The nontuberculous mycobacteria (NTM) comprise more than 190 species and subspecies of bacteria that can cause disease in people of all ages.1,2 Of note, these organisms are ubiquitous in the environment (eg, water sources, soil), with certain species being opportunistic pathogens affecting those who have underlying health conditions and/or who are immunocompromised, including in people living with HIV/AIDS (PLWHA).1 The prevalence of NTM infections is growing throughout the United States and internationally, which reinforces the necessity of optimized prophylactic and therapeutic options.3,4
Mycobacterium avium complex (MAC) is a group of multiple species of NTM that are related, including the important human pathogens M. avium, M. intracellulare, and M. chimaera.2,5 These species of NTM can cause disease in both adults and children, including pulmonary infections, lymphadenitis, and skin and soft tissue infections.5 Of note, PLWHA are more prone to opportunistic infections (OIs) from MAC due to the ability of HIV to impair a person’s immune system by lowering the CD4+ T cells.6,7 Prior to efficacious antiretroviral therapy (ART), NTM (including MAC), were common OIs in children living with HIV/AIDS in the United States.8,9 Although the progress with ART certainly has reduced the risk for all OIs in PLWHA, medication access and adherence are crucial to reducing the morbidity and mortality associated with these OIs.10-12
The primary objective of this article is to review the data surrounding the management of MAC disease in children living with HIV/AIDS. There is a lack of high-quality evidence in children; thus, many treatment recommendations stem from extrapolations from adult data, which are described.
Epidemiology
Retrospective studies of small data sets published decades ago have confirmed MAC disease in children who are severely immunosuppressed.13,14 A study conducted by Rutstein and colleagues aimed to describe the incidence of MAC infections in children infected with HIV.13 Following a review of 70 infants and children with HIV, 10% developed disseminated MAC disease (10% of those with HIV; 18% of those with AIDS).13 Another similar study conducted by Lewis and colleagues reviewed 22 cases of MAC infections that occurred in nearly 200 children with an overall incidence of 11% and up to nearly 25% in individuals whose absolute CD4+ T-cell counts were less than 100 cells/mm3.14 Of these, all patients had disseminated MAC disease with the exception of 1 adolescent with only localized lymphadenitis.14 Of note, these studies were conducted before the availability of efficacious ART for children, and the risk for MAC disease is reduced in children living with HIV/AIDS who are on optimized ART.13,14
Treatment Recommendations
The Guidelines for the Prevention and Treatment of Opportunistic Infections in Children with and Exposed to HIV provide treatment recommendations for MAC disease that includes macrolide-based therapy, ethambutol, rifabutin, fluoroquinolones, and/or amikacin.8 To optimize therapy and prevent the emergence of resistance, at least 2 drugs are always recommended.8 An early study conducted by Wallace Jr and colleagues evaluated initial clarithromycin monotherapy in patients with MAC complex lung disease with 3 of 19 isolates (16%) developing highlevel clarithromycin resistance.15 Furthermore, the guidelines list considerations for 3 to 4 drugs in patients with severe or disseminated disease.8 Treatment recommendations within these guidelines for children living with HIV with MAC disease are shown in Table 1.
| Table 1. Treatment Recommendations for MAC Infections In Children With and Exposed to HIV | |
| First choice | Alternatives |
|---|---|
| Initial treatment (=2 medications): clarithromycin plus ethambutol; for severe disease, add rifabutin | If intolerant to clarithromycin, azithromycin plus ethambutol If administration of rifabutin is not ideal/possible and third medication is needed OR if a fourth drug is needed due to disseminated diseases and/or more severe symptoms, add:
|
| MAC, Mycobacterium avium complex. Based on reference 8. | |
Most of the evidence surrounding MAC treatment in children living with HIV/AIDS is limited to case reports and case series.16-18 Phongsamart and colleagues evaluated 169 children living with HIV, with 10 (6%) developing a MAC infection (7 with disseminated disease and 3 with pneumonia).18 Most (90%) were in the advanced stage of their HIV at the time of MAC disease diagnosis. The clinical outcomes of these children were poor, with a mortality rate of 60%. Although the authors stated that clarithromycin exhibited the least resistance, specific treatment regimens used for individual patients within this study were not found.18
A prospective, open-label, phase 3, randomized trial conducted by Benson and colleagues evaluated the efficacy and safety of clarithromycin plus ethambutol (C + E), clarithromycin plus rifabutin (C + R), or clarithromycin plus ethambutol plus rifabutin (C + E + R) for the treatment of disseminated MAC disease in adults living with AIDS.19 In total, 160 eligible patients with bacteremic MAC disease were randomized to the 3 regimens (C + E: 53 patients; C + R: 50 patients; C + E + R: 57 patients) for 48 weeks. After 12 weeks of therapy, there was no statistically significant difference in the proportion of patients with a complete microbiologic response among the treatment arms (C + E: 40%; C + R: 42%; C + E + R: 51%; P=0.454). In patients who had complete or partial responses, MAC disease relapse was significantly higher in those receiving C + R (24%) versus those receiving C + E + R (6%; P=0.027), and numerically higher in those receiving C + E (7%; P=0.057). In patients receiving C + E + R, improved survival was noted compared with those receiving C + E (hazard ratio [HR], 0.44; 95% CI, 0.23-0.83) and in those receiving C + R (HR, 0.49; 95% CI, 0.26-0.92).19 Based on this information, some experts recommend the addition of rifampin to macrolide plus ethambutol therapy, particularly in patients lacking optimized ART, in those with high mycobacterial bacterial burdens, and/or in severe or disseminated disease.8
There is insufficient evidence to recommend the use of clarithromycin over azithromycin for MAC disease therapy.8 Dunne and colleagues conducted a randomized, double-blind trial comparing azithromycin and clarithromycin in the treatment of disseminated MAC disease in adults with HIV.20 Patients were randomized to receive 250 mg of azithromycin daily (n=65 patients), 600 mg of azithromycin daily (n=91 patients), or 500 mg of clarithromycin twice daily (n=90 patients), each in combination with ethambutol, for 24 weeks. The low-dose azithromycin arm was dropped following an interim analysis that showed a lower rate of bloodstream clearance. After 24 weeks of therapy, there were no statistically significant differences in the likelihood of developing 2 consecutive bacterial cultures (azithromycin, 46% vs clarithromycin, 56%; P=0.24) or 1 negative bacterial culture (azithromycin, 59% vs clarithromycin, 61%; P=0.80). Furthermore, there were no differences in relapse rates (39% vs 27%; P=0.21) or mortality (69% vs 63%; HR, 1.1; 95% CI, 0.7-1.7) in those patients receiving 600 mg of azithromycin daily versus 500 mg of clarithromycin twice daily.20
Another randomized trial (open-label) conducted by Ward and colleagues compared 600 mg of azithromycin daily plus ethambutol (n=16 patients) with 500 mg of clarithromycin twice daily (n=21 patients) plus ethambutol in adult patients living with HIV with disseminated MAC from 10 university-affiliated Veterans Affairs medical centers.21 At the final visit, bacteremia clearance was seen more commonly in clarithromycin-treated patients versus those treated with azithromycin (86% vs 38%; P=0.007). The estimated median time to bacteremia clearance was faster in those patients treated with clarithromycin (4 weeks vs >16 weeks; P=0.0018).21 Due to these data, some experts prefer clarithromycin as the macrolide of choice and reserve azithromycin in pediatric patients who cannot tolerate clarithromycin, or in those with concerning drug–drug interactions with clarithromycin.8
Therapy Considerations
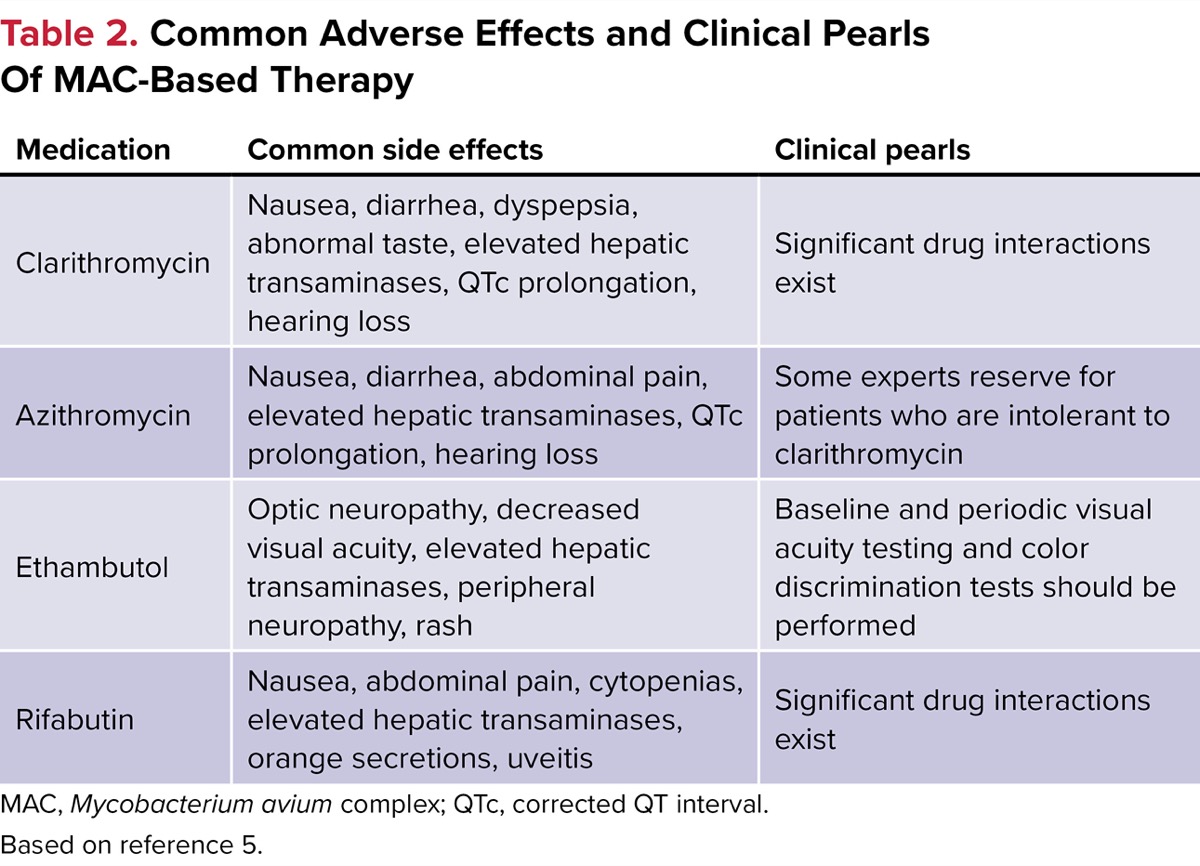
The antibiotics used most commonly to treat MAC disease have several considerations worth mentioning (Table 2).5 There are significant drug–drug interactions with C + R, and a medication interaction check is recommended for all patients receiving these therapies. Rifabutin is a cytochrome P450 3A4 (CYP3A4) inducer and substrate, while clarithromycin is a CYP3A4 inhibitor and substrate, as well as a P-glycoprotein inhibitor, which can lead to significant medication interactions. Furthermore, azithromycin and clarithromycin can induce corrected QT level prolongation, while notable side effects with rifabutin include hepatotoxicity and/or orange secretions. Finally, ethambutol can lead to optic neuropathy and reduced visual acuity, so visual testing and color discrimination testing are recommended.5
| Table 2. Common Adverse Effects and Clinical Pearls Of MAC-Based Therapy | ||
| Medication | Common side effects | Clinical pearls |
|---|---|---|
| Clarithromycin | Nausea, diarrhea, dyspepsia, abnormal taste, elevated hepatic transaminases, QTc prolongation, hearing loss | Significant drug interactions exist |
| Azithromycin | Nausea, diarrhea, abdominal pain, elevated hepatic transaminases, QTc prolongation, hearing loss | Some experts reserve for patients who are intolerant to clarithromycin |
| Ethambutol | Optic neuropathy, decreased visual acuity, elevated hepatic transaminases, peripheral neuropathy, rash | Baseline and periodic visual acuity testing and color discrimination tests should be performed |
| Rifabutin | Nausea, abdominal pain, cytopenias, elevated hepatic transaminases, orange secretions, uveitis | Significant drug interactions exist |
| MAC, Mycobacterium avium complex; QTc, corrected QT interval. Based on reference 5. | ||
Conclusion
MAC is a ubiquitous organism in the environment that has the potential to cause disease in PLWHA.1,2 Most of the evidence supporting treatment recommendations of MAC disease in children living with HIV stem from data that have been extrapolated from adults.8 Successful treatment commonly requires a prolonged course of at least 2 antibiotics that have the potential for notable adverse effects and drug–drug interactions.5 Given the lack of data in children and the fact that treatment outcomes for many patients are suboptimal, there is a need for new medications and treatment regimens, as well as pediatric-specific data, to help guide optimal therapy.5,8
References
- About nontuberculous mycobacteria (NTM) infections. CDC. April 15, 2024. Accessed June 5, 2025. cdc.gov/nontuberculous-mycobacteria/about/index.html
- Daley CL, Iaccarino JM, Lange C, et al. Treatment of nontuberculous mycobacterial pulmonary disease: an official ATS/ERS/ESCMID/IDSA clinical practice guideline. Eur Respir J. 2020;56(1):2000535.
- Prevots DR, Marras TK. Epidemiology of human pulmonary infection with nontuberculous mycobacteria: a review. Clin Chest Med. 2015;36(1):13-34.
- Stout JE, Koh WJ, Yew WW. Update on pulmonary disease due to non-tuberculous mycobacteria. Int J Infect Dis. 2016;45:123-134.
- Daley CL. Mycobacterium avium complex disease. Microbiol Spectr. 2017;5(2):10.1128/microbiolspec.tnmi7-0045-2017.
- About HIV. CDC. Last updated January 14, 2025. Accessed May 3, 2025. cdc.gov/hiv/about/index.html
- Zaremba ML. The essence of infection by opportunistic microorganisms. Przegl Epidemiol. 2001;55(suppl 3):91-99.
- Panel on Guidelines for the Prevention and Treatment of Opportunistic Infections in Children with and Exposed to HIV. Guidelines for the Prevention and Treatment of Opportunistic Infections in Children with and Exposed to HIV. National Institutes of Health, CDC, HIV Medicine Association, and Infectious Diseases Society of America; 2019. Accessed May 3, 2025. clinicalinfo.hiv.gov/en/guidelines/adult-and-adolescent-opportunistic-infection
- Nesheim SR, Kapogiannis BG, Soe MM, et al. Trends in opportunistic infections in the pre- and post-highly active antiretroviral therapy eras among HIV-infection children in the Perinatal AIDS Collaborative Transmission Study, 1986-2004. Pediatrics. 2007;120(1):100-109.
- Palmisano L, Vella S. A brief history of antiretroviral therapy of HIV infection: success and challenges. Ann Ist Super Sanita. 2011;47(1):44-48.
- Djawe K, Buchacz K, Hsu L, et al. Mortality risk after AIDS-defining opportunistic illness among HIV-infected persons—San Francisco, 1981-2012. J Infect Dis. 2015;212(9):1366-1375.
- Meng S, Tang Q, Xie Z, et al. Spectrum and mortality of opportunistic infections among HIV/AIDS patients in southwestern China. Eur J Clin Microbiol Dis. 2023;42(1):113-120.
- Rutstein RM, Cobb P, McGowan KL, et al. Mycobacterium avium intracellulare complex infection in HIV-infected children. AIDS. 1993;7(4):507-512.
- Lewis LL, Butler KM, Husson RN, et al. Defining the population of human immunodeficiency virus-infected children at risk for Mycobacterium avium-intracellulare infection. J Pediatrics. 1992;121(5 pt 1):677-683.
- Wallace RJ Jr, Brown BA, Griffith DE, et al. Initial clarithromycin monotherapy for Mycobacterium avium-intracellulare complex lung disease. Am J Respir Crit Care Med. 1994;149(5):1335-1341.
- Keller C, Kirkpatrick S, Lee K, et al. Disseminated Mycobacterium avium complex presenting as hematochezia in an infant with rapidly progressive acquired immunodeficiency syndrome. Pediatr Infect Dis J. 1996;15(8):713-715.
- Hariadi NI, Blackwood RA. Disseminated Mycobacterium avium complex in an adolescent with perinatally-acquired HIV infection. Infect Dis Rep. 2017;9(2):6884.
- Phongsamart W, Chokephaibulkit K, Chaiprasert A, et al. Mycobacterium avium complex in HIV-infected Thai children. J Med Assoc Thai. 2002;85(suppl 2):S682-S689.
- Benson CA, Williams PL, Currier JS, et al. A prospective, randomized trial examining the efficacy and safety of clarithromycin in combination with ethambutol, rifabutin, or both for the treatment of disseminated Mycobacterium avium complex disease in persons with acquired immunodeficiency syndrome. Clin Infec Dis. 2003;37(9):1234-1243.
- Dunne M, Fessel J, Kumar P, et al. A randomized, double-blind trial comparing azithromycin and clarithromycin in the treatment of disseminated Mycobacterium avium infection in patients with human immunodeficiency virus. Clin Infect Dis. 2000;31(5):1245-1252.
- Ward TT, Rimland D, Kauffman C, et al. Randomized, open-label trial of azithromycin plus ethambutol vs. clarithromycin plus ethambutol as therapy for Mycobacterium avium complex bacteremia in patients with human immunodeficiency virus. Veterans Affairs HIV Research Consortium. Clin Infect Dis. 1998;27(5):1278-1285.
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() A Review of Mycobacterium avium Complex Disease Treatment in Children Living With HIV/AIDS
A Review of Mycobacterium avium Complex Disease Treatment in Children Living With HIV/AIDS