Antimicrobial resistance (AMR) has emerged as a leading global health threat with mortality rates due to infections from resistant organisms rising each year.1 In the United States, the CDC estimates that nearly 3 million AMR infections occur annually, while AMR is responsible for more than 35,000 deaths.2 Internationally, the WHO estimated that bacterial AMR was directly responsible for approximately 1.3 million deaths and contributed to nearly 5 million deaths in 2019.3
Four of the 6 leading bacteria most frequently contributing to AMR-related mortality are gram-negative pathogens, including Escherichia coli and Klebsiella pneumoniae of the Enterobacterales order.4 Of the mechanisms of resistance associated with the Enterobacterales, those exhibiting resistance to carbapenems have been listed as an urgent threat by the CDC.2
Specifically, carbapenem-resistant Enterobacterales (CRE) can be grouped broadly into carbapenemase-producing CRE (CP-CRE) and non–carbapenemase-producing CRE (non–CP-CRE).5,6 CP-CRE occur due to the presence of gene(s) that encode a carbapenemase, whereas the mechanisms of resistance of non–CP-CRE may be due to carbapenem efflux, outer membrane loss, mutations in penicillin-binding proteins, and/or production of other beta-lactamases (eg, extended-spectrum beta-lactamases and/or AmpC beta-lactamases) with the previously mentioned mechanisms of resistance.5,6
Invasive infections caused by CRE have been reported to have mortality rates as high as 70%; therefore, efficacious and safe antimicrobial options are vital.7 Treatment of infections caused by CRE have involved options such as the aminoglycosides and polymyxins, which are layered with complications, such as unfavorable pharmacokinetics and/or safety concerns.8-10 Furthermore, these alternative agents have been shown to be inferior to the current first-line treatment options in patients with infections caused by CRE.11,12
Ceftazidime-avibactam (CZA; Avycaz, AbbVie) and meropenem-vaborbactam (MVB; Vabomere, Melinta Therapeutics) are novel beta-lactams that were recently introduced to the market, which have shown promising in vitro activity and clinical outcomes data.13-23 The primary purpose of this review is to describe the in vitro activity and real-world evidence of CZA and MVB against CRE.
Ceftazidime-Avibactam
Overview and In Vitro Studies
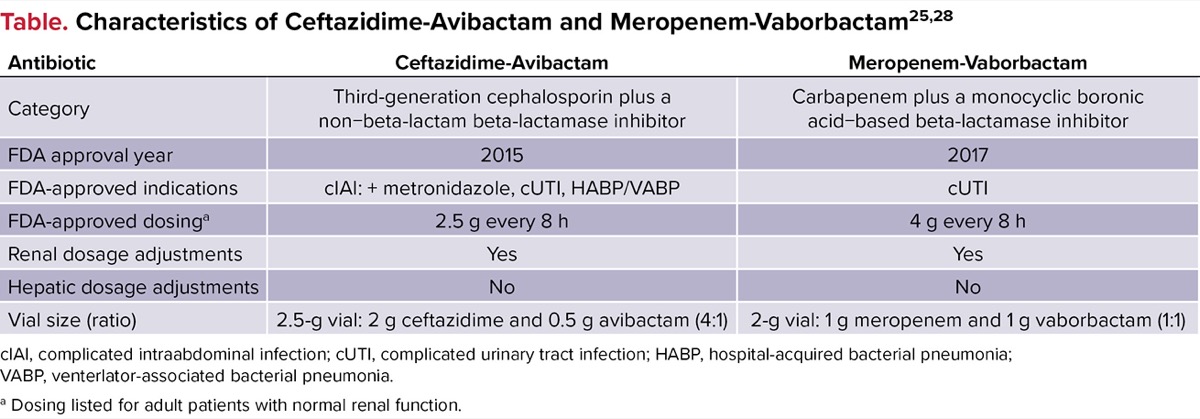
Ceftazidime-avibactam is a novel beta-lactam/beta-lactamase inhibitor that consists of a well-known, third-generation cephalosporin coupled with a non–beta-lactam beta-lactamase inhibitor that is available as a fixed dose in a 4:1 ratio.24,25 This novel agent initially received FDA approval in 2015, for the treatment of complicated urinary tract infections (UTIs), including pyelonephritis, and complicated intraabdominal infections with metronidazole.25 Importantly, the addition of avibactam to ceftazidime restores the susceptibility of ceftazidime to many ceftazidime-resistant gram-negative isolates and has demonstrated activity against many beta-lactamases, including extended-spectrum beta-lactamases, AmpC beta-lactamases, K. pneumoniae carbapenemases (KPC), and OXA-48–like enzymes.24 Characteristics of CZA can be found in the Table.
| Table. Characteristics of Ceftazidime-Avibactam and Meropenem-Vaborbactam25,28 | ||
| Antibiotic | Ceftazidime-Avibactam | Meropenem-Vaborbactam |
|---|---|---|
| Category | Third-generation cephalosporin plus a non-beta-lactam beta-lactamase inhibitor | Carbapenem plus a monocyclic boronic acid-based beta-lactamase inhibitor |
| FDA approval year | 2015 | 2017 |
| FDA-approved indications | cIAI: + metronidazole, cUTI, HABP/VABP | cUTI |
| FDA-approved dosinga | 2.5 g every 8 h | 4 g every 8 h |
| Renal dosage adjustments | Yes | Yes |
| Hepatic dosage adjustments | No | No |
| Vial size (ratio) | 2.5-g vial: 2 g ceftazidime and 0.5 g avibactam (4:1) | 2-g vial: 1 g meropenem and 1 g vaborbactam (1:1) |
| cIAI, complicated intraabdominal infection; cUTI, complicated urinary tract infection; HABP, hospital-acquired bacterial pneumonia; VABP, venterlator-associated bacterial pneumonia. a Dosing listed for adult patients with normal renal function. | ||
Numerous studies have shown that CZA has potent in vitro activity against many resistant gram-negative organisms, including CRE.13-16 Sader and colleagues evaluated the antimicrobial activity of CZA against 10,928 gram-negative isolates collected from 73 U.S. medical centers.13 Among the 8,640 Enterobacterales, 99.8% of strains were inhibited at a CZA minimum inhibitory concentration (MIC) of 4/4 mg/L or lower with an MIC required to inhibit 90% of isolates (MIC90) of 0.25 mg/L. Importantly, the current breakpoint for CZA per the Clinical and Laboratory Standards Institute (CLSI) is 8/4 mg/L or lower.26 Against ceftazidime (n=282) and meropenem (n=115) non-susceptible isolates, CZA susceptibility rates ranged between 98.8-100% (MIC90: 0.5-1 mg/L) and were 98.3% (MIC90 = 2 mg/L), respectively.13
Spiliopoulou and colleagues conducted a similar in vitro analysis of strictly carbapenem-nonsusceptible Enterobacterales.16 CZA susceptibility testing was performed on 1,460 isolates and overall susceptibility was found to be 73.0%. Importantly, this isolate panel included 367 isolates (25.1% of the total isolates) that produced metallo-beta-lactamases (MBL), in which avibactam lacks in vitro activity against these enzymes.24 When evaluating the CZA-resistant isolates (n=394), 98.4% of the screened isolates were found to carry a gene encoding an MBL enzyme. Among the isolates that were found to be carbapenemase-positive and MBL-negative (n=910), CZA susceptibility rates were 99.8%. Furthermore, susceptibility rates were also high for 98 isolates of non–CP-CRE at 95.9%.16
Overview of CZA Clinical Studies
Jorgensen and colleagues conducted a multicenter, retrospective study to assess the clinical characteristics, microbiology, including clinical outcomes data for CZA use in 203 patients with gram-negative bacterial infections, including 117 patients with CRE.19 In the subgroup analysis of patients treated for CRE infections, 48.7% of patients were Black, 53.8% were male, and the median age was 63 years (IQR, 52-73 years). Most patients had recent antibiotic exposure (82.1%) and/or hospitalizations (80.3%), 47.9% of patients had a multidrug-resistant organism infection or colonization within the past year, and 60.7% of the CRE infections were hospital-acquired. The most common sites of infection were respiratory (33.3%), intraabdominal (22.2%), urinary (20.4%), skin and soft tissue (8.8%), osteoarticular (6.0%), and/or primary bacteremia (6.0%). The most commonly isolated organisms included K. pneumoniae (63.2%), E. coli (14.5%), and Enterobacter spp. (12.8%). Of the K. pneumoniae isolates, 58.1% had CZA susceptibility testing performed with an MIC90 of 4 mg/L. Importantly, no data were reported regarding the mechanisms responsible for carbapenem resistance (eg, KPC, non–CP-CRE).
All patients received an infectious diseases consultation and nearly half (46.2%) required a CZA renal dosage adjustment. The median (IQR) time to CZA therapy was 93 (52-145) hours, and the median (IQR) duration of CZA therapy was 13 (6-18) days. Concomitant antibiotics were given to 38.5% of patients with aminoglycosides (28.9%), colistin/polymyxin B (22.2%), and/or tigecycline (22.2%) being the most common. Adverse events potentially attributable to CZA were reported in 8 patients (6.8%), with acute kidney injury (n=5) and Clostridioides difficile infection (n=3) being the most common.
Composite clinical failure, which was defined as 30-day mortality, 30-day recurrence, and/or worsening or failure to improve while on CZA, was reported in 29.1% of patients with CRE infections. When breaking down the components of the primary outcome, 16.2% of patients died within 30 days, 6.0% experienced a microbiological recurrence, and 15.4% worsened or failed to improve while receiving CZA. In patients with follow-up cultures, no isolates developed resistance to CZA.19
Another non-interventional study conducted by Soriano and colleagues evaluated the patterns of use of CZA, clinical success rates, and safety across European and Latin American regions in 516 patients who were treated with CZA for at least 72 hours.20 The most common types of infections included hospital-acquired pneumonia or ventilator-associated pneumonia (22.1%), complicated UTIs (20.0%), bloodstream infections (18.8%), and complicated intraabdominal infections (17.4%), while the most common organism was K. pneumoniae (59.3%), in which 88.5% exhibited carbapenem resistance. Of all isolates (n=710), 79.0% were tested for beta-lactamases, with 390 isolates having beta-lactamases identified (KPC, 34.6%; OXA-48, 20.0%; MBL, 17.9%). Treatment success was achieved in 77.3% of patients overall, and adverse events were reported in 6 patients.20
Meropenem-Vaborbactam
Overview of MVB And In Vitro Studies
Meropenem-vaborbactam is another novel beta-lactam/beta-lactamase inhibitor that consists of an established carbapenem co-formulated with a cyclic boronic acid–based beta-lactamase inhibitor that is available as a fixed dose in a 1:1 ratio.27,28 This agent received FDA approval in 2017, for the treatment of complicated UTIs, including pyelonephritis.28 The addition of vaborbactam to meropenem restores the susceptibility of meropenem to CRE isolates harboring KPC, but lacks activity against the other carbapenemases.27 Characteristics of MVB can be found in the Table.
Many studies have demonstrated that MVB exhibits potent in vitro activity against various gram-negative clinical isolates, including some CRE.17,18 Castanheira and colleagues evaluated the antimicrobial activity of MVB against 14,304 gram-negative isolates collected worldwide.17 Among the 10,426 Enterobacterales, 99.3% of strains were inhibited at a MVB MIC of 2/8 mg/L or lower with an MIC90 of 0.06 mg/L. Importantly, the current breakpoint for MVB per the CLSI is 4/8 mg/L or lower.26 Against carbapenem-resistant isolates (n=265), the MVB MIC90 was 32 mg/L; however, the MIC90 value was lowered substantially to 0.5 mg/L when limiting the carbapenem-resistant isolates to those producing KPC (n=135).17
Another evaluation from Hackel and colleagues was a related in vitro analysis against strictly KPC-producing Enterobacterales.18 MVB susceptibility testing was performed on 991 isolates (88.6% K. pneumoniae) and the overall susceptibility rate was found to be 99.0%. The addition of vaborbactam to meropenem was shown to lower the meropenem MIC90 from greater than 32 mg/L to an MVB MIC90 of 1 mg/L. Furthermore, there were no clinically relevant differences in MVB activity when the organisms were stratified by KPC variant type.18
Overview of MVB And Clinical Studies
Alosaimy and colleagues conducted a multicenter, retrospective descriptive study to evaluate the clinical characteristics, microbiology, and clinical outcomes data for MVB use in 126 patients with gram-negative bacterial infections (78.6% with CRE).21 Overall, 47.6% of the patients were White, 62.7% were male, and the median (IQR) age was 56 (37-68) years. The most common comorbidities included diabetes (39.7%), moderate to severe kidney disease or on chronic dialysis (29.4%), and/or heart failure (21.4%), while the most common infections were pneumonia (38.1%) including 19.8% of patients with ventilator-associated pneumonia, intraabdominal (19.0%), urinary (13.5%), and skin and soft tissue (10.3%). The most commonly isolated organisms included K. pneumoniae (42.1%), E. coli (19.8%), and E. cloacae (16.7%). Among the Enterobacterales isolates with MVB susceptibility testing reported (n=79), the MIC range was 0.023/8 to 8/8 mg/L. Importantly, no data were reported regarding the mechanisms responsible for carbapenem resistance (eg, KPC, non–CP-CRE).
Nearly half (49.2%) of patients were in the ICU upon collection of the index culture, with a median (IQR) Sequential Organ Failure Assessment (SOFA) score of 7 (4-10). In a subgroup analysis of patients treated for CRE infections, the median (IQR) time to MVB therapy was 85.1 (48.6-133.1) hours, while the median (IQR) duration of MVB therapy was 11.8 (6.7-16.0) days. Overall, combination antibiotic therapy for at least 48 hours was given to 34.1% of patients, with amikacin, levofloxacin, minocycline, tobramycin, and aztreonam being most common. Only 4 patients in the total cohort had a reported adverse effect, which included acute kidney injury (n=2), hepatotoxicity (n=1), and a severe dermatological reaction (n=1).
When evaluating patients treated with MVB for CRE, the primary outcome of 30-day mortality occurred in 19.2% of patients. Regarding other outcomes reported, 30-day recurrence was found in 13.1% of patients, 30-day hospital readmissions in 21.2%, and 25.3% worsened or failed to improve while on MVB. Overall, in patients with follow-up cultures (n=25), no isolates developed resistance to MVB.21
Another prospective, observational study conducted by Tiseo et al evaluated the clinical experience of MVB in treating resistant gram-negative organisms in patients hospitalized in the University of Pisa.22 Of the 104 patients, 63 (60.6%) patients treated with MVB had CRE, with 61 of the 63 (96.8%) infections being KPC-producing K. pneumoniae. Of the KPC-producing isolates, the most common infections were bacteremia (68.9%), hospital-acquired pneumonia (27.9%), and UTIs (16.4%). Clinical success, which was defined as a composite of resolution of signs and symptoms, survival, and lack of microbiological failure at day 30 from the onset of infection, was achieved in 81.9% of patients treated with MVB for KPC-producing K. pneumoniae.22
Conclusion
Enterobacterales exhibiting carbapenem resistance are associated with high rates of morbidity and mortality and have limited treatment options.5 Novel antibiotics, such as CZA and MVB, have been developed that offer therapeutic advantages over historically used options for CRE.8,9,11,12,19-23
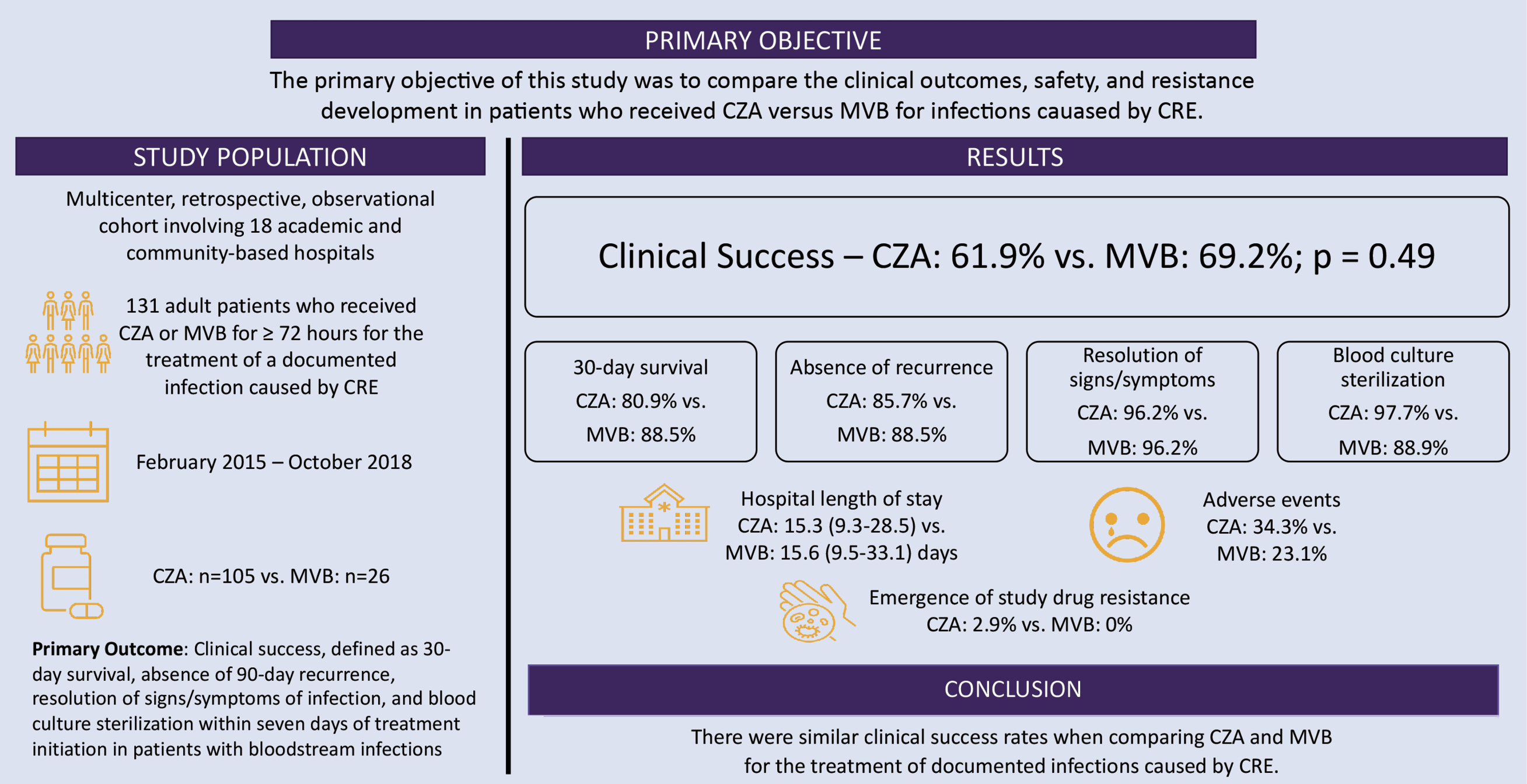
Furthermore, descriptive reports have emerged that show high clinical success rates and low rates of adverse effects.19-22 To my knowledge, the only study available comparing CZA and MVB for the treatment of CRE was conducted by Ackley and colleagues, and is described in the Figure.23 Additional prospective and larger real-world, observational studies of CZA use for infections caused by various types of CRE and comparing CZA and MVB for KPC-producing CRE are needed to guide the use of these antibiotics for carbapenem-resistant infections.
References
- Prestinaci F, Pezzottii P, Pantosi A. Pathog Glob Health. 2015;109(7):309-318.
- CDC. Accessed August 8, 2024. https://bit.ly/ 3WGWkpl-IDSE.
- WHO. Accessed July 17,2024. https://bit.ly/ 4dyhpsT-IDSE
- Zhang C, Fu X, Liu Y, et al. Lancet Reg Health West Pac. 2024;43:100972.
- Nordmann P, Dortet L, Poirel L. Trends Mol Med. 2012;18(5):263-272.
- Zou H, Xiong S-J, Lin Q-X, et al. Infect Drug Resist. 2019;12:3017-3027.
- Borer A, Saidel-Odes L, Riesenberg K, et al. Infect Control Hosp Epidemiol. 2009;30(10):972-976.
- Oliota AF, Penteado ST, Tonin FS, et al. Diagn Microbiol Infect Dis. 2019;94(1):41-49.
- Oliveira JF, Silva CA, Barbieri CD, et al. Antimicrob Agents Chemother. 2009;53(7):2887-2891.
- Avedissian SN, Liu J, Rhodes NJ, et al. Antibiotics (Basel). 2019;8(1):31.
- Shields RK, Nguyen MH, Chen L, et al. Antimicrob Agents Chemother. 2017;61(8):e00883-17.
- Van Duin D, Lok JJ, Earley M, et al. Clin Infect Dis. 2018;66(2):163-171.
- Sader HS, Castanheira M, Flamm RK, et al. Antimicrob Agents Chemother. 2014;58(3):1684-1692.
- Karlowsky JA, Kazmierczak KM, Bouchillon SK, et al. Antimicrob Agents Chemother. 2018;62(7):e02569-17.
- Soh TST, Salvinder S, Chen VSY. Med J Malaysia. 2022;77(2):174-184.
- Spiliopoulou I, Kazmierczak K, Stone GG. J Antimicrob Chemother. 2020;75(2):384-391.
- Castanheira M, Huband MD, Mendes RE, et al. Antimicrob Agents Chemother. 2017;61(9):e00567-17.
- Hackel MA, Lomovskaya O, Dudley MN, et al. Antimicrob Agents Chemother. 2017;62(1):e01904-17.
- Jorgensen SCJ, Trinh TD, Zasowski EJ, et al. Open Forum Infect Dis. 2019;6(12):ofz522.
- Soriano A, Montravers P, Bassetti M, et al. Infect Dis Ther. 2023;12(3):891-917.
- Alosaimy S, Lagnf AM, Morrisette T, et al. Open Forum Infect Dis. 2021;8(8):ofab371.
- Tiseo G, Galfo V, Riccardi N, et al. Eur J Clin Microbiol Infect Dis. 2024 Feb 20. doi:10.1007/s10096-024-04758-2
- Ackley R, Roshdy D, Meredith J, et al. Antimicrob Agents Chemother. 2020;64(5):e02313-19.
- Zasowski EJ, Rybak JM, Rybak MJ. Pharmacotherapy. 2015;35(8):755-770.
- Avycaz (ceftazidime/avibactam) [prescribing information]. Allergan USA Inc; December 2022.
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing. 34th ed. CLSI supplement M100.
- Jorgensen SCJ, Rybak MJ. Pharmacotherapy. 2018;38(4):444-461.
- Vabomere (meropenem and vaborbactam) [prescribing information]. Melinta Therapeutics LLC; June 2021.
Copyright © 2024 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Carbapenem-Resistant Enterobacterales (CRE): A Focus on Ceftazidime-Avibactam and Meropenem-Vaborbactam
Carbapenem-Resistant Enterobacterales (CRE): A Focus on Ceftazidime-Avibactam and Meropenem-Vaborbactam