The emergence of antimicrobial resistance against antibiotics with the highest efficacy and safety profiles continues to pose a major threat to global health.1,2 According to the CDC, 2.8 million antimicrobial-resistant infections occur each year within the United States.2 The CDC has further categorized many gram-negative pathogens, including carbapenem-resistant Enterobacterales (CRE), as urgent threats—the highest level of concern in their threat assessment framework.2 In July 2024, the CDC released an updated report demonstrating an increase in infections caused by several organisms classified as urgent and serious threats between 2019 and 2022, including hospital-onset CRE, extended-spectrum beta-lactamase (ESBL)-producing Enterobacterales, and multidrug-resistant (MDR) Pseudomonas aeruginosa.3
Gram-negative bacteria have the ability to harbor multiple concurrent resistance mechanisms, posing significant challenges to the optimization of antimicrobial therapy and contributing to increased mortality.4 Specifically, they are known for their ability to acquire all 4 main mechanisms of antibiotic resistance, including altered antimicrobial target sites, enhanced efflux pump expression, altered and/or downregulated porin channels, and enzymatic inactivation.4 Beta-lactamases, the classic example of antibacterial inactivation by enzymes, are a large and highly diverse group of enzymes that can break down the beta-lactam ring via hydrolysis.5,6
Beta-lactamases are divided into 4 classes that encompass the serine beta-lactamases (Ambler molecular classes A, C, and D) and metallo-beta-lactamases/20829" target="_blank">metallo-beta-lactamases (MBLs; Ambler molecular class B).5,6 Serine beta-lactamases use a serine in their active site to perform hydrolysis, whereas MBLs use at least 1 divalent zinc ion. Class A beta-lactamases include the narrow-spectrum beta-lactamases (eg, penicillinase, SHV-1), extended-spectrum beta-lactamases (eg, CTX-M), and serine carbapenemases (eg, Klebsiella pneumoniae carbapenemase [KPC]). The class C enzymes comprise cephalosporinases, with the most clinically relevant enzyme group being the inducible AmpC beta-lactamases, while the class D enzymes include the OXA-type enzymes (eg, OXA-48). Key enzymes within molecular class B include the New Delhi MBL (NDM), Verona Integron-encoded MBL (VIM), and imipenemase-type MBL (IMP).5,6
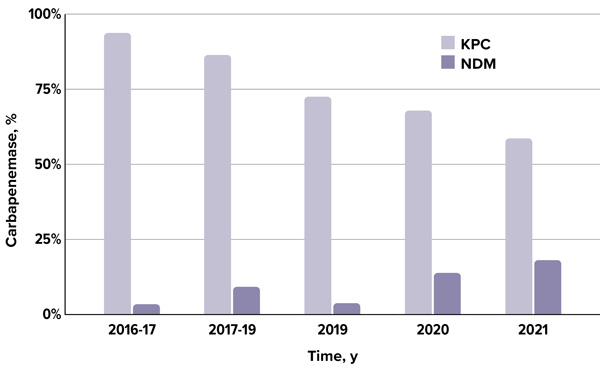
KPC has been recognized as the predominant carbapenemase in the United States, while the prevalence of MBLs have remained relatively low.5,6 However, the epidemiology of carbapenemases throughout the United States has significantly changed throughout the past decade (Figure 1).7-9 In the CRACKLE-2 prospective cohort study, conducted across 49 US hospitals between 2016 and 2017, 93% of carbapenemase-producing CRE harbored KPC-type carbapenemases, while only 3% were positive for NDMs.7 A subsequent study from 2017 to 2019 reported that KPCs accounted for 86% of isolates, and NDM comprised 9%.8 More recently, an analysis from 2019 to 2021 demonstrated that although KPC remained the predominant carbapenemase, its prevalence declined from 74% to 57%, coinciding with a notable increase in MBLs from 4% to 20%.9
Beta-lactams remain among the most valuable antibiotic classes in clinical practice.5,6 In response to the increasing prevalence of beta-lactamases, beta-lactamase inhibitors have been developed and are frequently co-formulated with beta-lactams to counteract enzymatic hydrolysis. Over the past decade, several novel beta-lactams with and without beta-lactamase inhibitors have received regulatory approval, exhibiting varying spectra of activity against enzymes across the Ambler classification system.5,6,10 Aside from aztreonam, the only other beta-lactam exhibiting in vitro activity against MBLs is cefiderocol (Fetroja, Shionogi), a siderophore cephalosporin with a unique mechanism of action.10,11 Aztreonam is a monobactam that is known for its ability to withstand hydrolysis by MBLs; however, many MBL-producing organisms co-express additional beta-lactamases, such as ESBLs, AmpC enzymes, KPCs, and/or OXA-48–like enzymes that would generally inactivate aztreonam. Consequently, coadministration of a beta-lactamase inhibitor, such as avibactam, can protect aztreonam from hydrolysis from these other enzymes.4,10-12
Overview of In Vitro Studies
Since its approval by the FDA in 2015, avibactam has been commercially available only in combination with ceftazidime.13 Given that many MBL-producing Enterobacterales coproduce multiple beta-lactamases, a therapeutic strategy employed in clinical practice has been the use of aztreonam in combination with ceftazidime-avibactam (CZA/AVI; Avycaz, AbbVie)4,10-12,14 This strategy capitalizes the beta-lactamase inhibitory activity of avibactam against coproduced serine beta-lactamases, thereby, protecting the MBL-stable aztreonam and enabling it to retain its antibacterial activity.4,10-12
Aztreonam-avibactam (ATM/AVI; Emblaveo, AbbVie) is a novel beta-lactam/beta-lactamase inhibitor that consists of a monobactam paired with a non–beta-lactam beta-lactamase inhibitor, available as a fixed-dose in a 3:1 ratio.15,16 This agent received FDA approval in February 2025 in adults with limited or no alternative treatment options for complicated intraabdominal infections (cIAIs), to be used in combination with metronidazole.15
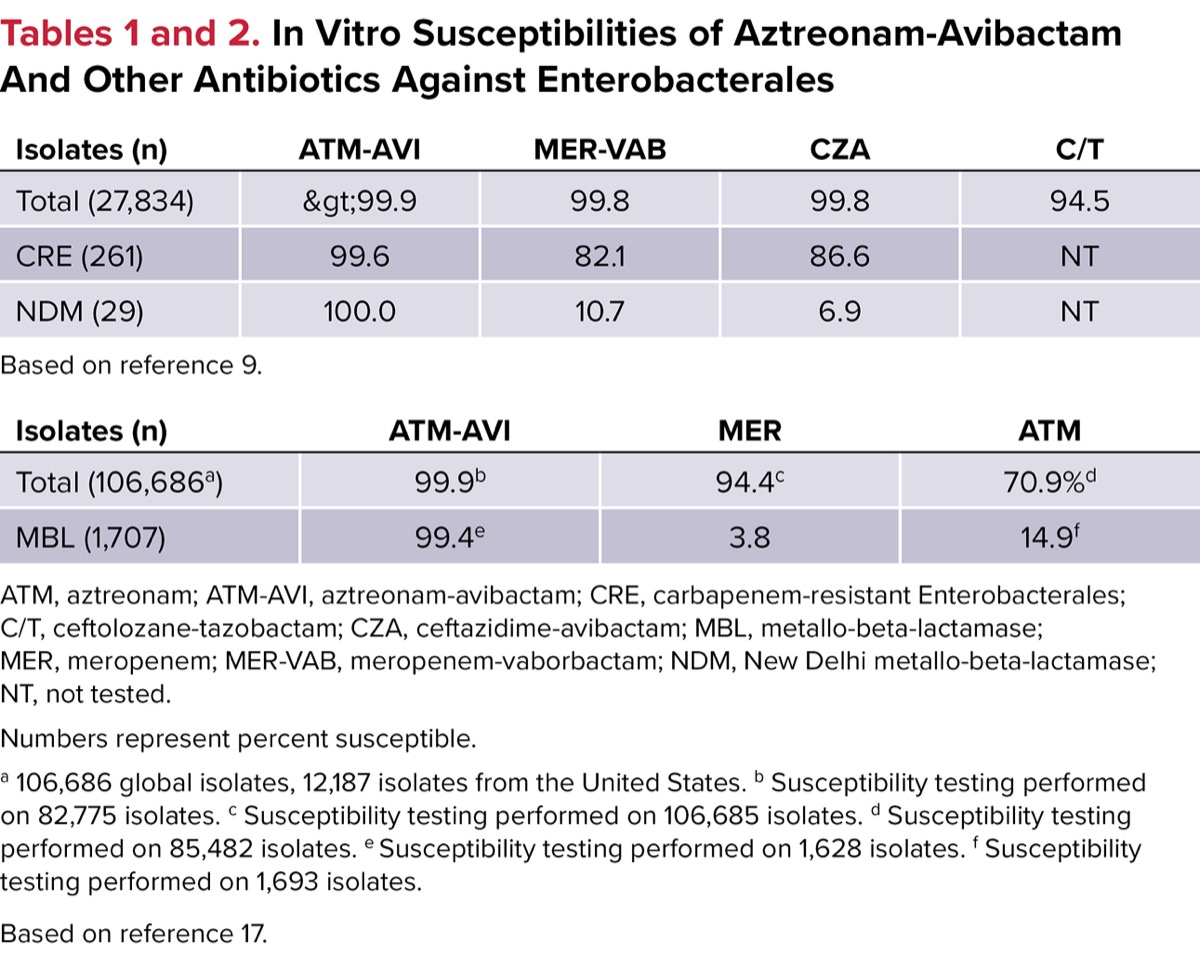
Multiple studies have assessed the in vitro activity of ATM-AVI and comparators against MDR gram-negative organisms (Tables 1-2).9,17 Sader and colleagues assessed the activity of ATM/AVI, CZA/AVI, and meropenem-vaborbactam (MER/VAB; Vabomere, Melinta Therapeutics) against 27,834 Enterobacterales isolates from 74 US hospitals from 2019 to 2021.9 Susceptibility testing was performed using broth microdilution specified by the Clinical and Laboratory Standards Institute standards, and a tentative breakpoint of 8/4 mg/L was applied for ATM-AVI.9
| Tables 1 and 2. In Vitro Susceptibilities of Aztreonam-Avibactam And Other Antibiotics Against Enterobacterales | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
The 3 most common isolated Enterobacterales included Escherichia coli (34.4%), K. pneumoniae (20.8%), and Enterobacter cloacae (8.7%). With an MIC90 (minimum inhibitory concentration needed to inhibit 90% bacterial growth) of 0.12 mg/L, more than 99.9% of all isolates were inhibited by ATM/AVI.9 Of note, 261 isolates were identified as CRE with most being KPC- (65.5%) or MBL-producing (12.6%). For all CRE, susceptibility was reported to be higher with ATM/AVI (99.6%) compared with CZA/AVI (86.2%) and MER/VAB (82.1%). Specifically for NDM MBL-producing isolates, ATM/AVI activity (100.0%) also remained higher than CZA/AVI (6.9%) and MER/VAB (10.7%); the same was true for IMP-type–producing organisms (ATM/AVI, 100.0% vs CZA/AVI, 25.0% vs MER/VAB, 50.0%).9
Rossolini and colleagues compared the activity of ATM/AVI and comparators against MBL-producing and non-producing Enterobacterales at global and regional levels between 2016 and 2020.17 The investigators employed a similar methodology that was described with the previous in vitro study, including the application of a tentative susceptibility breakpoint of 8/4 mg/L. Overall, 106,686 Enterobacterales isolates were evaluated, with the 3 most common species being E. coli (33.4%), K. pneumoniae (29.4%), and E. cloacae (10.6%). CRE and MBL-producing Enterobacterales were identified in 5,356 (5.0%) and 1,707 (1.6%) of the total isolates, respectively.17
With an MIC90 of 0.25 mg/L, ATM/AVI displayed the highest in vitro susceptibility rates against all Enterobacterales isolates (99.9%).17 Specifically against MBL-producing isolates, ATM/AVI also displayed potent in vitro activity inhibiting nearly all isolates (99.4%), compared with drastically lower susceptibility rates demonstrated by most other agents (amikacin, 50.0%; gentamicin, 29.0%; aztreonam, 14.9%). The activity of ATM/AVI was consistent among both NDM- and VIM-producing Enterobacterales. Additionally, the study showed ATM/AVI to have high potency against MBL-positive isolates harboring other carbapenemase genes like KPC (100.0%) and OXA-48–like (99.7%).17
Overview of Clinical Studies
Prior to the FDA approval of ATM/AVI, several clinical studies evaluated the combination of aztreonam with CZA/AVI for the treatment of infections caused by MBL-producing Enterobacterales.12,14 Overall, these studies demonstrated that the CZA/AVI plus aztreonam combination was associated with improved clinical outcomes compared with other active antibiotics under these highly resistant phenotypes.12,14
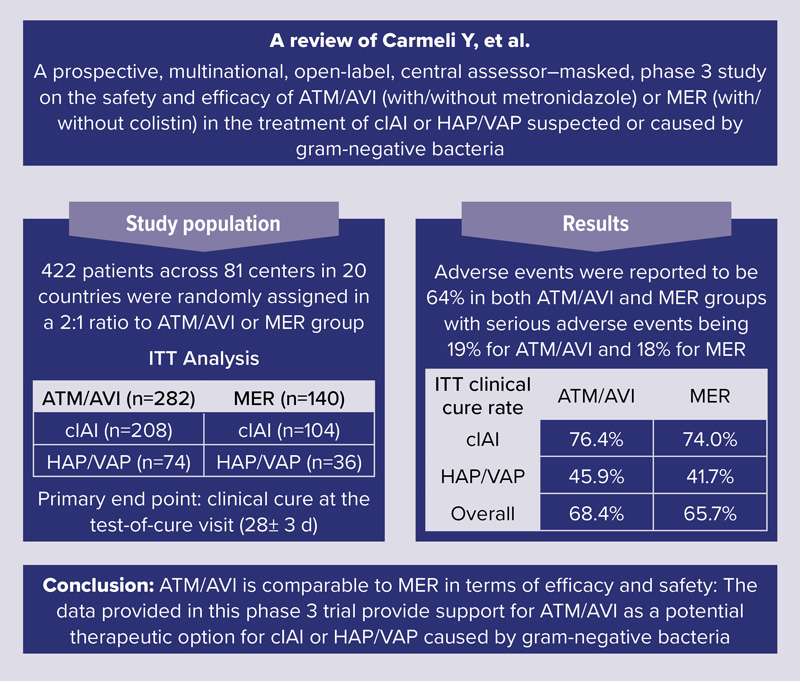
The REVISIT trial was a phase 3, open-label, multinational, randomized trial that investigated ATM/AVI versus meropenem for the treatment of cIAIs or hospital-acquired pneumonia/ventilator-associated pneumonia (HAP/VAP) caused (or suspected to be caused) by gram-negative bacteria (Figure 2).18 Specifically, the investigators evaluated the safety and efficacy of ATM/AVI with (cIAIs) or without metronidazole (HAP/VAP), which was compared with meropenem with or without colistin in a 2:1 ratio at 81 centers across 20 countries. The primary end point was clinical cure at the test-of-cure visit (28±3 days) in the intention-to-treat (ITT) population.18
Overall, there was a total of 422 patients that were included within the ITT analysis (ATM/AVI, 66.8% vs meropenem, 33.2%).18 Most patients included were younger than 65 years of age (67.1%), male (68.0%), and White (54.0%) or Asian (41.7%). Overall, more patients had cIAIs (ATM/AVI, 49.3%; meropenem, 24.7%) versus HAP/VAP (ATM/AVI, 17.5%; meropenem, 8.5%). In total, 64.2% of patients had at least 1 gram-negative pathogen identified at baseline and included in the microbiological-ITT analysis set, most commonly E. coli in patients with cIAIs and K. pneumoniae in those with HAP/VAP. Overall, Enterobacterales comprised 93.0% of all isolates. Of note, 80 gram-negative isolates were tested for carbapenemases with a positivity rate of 23.8%; however, only 7 isolates were MBL-positive in the ATM/AVI group, and 3 in the meropenem group.18
Overall, the adjudicated clinical cure rate within the ITT population at the test-of-cure visit was 68.4% and 65.7% in the ATM/AVI and meropenem groups, respectively, exhibiting a treatment difference of 2.7% (–6.6% to 12.4%).18 Similar cure rates were also seen between the 2 groups for patients with cIAIs and HAP/VAP. Finally, there were no major differences in adverse event rates between the 2 groups, with the most common reported side effects being anemia (6.6%), increases in alanine aminotransferase (6.1%), and diarrhea (5.1%). Overall, the phase 3 REVISIT trial provides support for ATM/AVI as an option for patients with cIAIs (with metronidazole) or HAP-VAP. However, a major limitation of this study were the low numbers of infections caused by MBL-producing Enterobacterales.18
The ASSEMBLE study was a prospective, randomized, multicenter, open-label, comparative study that aimed to assess the efficacy and safety of ATM/AVI compared with best available therapy for serious infections caused by MBL-producing gram-negative bacteria.19,20 Unfortunately, the trial was terminated early due to low enrollment (15 participants) but showed clinical cure rates at the test-of-cure visit of 41.7% and 0% for ATM/AVI and best available therapy, respectively. However, the small sample size severely limits the results and their generalizability.19,20
Conclusion
The rise of antimicrobial resistance, particularly among carbapenem-resistant and MBL-producing Enterobacterales, continues to challenge clinicians and limit effective treatment options.1,2 These organisms frequently harbor multiple resistance mechanisms, complicating therapy and contributing to poor outcomes.4,10,12 ATM/AVI, a novel fixed-dose combination of a monobactam and non–beta-lactam beta-lactamase inhibitor, has demonstrated potent in vitro activity against a broad range of multidrug-resistant gram-negative pathogens, including MBL-producing Enterobacterales.9,15-17
As a fixed-dose formulation, ATM/AVI provides improved pharmacokinetic and pharmacodynamic properties ensuring synchronized drug exposure, thus enhancing the reliability of antibacterial activity.15 It also offers greater dosing convenience, reducing the complexity and error potential associated with administering separate agents.15 Clinical studies provide growing support for the use of ATM/AVI.18 Although limitations remain, particularly in the volume of data for infections caused by confirmed MBL-producing organisms, the available evidence positions ATM/AVI as a valuable and necessary addition to the antimicrobial arsenal for treating severe infections cause by drug-resistant gram-negative pathogens.
References
- Antimicrobial resistance. WHO. November 21, 2023. Accessed August 7, 2025. who.int/news-room/fact-sheets/detail/antimicrobial-resistance
- CDC. Antibiotic Resistance Threats in the United States, 2019. Department of Health and Human Services, CDC; 2019. Accessed July 22, 2025. bit.ly/3Hf78Y4-IDSE
- Antimicrobial resistance threats in the United States, 2021-2022. CDC. April 11, 2025. Accessed July 22, 2025. bit.ly/4fr1YEV-IDSE
- Gauba A, Rahman KM. Evaluation of antibiotic resistance mechanisms in gram-negative bacteria. Antibiotics (Basel). 2023;12(11):1590.
- Bush K, Bradford PA. Epidemiology of ß-lactamase-producing pathogens. Clin Microbiol Rev. 2020;33(2):e00047-19.
- Bush K. Past and present perspectives on ß-lactamases. Antimicrob Agents Chemother. 2018;62(10):e01076-18.
- van Duin D, Arias CA, Komarow L, et al. Molecular and clinical epidemiology of carbapenem-resistant Enterobacterales in the USA (CRACKLE-2): a prospective cohort study. Lancet Infect Dis. 2020;20(6):e116.
- Sabour S, Huang JY, Bhatnagar A, et al. Detection and characterization of targeted carbapenem-resistant health care-associated threats: findings from the Antibiotic Resistance Laboratory Network, 2017 to 2019. Antimicrob Agents Chemother. 2021;65(12):e0110521.
- Sader HS, Mendes RE, Carvalhaes CG, et al. Changing epidemiology of carbapenemases among carbapenem-resistant Enterobacterales from United States hospitals and the activity of aztreonam-avibactam against contemporary Enterobacterales (2019-2021). Open Forum Infect Dis. 2023;10(2):ofad046.
- Tamma PD, Hsu AJ. Defining the role of novel ß-lactam agents that target carbapenem-resistant gram-negative organisms. J Pediatric Infect Dis Soc. 2019;8(3):251-260.
- Tamma PD, Aitken SL, Bonomo RA, et al. Infectious Diseases Society of America 2024 guidance on the treatment of antimicrobial-resistant gram-negative infections. Clin Infect Dis. Published online August 7, 2024. doi:10.1093/cid/ciae403 bit.ly/4f0waGG-IDSE
- Mauri C, Maraolo AE, Di Bella S, et al. The revival of aztreonam in combination with avibactam against metallo-ß-lactamase-producing gram-negatives: a systematic review of in vitro studies and clinical cases. Antibiotics (Basel). 2021;10(8):1012.
- Avycaz. Prescribing information. Allergan USA Inc; December 2022. bit.ly/3GDsq1n-IDSE
- Falcone M, Daikos GL, Tiseo G, et al. Efficacy of ceftazidime-avibactam plus aztreonam in patients with bloodstream infections caused by metallo-ß-lactamase-producing Enterobacterales. Clin Infect Dis. 2021;72(11):1871-1878.
- Emblaveo. Prescribing information. AbbVie Inc; February 2025. bit.ly/4lHYEru-IDSE
- Al Musawa M, Bleick CR, Herbin SR, et al. Aztreonam-avibactam: the dynamic duo against multidrug-resistant gram-negative pathogens. Pharmacotherapy. 2024;44(12):927-938.
- Rossolini GM, Arhin FF, Kantecki M. In vitro activity of aztreonam-avibactam and comparators against metallo-ß-lactamase-producing Enterobacterales from ATLAS Global Surveillance Program, 2016-2020. J Glob Antimicrob Resist. 2024;36:123-131.
- Carmeli Y, Cisneros JM, Paul M, et al. Aztreonam-avibactam versus meropenem for the treatment of serious infections caused by gram-negative bacteria (REVISIT): a descriptive, multinational, open-label, phase 3, randomised trial. Lancet Infect Dis. 2025;25(2):218-230.
- Sangiorgio G, Calvo M, Stefani S. Aztreonam and avibactam combination therapy for metallo-ß-lactamase-producing gram-negative bacteria: a narrative review. Clin Microbiol Infect. 2025;31(6):971-978.
- A Prospective, Randomized, Open-Label, Comparative Study to Assess the Efficacy, Safety and Tolerability of Aztreonam-Avibactam (ATM-AVI) and Best Available Therapy for the Treatment of Serious Infections Due to Multidrug-Resistant Gram-Negative Bacteria Producing Metallo-ß-Lactamase (MBL). ClinicalTrials.gov Identifier NCT03580044. July 22, 2025. clinicaltrials.gov/ct2/show/NCT03580044
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.
Download to read this article in PDF document:![]() Rearming Beta-Lactams: A Focus on Aztreonam-Avibactam and Metallo-Beta-Lactamases
Rearming Beta-Lactams: A Focus on Aztreonam-Avibactam and Metallo-Beta-Lactamases