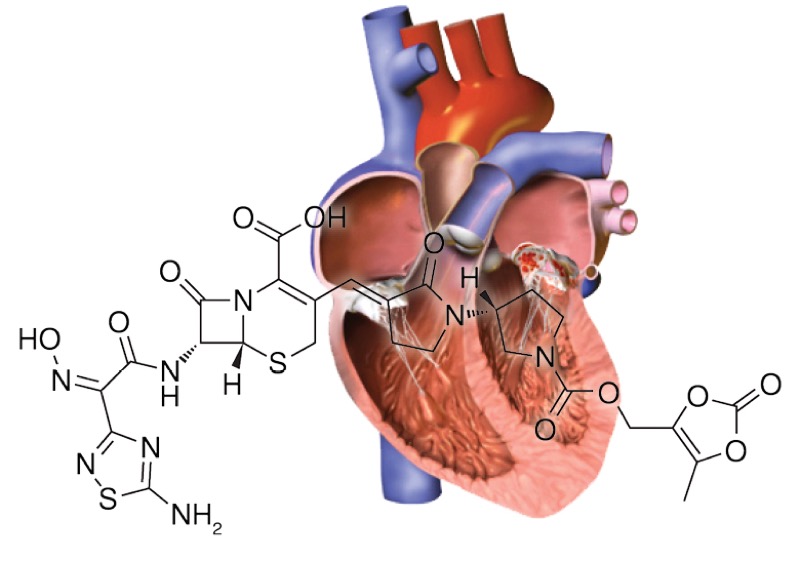
Ceftobiprole medocaril (Zevtera, Innoviva), a new fifth-generation cephalosporin antibiotic, became commercially available this past May after FDA approval in April. Ceftobiprole medocaril has three indications: adults and children 3 months and older with community-acquired bacterial pneumonia, adults with acute bacterial skin and skin structure infections, and adults with Staphylococcus aureus bacteremia (SAB), including right-sided infective endocarditis (IE). Experts spoke to Infectious Disease Special Edition about what having this new drug at their disposal will mean for treating right-sided IE, as this is the newest approval for SAB caused by methicillin-resistant S. aureus (MRSA) isolates since 2006.
Treating Right-Sided IE
Right-sided IE, which typically involves the heart’s tricuspid valve, accounts for 5% to 10% of all IE cases, according to a 2020 review article in the Journal of the American Heart Association (2020;9[15]:e017293).
It is also more commonly associated with IV drug use, intracardiac devices like prosthetic heart valves and central venous catheters, “all of which have become more prevalent over the past 20 years,” according to the authors.
It can also be a result of dental work in people with heart valve issues, where infection moves to the heart and can embed and then grow in the valve, said Brian Baturin, MD, an imaging cardiologist with MedStar Heart & Vascular Institute, at MedStar Washington Hospital Center, in Washington, D.C. “Left untreated, the infection can cause a leaky valve that could require surgery to replace it, or even result in death for the patient,” Dr. Baturin said.
IE symptoms can include fever, chills, lethargy, leg swelling, difficulty breathing and progression of any of those symptoms, Dr. Baturin said. If a SAB diagnostic test comes back positive, providers are likely to perform an echocardiogram or the more sensitive transthoracic echocardiogram to check the heart and heart devices.
A New Option
Hunter Fraker, MD, an attending physician in infectious disease at Tufts Medical Center, in Boston, said infectious disease specialists are grateful to have a new drug to choose from to treat IE. “This drug gives us another option,” he said.
Dr. Fraker said although the drug price point may play a role in treatment choice, isolating MRSA as the cause of bacteremia will be when doctors consider using the drug.
Ceftobiprole medocaril has already been used in Europe, Dr. Fraker noted. The drug was first approved by the European Medicines Agency in 2013 for the treatment of hospital-acquired pneumonia (excluding ventilator-associated pneumonia) and community-acquired pneumonia in adults. The U.S. indication is broader.
It’s not just the indication that is broader. Dr. Fraker said ceftobiprole medocaril may also be an option for physicians who want to use a broader-spectrum antibiotic to treat the infection.
Mandee Booth, PharmD, BCIDP, an assistant professor in the Department of Practice, Sciences, and Health Outcomes Research at the University of Maryland School of Pharmacy, in Baltimore, said the data on the drug published in The New England Journal of Medicine are “pretty compelling” (2023;389[15]:1390-1401). The drug is “a little bit more broad spectrum in terms of using it off the bat,” Dr. Booth said, and patient candidates might include people who can’t take other treatments for right-sided IE like daptomycin or vancomycin because of side effects such as rhabdomyolysis, renal dysfunction or any difficulties tolerating the drug.
Drs. Baturin, Booth and Fraker reported no relevant financial disclosures.
This article is from the October 2025 print issue.