The novel antibiotic aztreonam-avibactam (ATM-AVI; Emblaveo, AbbVie) may be a potential therapeutic option for difficult-to-treat infections caused by metallo-beta-lactamase (MBL)-producing gram-negative pathogens, according to results of the phase 3 ASSEMBLE trial (JAC Antimicrob Resist 2025;7[4]:dlaf131). However, the trial included just 15 patients due to recruitment challenges experienced during the 2020 peak of the COVID-19 pandemic.
The researchers compared ATM-AVI with best available therapy (BAT) for treatment of complicated intraabdominal infection (cIAI), complicated urinary tract infection (cUTI), hospital-acquired/ventilator-associated pneumonia (HAP/VAP) or bloodstream infection (BSI) caused by confirmed MBL-producing multidrug-resistant pathogens.
Clinical cure—resolution or improvement of the infection to the point where no further treatment was required—was achieved in five of 12 patients (42%) receiving ATM-AVI and zero of three (0%) receiving other therapies. All-cause mortality rates at 28 days were one of 12 (8%) for those receiving the study drug and one of three (33%) for those prescribed other therapies. The two deaths were not considered treatment-related.
Although the number of participants in ASSEMBLE was small, the presented data support the potential role of ATM-AVI in the management of these infections, said lead study author George L. Daikos, MD, a professor of infectious diseases at the National and Kapodistrian University of Athens, in Greece.
“The infections caused by Enterobacterales-producing MBLs are on the rise worldwide and have become endemic in several geographic areas,” Dr. Daikos told Infectious Disease Special Edition. They present “a significant threat to patients and healthcare systems, as treatment options are extremely limited.”
Adding More Data to Support Drug’s Use
The multicenter trial in nine countries randomized hospitalized adults 2:1 to ATM-AVI (plus metronidazole for cIAI) or BAT for five to 14 days (cIAI, cUTI and BSI) or seven to 14 (HAP/VAP) days. Patients taking the study drug received a loading dose of 500 to 167 mg followed immediately by 1,500 to 500 mg, and then 1,500 to 500 mg every six hours in three-hour infusions. BAT was monotherapy or combination therapy at the investigator’s discretion based on local epidemiology, Dr. Daikos said. For example, one patient received amikacin, colistin and meropenem.
The most frequent baseline pathogens were Enterobacterales with Klebsiella pneumoniae being most common.
Treatment-related adverse events occurred in two of 12 (17%) patients in the ATM-AVI group. One patient had frequent bowel movements and chest discomfort, while the other experienced increased alanine aminotransferase, aspartate aminotransferase, blood alkaline phosphatase and gamma-glutamyl transferase.
In February, the FDA approved the drug with metronidazole for the treatment of patients 18 years and older with limited or no alternative options for cIAIs, including those caused by gram-negative microorganisms. ATM-AVI has additional indications in Europe.
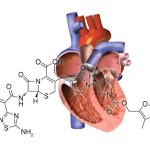
The drug contains aztreonam, a monobactam antibiotic resistant to hydrolysis by MBLs but that can be degraded by other beta-lactamases, and avibactam, a novel beta-lactamase inhibitor that neutralizes extended-spectrum beta-lactamases and AmpC beta-lactamases, Dr. Daikos said. The combination restores aztreonam’s activity against bacteria that coproduce MBLs and other beta-lactamases, he said.
“It’s certainly a welcome addition, I mean, we’re never going to turn down more data. But it is a small study,” said James Lewis, PharmD, FIDSA, the clinical supervisor of infectious disease at Oregon Health & Science University, in Portland. To that end, he said, it could be viewed almost as a case series.
The FDA labeling for ATM-AVI says it should be used in circumstances where there are limited or no other options, Dr. Lewis added: “I think that kind of tells you the amount of data we’re dealing with here with regards to this agent.” However, it provides activity against MBL producers, which has been one of the most problematic organisms to kill, he said.
“It’s going to be interesting to see how the market adapts, and whether or not they buy this prepackaged formulation” versus making their own combination by running simultaneous drips for aztreonam, avibactam and maybe a third medication that they have on the shelf, he said.
Cefiderocol (Fetroja, Shionogi) is another viable option for managing MBL-driven infections, he noted.
The study was sponsored by Pfizer. Dr. Daikos is an advisor for and consultant to Pfizer, and has received honoraria from MSD, Pfizer and Viatris. Dr. Lewis served as a consultant to AbbVie during the drug’s development and has served as a consultant to Shionogi. He is also a member of the Infectious Disease Special Edition editorial advisory board.
This article is from the October 2025 print issue.