Infective endocarditis (IE), a serious infection involving the endocardium and heart valves, is associated with a high degree of illness and mortality, and has an annual incidence that has continued to increase throughout the United States.1-3 Risk factors for developing IE include prior IE, the presence of a prosthetic valve or implanted device, structural or congenital heart disease, and history of invasive procedures. People who inject drugs are also at risk for IE.2,4 Importantly, IE has an array of manifestations and sequelae, and can lead to a myriad of intracardiac and extracardiac complications if not identified early and appropriately treated.1 Furthermore, IE is associated with high recurrence and mortality rates despite appropriate surgical intervention and/or antimicrobial therapy, which further emphasizes the importance and clinical relevance within the infectious diseases and cardiology communities.3,5
IE requires the simultaneous occurrence of several independent factors, including the alteration of the cardiac valve surface to create a suitable site for microorganism attachment and colonization, a bloodstream infection with an organism capable of attaching to valve tissue, and the creation of a vegetation that is a protective mass for the proliferating organism consisting of molecules such as fibrin and platelets.6 Notably, IE can be broadly classified as being associated with either a native or prosthetic valve, with prosthetic valve IE being associated with worse outcomes.2,7 Although a wide variety of pathogens can cause IE, predominant causative organisms have been reported to be of the Enterococcus genus.8,9 The most common enterococci associated with infection include E faecalis and E faecium, with E faecalis being most prevalent but E faecium being associated with more drug resistance to agents, such as ampicillin and vancomycin.10
Despite IE being a common infection that is associated with a high degree of morbidity and mortality, treatment recommendations for gram-positive IE have evolved little over the past few decades.2,11 This report aims to review antimicrobial treatment principles for IE and summarize the available evidence for the adjunctive use of aminoglycosides and beta-lactams for the treatment of enterococcal IE.
Therapeutic Principles For Infective Endocarditis
Empiric antimicrobial regimens for IE should be chosen based on patient-specific risk factors (ie, intolerance) and local pathogen epidemiology, which may differ based on geography, while definitive therapy should be chosen based on the identified etiology, minimum inhibitory concentration (MIC), and valve origin of infection (native vs prosthetic valve).2,11 Importantly, IE can pose a variety of treatment challenges, as infected vegetations classically are considered to be associated with a high bacterial inoculum, low pathogen metabolic activity, and a slow rate of microorganism growth within biofilms.2,12 The inoculum effect is a phenomenon in which commonly used antibiotics to treat bacterial IE, such as the beta-lactams and vancomycin, are less active against highly dense populations of bacteria.2,13-14 This may be due, in part, to the stationary growth-phase conditions of bacteria within high-inoculum vegetations that reduce the likelihood that cell wall–active antibiotics (beta-lactams, vancomycin) are optimally effective.15,16 Notably, other antibiotics with different mechanisms of action have been shown to be less affected by these conditions.15-17 Furthermore, it is important to note these high bacterial densities (~10^8-10^11 colony-forming units [CFU] per 1 g of tissue) can lead to MICs at the site of infection that are higher than reported with in vitro susceptibility testing that uses a standard inoculum (~10^5.5 CFU/mL).2 In addition, antimicrobials may fail to eradicate the infection because of reduced active concentrations due to increased protein binding, fluctuations of antibiotic penetration into the vegetations and unique antimicrobial pharmacokinetic/pharmacodynamic properties.2
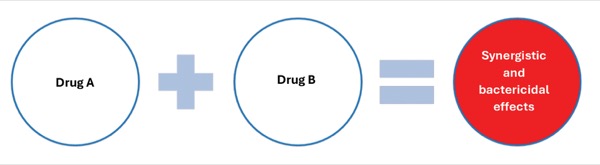
These phenomena, among others, have led to numerous investigations as well as the American Heart Association/Infectious Diseases Society of America (AHA/IDSA) endocarditis guideline recommendations that endorse the use of bactericidal antibiotics for IE to sterilize these vegetations with high bacterial densities.2,18-20 Furthermore, antimicrobial combinations have been studied throughout in vitro and in vivo models that appear to produce better synergistic and bactericidal effects compared with monotherapy in enterococci and other gram-positive isolates.13,21-26 The concept of synergism, which is the notion that 2 antimicrobials together exhibit greater activity than either agent alone, is shown in Figure 1.27 Important to highlight, most of the recommendations regarding combination therapy provided in the 2015 AHA/IDSA endocarditis guidelines are based on level B and C evidence and have limited clinical validation.2
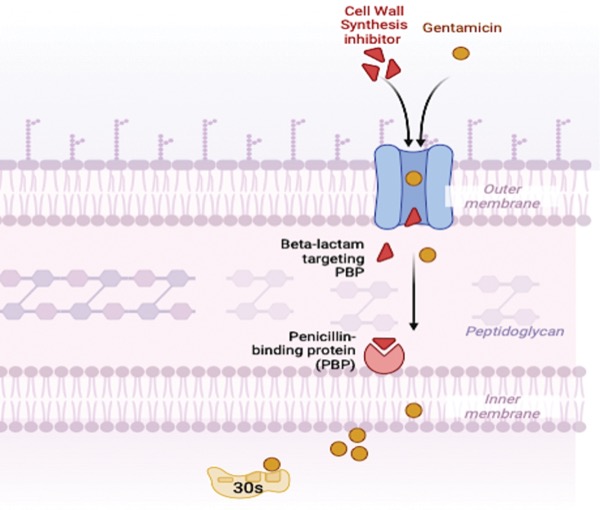
Enterococcus spp are a frequent cause of IE, with E faecalis being the most common.2,8-9 The AHA/IDSA endocarditis guidelines recommend ampicillin in addition to gentamicin or ceftriaxone for IE caused by enterococci susceptible to penicillin.2 Data on beta-lactam monotherapy for IE caused by ampicillin-susceptible enterococci are limited, and combination therapy with a beta-lactam and an aminoglycoside has been the historical standard-of-care therapy based on early case reports that showed positive outcomes following in vitro analyses displaying combination therapy as a necessity to produce bactericidal effects.21,28-32 Notably, treatment durations were short, and doses were low in the early studies of IE that described beta-lactam monotherapy failures. Importantly, the enterococci have been shown to be relatively impermeable to aminoglycosides.33 The theory behind the synergistic effects that have been shown with beta-lactam and aminoglycoside combination therapy relies on the cell wall–active beta-lactam increasing the permeability of the enterococcal cell by cell wall inhibition, facilitating the diffusion of the aminoglycoside into the site of the ribosomal target to exert its bactericidal effects (Figure 2).2,22,34
The historical data supporting combination therapy with a beta-lactam and an aminoglycoside have likely been overly relied upon by clinicians and have significant limitations, creating a sense of confidence about the perceived benefit that may be inaccurate.11 As one example from the mid-1950s, Geraci and colleagues described 33 cases of IE caused by enterococci, in which 19 patients received penicillin monotherapy and 14 patients received combination therapy with penicillin plus streptomycin.31 Overall, the rates of treatment cures were reported to be much higher with combination therapy (monotherapy, 26%; combination therapy, 71%). However, there are important limitations that are worth discussing. First and foremost, clinical cure was not defined, the duration of follow-up was not listed for all patients, and many patients received suboptimal doses of penicillin and treatment durations. Second, other patients that were classified as beta-lactam monotherapy failures did not receive a beta-lactam at all, received therapy for a brief duration due to allergic reactions, or had mixed infections.31 Another major aspect to consider in many of these earlier studies is that contemporary features of clinical care were not available. Therefore, relying on historical descriptive case reports such as these as the backbone of evidence for current practice is problematic.11 Although a more recent study did show higher rates of relapse and mortality in patients receiving beta-lactam monotherapy compared with those receiving combination therapy with an aminoglycoside, the beta-lactam monotherapy group had only 9 patients.35
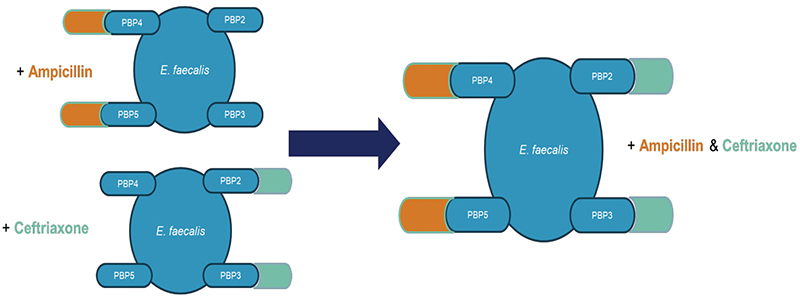
Given the extensive clinical history of using combination therapy for IE caused by Enterococcus spp, it has been tempting within the clinical realm to substitute beta-lactam–aminoglycoside combination therapy with another combination in place of beta-lactam monotherapy in ampicillin-susceptible isolates.11 The combination of ampicillin and ceftriaxone is an alternative AHA/IDSA endocarditis guidelines–recommended therapy for IE caused by enterococci.2 The history of this dual beta-lactam therapy stems from an evaluation conducted by Mainardi and colleagues in 1995, in which a synergistic effect was observed in vitro between amoxicillin and cefotaxime against various isolates of E faecalis.23 Following this report, the combination of ampicillin and ceftriaxone has also been shown to produce a bactericidal synergistic effect by the complementary saturation of multiple penicillin-binding proteins (PBPs); ampicillin binds to PBP 4 and PBP 5 while ceftriaxone binds to PBP 2 and PBP 3, leading to increased bacterial kill (Figure 3).24,25
Mirna and colleagues conducted a systematic review and meta-analysis to evaluate if ampicillin plus ceftriaxone was noninferior to ampicillin/amoxicillin plus gentamicin.36 The authors concluded that therapy with ampicillin plus ceftriaxone was noninferior to ampicillin/amoxicillin plus gentamicin, as there were no statistically significant differences noted in treatment failure, relapse, in-hospital mortality, and 3-month mortality. Furthermore, they found that ampicillin plus ceftriaxone was safer than therapy with ampicillin/amoxicillin plus gentamicin, as there were a lower odds of nephrotoxicity (odds ratio [OR], 0.45 [0.26-0.77]) and drug withdrawal due to side effects (OR, 0.11 [0.03-0.46]).36 Following publication of this meta-analysis, Danneels and colleagues conducted a multicenter, retrospective study of patients diagnosed with IE caused by E faecalis and found no differences in 1-year relapse in patients treated with dual beta-lactam therapy and with beta-lactam–aminoglycoside combination therapy.35
The proposed synergistic benefits associated with using beta-lactams with adjunctive aminoglycosides for treating enterococcal IE have favorable in vitro activity; however, there is limited evidence to support their benefit on patient outcomes.11,21-22,26 Furthermore, there are no persuasive data to suggest that dual beta-lactam therapy is superior to beta-lactam monotherapy.11 Although the current AHA/IDSA guidelines consider ampicillin plus ceftriaxone an alternative to ampicillin plus gentamicin, studies have found ampicillin plus ceftriaxone to be noninferior to ampicillin plus gentamicin for the treatment of IE caused by E faecalis.36 Notably, the combination of ampicillin plus ceftriaxone was also found to be safer and more tolerable than ampicillin plus gentamicin.36
Many clinicians opt to avoid the routine use of aminoglycosides for IE caused by Enterococcus spp because their benefits are uncertain, resistance is increasing, the risks for adverse effects are larger, and they have displayed similar rates of clinical outcomes compared with dual beta-lactam therapy.11,35-36 It is imperative to consider the potential benefits and risks, as well as acknowledge the limitations of the available evidence associated with the addition of adjunctive agents, when treating patients to optimize outcomes and minimize adverse events.11
References
- Yallowitz AW, Decker LC. Infectious endocarditis. StatPearls [internet]. StatPearls Publishing; 2024.
- Baddour LM, Wilson WR, Bayer AS, et al. Infective endocarditis in adults: diagnosis, antimicrobial therapy, and management of complications: a scientific statement for healthcare professionals from the American Heart Association. Circulation. 2015;132(15):1435-1486.
- Taduru SS. 30-year trends of incidence and mortality of infective endocarditis in the United States–unveiling the age- and gender-related and regional disparities. Am J Cardiol. 2023:204:421-422.
- Pierce D, Calkins BC, Thornton K. Infectious endocarditis: diagnosis and treatment. Am Fam Physician. 2012;85(10):981-986.
- Alkhouli M, Alqahtani F, Alhajji M, et al. Clinical and economic burden of hospitalizations for infective endocarditis in the United States. Mayo Clin Proc. 2020;95(5):858-866.
- Holland TL, Baddour LM, Bayer AS, et al. Infective endocarditis. Nat Rev Dis Primers. 2016;2:16059.
- Romano G, Carozza A, Della Corte A, et al. Native vs. primary prosthetic valve endocarditis: comparison of clinical features and long-term outcome in 353 patients. J Heart Valve Dis. 2004;13(2):200-208.
- Habib G, Erba PA, Iung B, et al. Clinical presentation, aetiology and outcome of infective endocarditis. Results of the ESC-EORP EURO-ENDO (European infective endocarditis) registry: a prospective cohort study. Eur Heart J. 2019;40(39):3222-3232.
- Cresti A, Chiavarelli M, Scalese M, et al. Epidemiological and mortality trends in infective endocarditis, a 17-year population-based prospective study. Cardiovasc Diagn Ther. 2017;7(1):27-35.
- García-Solache M, Rice LB. The Enterococcus: a model of adaptability to its environment. Clin Microbiol Rev. 2019;32(2):e00058-18.
- McDonald EG, Aggrey G, Aslan AT, et al. Guidelines for diagnosis and management of infective endocarditis in adults: a WikiGuidelines Group consensus statement. JAMA Netw Open. 2023;6(7):e2326366.
- Durack DT, Beeson PB. Experimental bacterial endocarditis. II. Survival of a bacteria in endocardial vegetations. Br J Exp Pathol. 2917;53(1):50-53.
- LaPlante KL, Rybak MJ. Impact of high-inoculum Staphylococcus aureus on the activities of nafcillin, vancomycin, linezolid, daptomycin, alone and in combination with gentamicin, in an in vitro pharmacodynamic model. Antimicrob Agents Chemother. 2004;48(12):4665-4672.
- Rose WE, Leonard SN, Rossi KL, et al. Impact of inoculum size and heterogeneous vancomycin-intermediate Staphylococcus aureus (hVISA) on vancomycin activity and emergence of VISA in an in vitro pharmacodynamic model. Antimicrob Agents Chemother. 2009;53(2):805-807.
- Eng RH, Padberg FT, Smith SM, et al. Bactericidal effects of antibiotics on slowly growing and nongrowing bacteria. Antimicrob Agents Chemother. 1991;35(9):1824-1828.
- Stevens DL, Yan S, Bryant AE. Penicillin-binding protein expression at different growth stages determines penicillin efficacy in vitro and in vivo: an explanation for the inoculum effect. J Infect Dis. 1993;167(6):1401-1405.
- Mizunga S, Kamiyama T, Fukuda Y, et al. Influence of inoculum size of Staphylococcus aureus and Pseudomonas aeruginosa on in vitro activities and in vivo efficacy of fluoroquinolones and carbapenems. J Antimicrob Chemother. 2005;56(1):91-96.
- Hunter TH. Speculations on the mechanism of cure of bacterial endocarditis. J Am Med Assoc. 1950;144(7):524-527.
- Schuler G. Antibiotic therapy of infectious endocarditis (when, with what drug, how long?). Z Kardiol. 1994;83(1):2-8.
- Finberg RW, Moellering RC, Tally FP, et al. The importance of bactericidal drugs: future directions in infectious disease. Clin Infect Dis. 2004;39(9):1314-1320.
- Jawetz E, Gunnison JB, Coleman VR. Combined action of penicillin with streptomycin and chloromycetin on enterococci in vitro. Am J Med. 1950;8(4):532.
- Herrera-Hidalgo L, Fernández-Rubio B, Luque-Márques R, et al. Treatment of Enterococcus faecalis infective endocarditis: a continuing challenge. Antibiotics (Basel). 2023;12(4):704.
- Mainardi JL, Gutmann L, Acar JF, et al. Synergistic effect of amoxicillin and cefotaxime against Enterococcus faecalis. Antimicrob Agents Chemother. 1995;39(9):1984-1987.
- Cusamano JA, Khan R, Shah Z, et al. Penicillin plus ceftriaxone versus ampicillin plus ceftriaxone synergistic potential against clinical Enterococcus faecalis blood isolates. Microbiol Spectr. 2022;10(4):e0062122.
- Jimenez-Toro I, Rodriguez CA, Zuluaga AF, et al. A new pharmacodynamic approach to study antibiotic combinations against enterococci in vivo: application to ampicillin plus ceftriaxone. PLoS One. 2020;15(12):e0243365.
- Beganovic M, Luther MK, Rice LB, et al. A review of combination antimicrobial therapy for Enterococcus faecalis bloodstream infections and infective endocarditis. Clin Infect Dis. 2018;67(2):303-309.
- Doern CD. When does 2 plus 2 equal 5? A review of antimicrobial synergy testing. J Clin Microbiol. 2014;52(12):4124-4128.
- Hein GE, Berg BM. Recovery from subacute bacterial endocarditis; report of two cases. Am Heart J. 1949;38(3):433-437.
- Harris R. Subacute bacterial endocarditis; report of enterococcal infection treated with penicillin. Bulletin NY Med Coll. 1945;8:61-63.
- Robbins WC, Tompsett R. Treatment of enterococcal endocarditis and bacteremia; results of combined therapy with penicillin and streptomycin. Am J Med. 1951;10(3):278-299.
- Geraci JE, Martin WJ. Antibiotic therapy of bacterial endocarditis. VI. Subacute enterococcal endocarditis; clinical pathologic and therapeutic consideration of 33 cases. Circulation. 1954;10(2):173-194.
- Griffith GC, Levinson DC. Subacute bacterial endocarditis; a report on 57 patients treated with massive doses of penicillin. Calif Med. 1949;71(6):403-408.
- Gutiérrez J, Hoyos A, Piedrola G. High-level aminoglycoside resistance in enterococci. Ann Biol Clin (Paris). 1992;50(10-11):671-674.
- Moellering Jr RC, Weinberg AN. Studies on antibiotic synergism against enterococci. II. Effect of various antibiotics on the uptake of 14 C-labeled streptomycin by enterococci. J Clin Invest. 1971;50(12):2580-2584.
- Danneels P, Hamel J-F, Picard L, et al. Impact of Enterococcus faecalis endocarditis treatment on risk of relapse. Clin Infect Dis. 2023; 76(2):281-290.
- Mirna M, Topf A, Schmutzler L, et al. Time to abandon ampicillin plus gentamicin in favour of ampicillin plus ceftriaxone in Enterococcus faecalis infective endocarditis? A meta-analysis of comparative trials. Clin Res Cardiol. 2022;111(10):1077-1086.
Copyright © 2024 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Synergism sparks energy, or does it? Antimicrobial Duos for Infective Endocarditis Caused by Enterococcus Species
Synergism sparks energy, or does it? Antimicrobial Duos for Infective Endocarditis Caused by Enterococcus Species