By IDSE News Staff
The FDA issued a 510(k) clearance and Clinical Laboratory Improvement Amendments (CLIA) waiver for the bioMérieux Biofire Spotfire System and its Biofire Spotfire Respiratory (R) Panel.
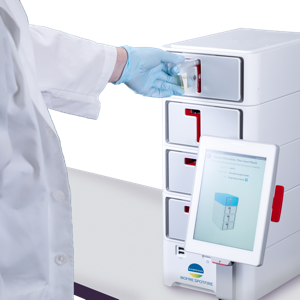
The COVID-19 pandemic demonstrated the need for healthcare professionals to have diagnostic tests available as close as possible to the patient, providing actionable results quickly. With the totally new and innovative Biofire Spotfire System and its Biofire Spotfire R Panel,
