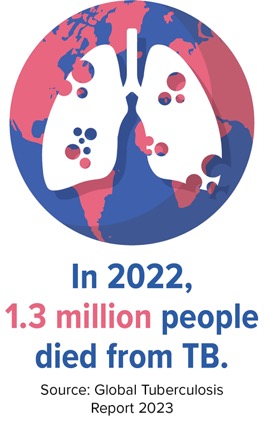
Tuberculosis (TB) is one of the leading causes of death among people with HIV globally. According to the latest data from the WHO, the number of deaths in 2022 attributed to TB globally was 1.30 million.1 People with HIV are also at a higher risk for acquisition of TB and are more likely to become ill compared with HIV-negative individuals. Data from the WHO estimate that individuals with HIV and latent TB are 18 (15-21) times more likely to reactivate TB compared with those without HIV.2
In 2021, estimates predict that approximately 703,000 people with HIV had TB, of which only half of those received an actual TB diagnosis. There were approximately 187,000 TB-related deaths in people with HIV that year. When looking at the United States specifically, a significantly higher mortality rate is observed in individuals with TB and HIV, with a mortality rate of 11.8% compared with 6.7% in those with TB who are HIV-negative.3 The WHO reports that as of 2019, approximately 50% of individuals with HIV currently on antiretroviral therapy (ART) and diagnosed with latent TB have received treatment for latent TB. Conversely, reports demonstrate that only 41% of people with HIV and latent TB are receiving ART.2
Data demonstrate that individuals with HIV have a 3 times higher likelihood of mortality while receiving TB treatment compared with those without HIV.2 These statistics are rather alarming, considering the increased mortality risk in people with HIV who have latent TB compared with those with latent TB only. The data above highlight the importance of ensuring increased access to ART, improving access to high-quality TB screening, and expanding the availability of treatment for latent TB.
This review provides an overview of treatment recommendations in individuals with HIV and latent TB, focusing on important considerations that need to be considered in those on ART.
Diagnostic Considerations
The Department of Health and Human Services (DHHS) guidelines recommend evaluation for latent TB in all individuals at the time of HIV diagnosis regardless of TB risk factors.3 The compliance with these recommendations is unfortunately low; 47% to 69% of individuals in care completed initial screening at the time of HIV diagnosis, with even less initiating treatment for latent TB (42%).3
Guidelines recommend the use of either a tuberculin skin test (TST) or interferon-gamma release assays (IGRA). However, they note these tests have limited accuracy particularly in individuals with significant immunodeficiencies (CD4 count <200 cells/mm3). Both the TST and IGRA have limitations in their testing, with no direct comparisons of the 2 different testing modalities on screening, detection, positivity, etc, and are considered appropriate for latent TB screening in people with HIV.3
For those who test negative at initial screening and have a CD4 count of less than 200 cells/mm3, repeat testing should be performed after initiation of ART and until subsequent lab results demonstrate a CD4 count of more than 200 cells/mm3. Annual testing is not recommended unless individuals had a prior negative test result and are at high risk for infection due to continued exposure to people with active TB infection.3
People with HIV and a positive TST or IGRA should be evaluated for active TB. Most with active TB will be symptomatic (ie, cough, weight loss, fever, lymphadenopathy); however, this may not always be the case. Of note, for those individuals with a CD4 count less than 200 cells/mm3 and TB-related symptoms, a negative TST or IGRA does not rule out active TB.
Latent TB Treatment Recommendations
Latent TB treatment is indicated in people who have a positive screening test or those who have close contact with an individual with active TB regardless of current CD4 count and results of screening test. Recommendations per the DHHS guidelines for treatment of latent TB are shown in Table 1 with preferred regimens listed first followed by alternative regimens.3
| Table 1. DHHS Latent TB Treatment Recommendations | ||||
| Regimen | Adult dosing | Duration | Clinical considerations | |
|---|---|---|---|---|
| Preferred therapy | ||||
| 3HP | INH + RPT + pyridoxine |
| 12 wk | Recommended for virally suppressed patients |
| 3HR | INH + rifampin + pyridoxine |
| 3 mo | Beware of drug–drug interactions |
| Alternative therapy | ||||
| INH + pyridoxine |
| 6-9 mo | Rates of treatment completion are suboptimal due to long duration | |
| 4R | rifampin | 600 mg PO daily | 4 mo | Beware of drug–drug interactions |
| 1HP | INH + RPT + pyridoxine |
| 4 wk | |
| 1HP, 1-mo course of daily isoniazid plus rifapentine; 3HP, 3-mo course of weekly isoniazid plus rifapentine; 3HR, isoniazid and rifampin; 4R, 4-mo course of daily rifampin; INH, isoniazid; PO, by mouth; RPT, rifapentine; TB, tuberculosis. Based on reference 1. | ||||
For individuals exposed to drug-resistant TB, providers should consult public health authorities and TB experts to determine the most appropriate treatment regimen for latent TB treatment.
Clinical Considerations
Initiation of ART should not be deferred, as the treatments of latent TB and HIV act independent of each other to help decrease the risk of developing active TB. When using antimicrobials for treatment of latent TB with ART, one important consideration when choosing the appropriate regimen is the presence or possibility of drug–drug interactions.4 Table 2 summarizes some important considerations regarding drug–drug interactions with ARTs and antimicrobial agents used for treatment of latent TB. Note this table is not all inclusive, and drug–drug interactions should be assessed for each patient based on the current medications they are receiving.
| Table 2. Drug–Drug Interactions With ART and Antimicrobial Agents Used for Latent TBa | ||
| Anti-tuberculosis antimicrobial | ARVs | Recommendations |
|---|---|---|
| Isoniazid | All ARVs | No dosage adjustment necessary for INH or ARVs |
| Rifampin | NRTIs |
|
| Efavirenz | No dosage adjustment necessary for rifampin | |
| Dolutegravir-raltegravir |
| |
| Bictegravir-emtricitabine-TAF | Do not coadminister | |
| PIs | Do not coadminister | |
| Maraviroc without strong CYP3A4 inhibitor |
| |
| Rifapentine | Efavirenz | Rifapentine 1,200 mg/day for individuals =40 kg |
| NRTIs |
| |
| Raltegravir |
| |
| Dolutegravir |
| |
| Bictegravir-emtricitabine-TAF | Do not coadminister | |
| PIs | Do not coadminister | |
| Pyrazinamide | All ARVs | No dosage adjustments needed |
| ART, antiretrovial therapy; ARVs, antiretrovirals; CYP3A4, cytochrome P450 3A4; NRTIs, nucleoside reverse transcriptase inhibitors; PIs, protease inhibitors; TAF, tenofovir alafenamide; TB, tuberculosis. a Drug–drug interaction chart is not all inclusive. Please check the patient’s entire medication list for drug–drug interactions. Liverpool HIV drug interaction checker is an excellent resource that can be used to assess for any potential drug–drug interactions and is available at https://www.hiv-druginteractions.org/checker. Based on reference 2. | ||
Conclusion
Individuals with HIV and latent TB are at a significantly higher risk of developing active TB, with a higher mortality rate compared with those who are HIV-negative with latent TB. This risk can be mitigated by increasing access to ART and diagnostic testing for latent TB, and increased access to latent TB treatment. When evaluating these patients and prescribing treatment for latent TB, it is essential to review the patient’s medication list for not only their antiretroviral regimen but any other mediations they may be taking, including over-the-counter drugs and herbal supplements.3,4 With collaboration among the various healthcare providers involved in the care of the patient including their HIV provider as well as a pharmacist, patients can safely receive treatment for latent TB and HIV, thereby decreasing their risk of developing active TB.
References
- WHO. Global Tuberculosis Report 2023. Accessed November 27, 2024. https://bit.ly/4eKankN-IDSE
- WHO. Tuberculosis & HIV. Accessed October 15, 2024. https://www.who.int/teams/global-hiv-hepatitis-and-stis-programmes/hiv/treatment/tuberculosis-hiv
- Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents With HIV. National Institutes of Health, CDC, HIV Medicine Association, and Infectious Diseases Society of America; 2024. Accessed November 27, 2024. https://bit.ly/3ZzVWf7-IDSE
- Liverpool HIV drug interaction checker. Accessed October 15, 2024. https://www.hiv-druginteractions.org/checker
Copyright © 2024 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.
Download to read this article in PDF document:![]() HIV and Latent TB: Opportunities to Optimize Treatment and Decrease Risk for Active TB
HIV and Latent TB: Opportunities to Optimize Treatment and Decrease Risk for Active TB