The fall season not only brings with it the cooler weather and changing color of the leaves but also is associated with an increased incidence of respiratory viral infections such as influenza, respiratory syncytial virus (RSV), and COVID-19. As of early September, the incidence of influenza and RSV is low/very low, respectively, while some areas of the country are seeing increased incidence of COVID-19. Despite the low activity of these viruses, all 3 are expected to see increased cases over the coming weeks to months with peak rates similar to the 2024-2025 season.1
People with HIV (PWH) may be more susceptible to infections including respiratory viruses because of disruptions in their immune function, particularly due to decreases in the CD4 T lymphocytes. Antiretroviral therapy (ART) has helped to lessen the effect HIV has on the individual’s immune system, especially in those who initiate early ART. Despite the initiation of ART, the immune system may not completely normalize, potentially making these individuals more susceptible to certain respiratory viruses. This article reviews the current epidemiology and recommendations for prevention as well as treatment for several common respiratory viruses including influenza, RSV, and COVID-19.2
Influenza
According to the Department of Health and Human Services (HHS) guidelines, there is varying information with some studies demonstrating an increased rate of hospitalization and mortality in PWH, whereas others have observed no increased risk. Individuals who are not currently on ART or with lower CD4+ counts appear to be at the highest risk.3
Prevention
One of the best modalities for prevention of influenza includes vaccination along with good hand hygiene and maintenance of daily healthy habits. Data have demonstrated that PWH who are on ART and have normal or near-normal CD4+ counts mount appropriate responses to influenza vaccinations; however, those with advanced disease or low CD4+ counts may not develop the same protection. Published literature has demonstrated the effectiveness and immunogenicity of the influenza vaccine in PWH. Fine and colleagues demonstrated that vaccination was the most effective method for preventing influenza-like illnesses (ILIs) during an outbreak at a facility in PWH with CD4+ counts greater than 100 cells/mm3 and HIV-1 RNA less than 30,000 copies.4 Similarly, data from a randomized clinical trial demonstrated that influenza vaccination was efficacious in prevention of laboratory-confirmed influenza infection in PWH who were naive to ART.5 Recommendations from the CDC and Advisory Committee on Immunization Practices (ACIP) advise that individuals 6 months of age and older should receive the age-appropriate annual influenza vaccine as long as no contraindications exist.6 See Table 1 for flu vaccination recommendations.3,6
| Table 1. Recommended Influenza Vaccinations 2025-2026 | |||
| Influenza vaccinations | Indication | Clinical considerations | |
|---|---|---|---|
|
|
| |
| HD-IIV, high-dose inactivated influenza vaccine; IIV, Inactivated influenza vaccine; LAIV, live-attenuated influenza vaccine; RIV, recombinant hemagglutinin vaccine. Based on references 3 and 6. | |||
Treatment
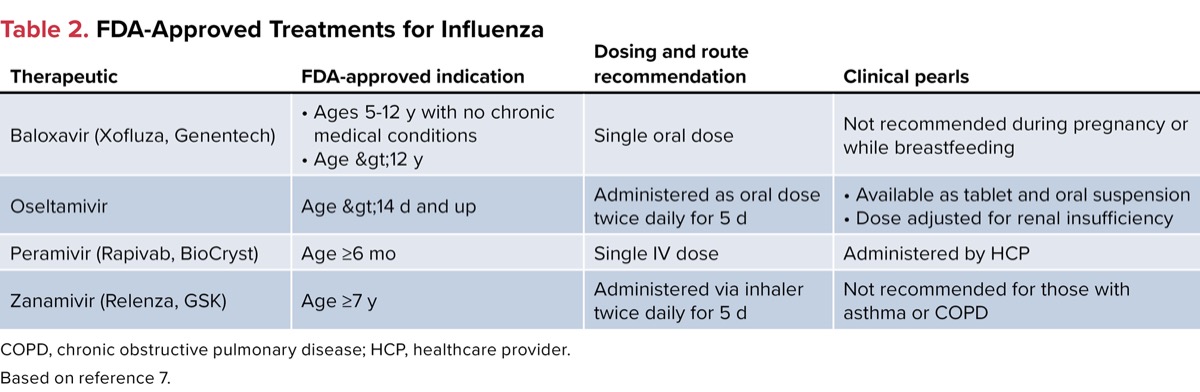
For those who develop ILI and test positive for influenza A or B, prompt treatment is indicated. Recommended treatments are listed in Table 2 and should be initiated in those with ILIs, ideally within 48 hours of symptom onset. Initiation of treatment should not be delayed while waiting for laboratory confirmation, especially in those with severe influenza and/or at high risk for complications.7
| Table 2. FDA-Approved Treatments for Influenza COPD, chronic obstructive pulmonary disease; HCP, healthcare provider. Based on reference 7. | |||
| Therapeutic | FDA-approved indication | Dosing and route recommendation | Clinical pearls |
|---|---|---|---|
| Baloxavir (Xofluza, Genentech) |
| Single oral dose | Not recommended during pregnancy or while breastfeeding |
| Oseltamivir | Age >14 d and up | Administered as oral dose twice daily for 5 d |
|
| Peramivir (Rapivab, BioCryst) | Age =6 mo | Single IV dose | Administered by HCP |
| Zanamivir (Relenza, GSK) | Age =7 y | Administered via inhaler twice daily for 5 d | Not recommended for those with asthma or COPD |
Respiratory Syncytial Virus
RSV is a common respiratory illness observed in children and older adults that can result in significant disease requiring hospitalization and significant mortality. The CDC estimates that out of the 3.6 million to 6.5 million outpatient visits for RSV, 190,000 to 350,000 of those individuals were hospitalized with 10,000 to 23,000 deaths during the 2024-2025 season.8 Almeida and colleagues conducted a systematic review to determine whether there was a difference in the incidence and severity of RSV in PWH.9 After a review of the literature, the authors identified 36 articles that were included in the review and demonstrated there was no significant difference in the incidence or severity of infection in adults with HIV. The data demonstrated that children with HIV and those exposed to HIV but noninfected had a higher incidence of disease severity as well as case fatality rate compared with those who were not exposed to HIV.
Prevention
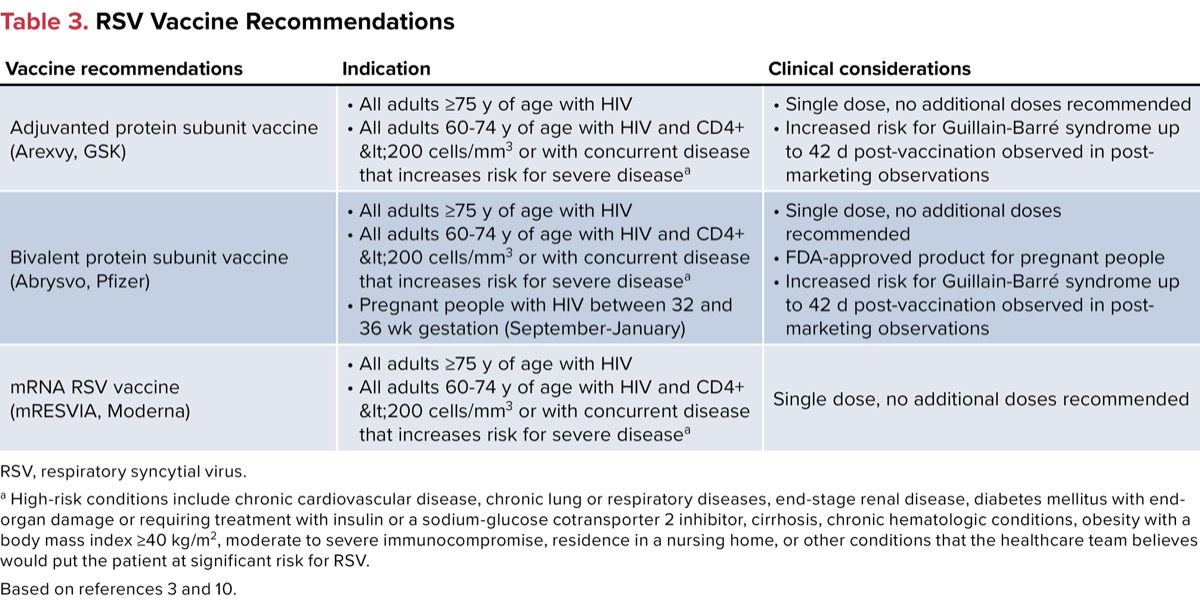
The first RSV vaccines were approved by the FDA in May 2023 for adults 60 years of age and older. With the approval of these vaccines, ACIP updated its recommendations to include RSV vaccination in adults 75 years of age and older and those 60 to 74 years who are at high risk for severe disease. Risk for severe disease includes conditions such as chronic cardiovascular disease, chronic lung or respiratory diseases, end-stage renal disease, diabetes mellitus with end-organ damage or requiring treatment with insulin or a sodium-glucose cotransporter 2 inhibitor, cirrhosis, chronic hematologic conditions, obesity with a body mass index of 40 kg/m2 or higher, moderate to severe immunocompromise, residence in a nursing home, or other conditions that the healthcare team believes would put the patient at significant risk for RSV. PWH are considered at risk if they meet any of the following criteria: having a CD4+ count lower than 200 cells/mm3, having an AIDS-defining illness (or have had in the past), or are currently not on ART.3,10 See Table 3 for RSV vaccination recommendations.3,10
| Table 3. RSV Vaccine Recommendations | ||
| Vaccine recommendations | Indication | Clinical considerations |
|---|---|---|
| Adjuvanted protein subunit vaccine (Arexvy, GSK) |
|
|
| Bivalent protein subunit vaccine (Abrysvo, Pfizer) |
|
|
| mRNA RSV vaccine (mRESVIA, Moderna) |
| Single dose, no additional doses recommended |
| RSV, respiratory syncytial virus. a High-risk conditions include chronic cardiovascular disease, chronic lung or respiratory diseases, end-stage renal disease, diabetes mellitus with end-organ damage or requiring treatment with insulin or a sodium-glucose cotransporter 2 inhibitor, cirrhosis, chronic hematologic conditions, obesity with a body mass index =40 kg/m2, moderate to severe immunocompromise, residence in a nursing home, or other conditions that the healthcare team believes would put the patient at significant risk for RSV. Based on references 3 and 10. | ||
Treatment
Currently there are no FDA-approved treatments for RSV in anyone older than 12 months. For those with RSV, the recommendation is to manage symptoms with over-the-counter medications. Pain relievers such as acetaminophen or ibuprofen may be used to help manage fever and/or pain, if needed. People with RSV should ensure they are continuing to take in enough fluids to prevent possible dehydration. Healthcare providers can assist their patients by providing recommendations to help manage the symptoms associated with RSV and recommend a higher level of care as necessary based on the individual’s symptoms and risk for progression to severe disease.
COVID-19
Since the first identified cases of severe acute respiratory syndrome coronavirus 2—or SARS-CoV-2, the virus that causes COVID-19—evidence for prevention and treatment has been evolving constantly. COVID-19 was associated with high rates of hospitalizations and mortality with marked changes over the last few years, due to the different variants associated with lower virulence.
PWH may be at increased risk due to their compromised immune systems as well as other comorbidities, which are seen in the aging population that are associated with a higher risk for severe disease.12 Several authorities have issued guidance regarding prevention, while the Infectious Diseases Society of America (IDSA) offers guidance for treatment of COVID-19.3,11
Prevention
Similarly to the influenza vaccines, COVID-19 vaccines are updated annually based on the currently circulating variants. HHS currently recommends all individuals older than 5 years of age with HIV receive a COVID-19 vaccine regardless of their current HIV-1 RNA or CD4+ counts. Individuals with CD4+ counts under 200 cells/mm3 or those with advanced HIV may be eligible for an additional dose 8 weeks or more after their last COVID-19 vaccine.3
In addition to vaccinations, the FDA has authorized pemivibart (Pemgarda, Invivyd) via an emergency use authorization (EUA) in individuals who are considered moderately or severely immunosuppressed due to medical conditions or treatments.12 Those with HIV and CD4+ counts lower than 200 cells/mm3 are included in the approval for pemivibart. The IDSA recommends use of pemivibart in those individuals who meet the EUA eligibility where the COVID-19 variants are confirmed susceptible to pemivibart.13
Treatment
Regarding treatment of COVID-19 in PWH, data have been evolving, with several agents currently having received FDA approval (EUA or full approval) for treatment of COVID-19 including molnupiravir (Lagevrio, Merck),14 nirmatrelvir+ritonavir (Paxlovid, Pfizer),15 and remdesivir (Veklury, Gilead).16 For more information on COVID-19 treatment considerations, please see the cover story “How Should We Be Managing ‘COVID-25’?”.
Discussion
Vaccination can play a key component in prevention as well as in decreasing the severity of respiratory viral infections. All individuals, especially those with chronic conditions, are at higher risk for influenza. Those 65 years of age and older also are at higher risk for RSV. The CDC, ACIP, and HHS recommend that all people, regardless of HIV status, receive the seasonal influenza vaccine and those who meet the criteria be vaccinated for RSV as well.
With the changing seasons, as healthcare providers, we can help educate our patients and advocate for them to decrease their risk for acquiring these viral infections—and decrease the severity of disease if they do end up with either viral infection. For those who test positive for influenza, prompt treatment is indicated to help decrease the severity and duration of their symptoms. For the most updated information regarding vaccination recommendations, refer to the CDC.gov webpage, recommendations from ACIP, and recommendations from professional medical societies and US states. The IDSA and several other associations, as well as state governments, have made statements and recommendations that differ from ACIP.11,13,17,18
References
- Respiratory illnesses data channel. Respiratory illnesses. CDC. September 5, 2025. Accessed September 5, 2025. bit.ly/CDC-cold
- Gispen F, et al. Curr HIV/AIDS Rep. 2025;22(1):17.
- Panel on Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV. Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV. Updated July 14, 2025. Accessed September 3, 2025. bit.ly/HIV-infection
- Fine AD, et al. Clin Infect Dis. 2001;32(12):1784-1791.
- Cohen C, et al. Emerg Infect Dis. 2013;19(11):1766-1774.
- Grohskopf LA, et al. MMWR Morb Mortal Wkly Rep. 2025;74(32):500-507.
- Treatment of flu. Influenza (flu). CDC. Updated September 9, 2024. Accessed September 9, 2025. bit.ly/CDC-FLU
- Preliminary estimates of RSV burden for 2024-2025. Respiratory syncytial virus infection (RSV). CDC. July 8, 2025. Accessed September 3, 2025. bit.ly/CDC_RSV
- Almeida A, et al. Pathogens. 2024;13(9):802.
- Britton A. et al. MMWR Morb Mortal Wkly Rep. 2024;73(32):696-702.
- Nadig N, et al. Clin Infect Dis. Published online September 9, 2025. doi:10.1093/cid/ciaf354
- Pemgarda. Emergency use authorization. Invivyd. August 26, 2024. Accessed September 15, 2025. bit.ly/pemgarda_EUA
- Bhimraj A, et al. Clin Infect Dis. Published online August 4, 2025. doi:10.1093/cid/ciaf424
- Lagevrio. Fact sheet for healthcare providers: emergency use authorization for Lagevrio. Merck. June 2024. Accessed September 15, 2025. bit.ly/lagevrio
- Paxlovid. Prescribing information. Pfizer; February 12, 2025. Accessed September 15, 2025. bit.ly/paxlovid_PI
- Veklury. Prescribing information. Gilead; October 22, 2020. Accessed September 15, 2025. bit.ly/Veklury_PI
- Shaw G. Infectious Disease Special Edition. Updated September 10, 2025. Accessed September 11, 2025.
- FDA’s narrow COVID-19 vaccine label ignores science and puts millions of lives at risk. IDSA. Updated August 27, 2025. Accessed September 11, 2025. bit.ly/IDSA-COVID
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Respiratory Virus Season: Prevention and Treatment Considerations in People With HIV
Respiratory Virus Season: Prevention and Treatment Considerations in People With HIV