Cefiderocol (Fetroja, Shionogi) has potent in vitro activity against Stenotrophomonas maltophilia isolates in the United States, including against those that were nonsusceptible to comparator antibiotics, according to new research presented at IDWeek 2025, in Atlanta.
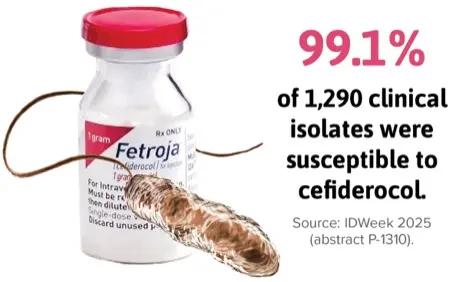
In a study of 1,290 clinical isolates collected from U.S. adult and pediatric patients (abstract P-1310), 99.1% of isolates were susceptible to cefiderocol. Susceptibilities to trimethoprim-sulfamethoxazole, levofloxacin, and minocycline were 97.7%, 81.8%, and 92.1%, respectively, investigators from Shionogi and Element Iowa City (JMI Laboratories) reported.
High Marks
Cefiderocol maintained high susceptibility (=96.7%) against 12 isolates that were nonsusceptible to trimethoprim-sulfamethoxazole, levofloxacin, and/or minocycline. Some 89 S. maltophilia isolates had a minimum inhibitory concentration (MIC) of at least 8 mcg/mL to aztreonam-avibactam (Emblaveo, AbbVie). Among these isolates, cefiderocol maintained 100% susceptibility, whereas susceptibilities to trimethoprim-sulfamethoxazole, levofloxacin, and minocycline were 95.5%, 73%, and 89.9%, respectively.
The most common infection types from which isolates were collected included pneumonia (n=942; 73%), skin and soft tissue infection (n=112; 8.7%), and bloodstream infection (n=100; 7.8%). Cefiderocol was the most potent agent tested, with MIC50/90 values of 0.06/0.25 mcg/mL. In comparison, MIC50/90 of aztreonam-avibactam was 2/4 mcg/mL.
Data Support Choosing Cefiderocol
The results may offer clinicians evidence to support the use of cefiderocol in adult patients with suspected resistant infections, including those caused by S. maltophilia, said Christine M. Slover, PharmD, the executive director of medical affairs at Shionogi.
“As antimicrobial resistance patterns are constantly changing, surveillance is essential to understanding the current real-world landscape of resistance to better inform treatment decision-making,” Dr. Slover said.
The study reinforces findings of initial surveillance studies that cefiderocol “is really an in vitro superstar when it comes to Stenotrophomonas,” commented Emir Kobic, PharmD, BCIDP, an antimicrobial stewardship and infectious disease pharmacist at Banner - University Medical Center Phoenix. “But in vitro data doesn’t always correlate with in vivo success, so we really need to see this in vitro data pair up with clinical success.”
Among the 12 isolates that did not respond to other agents, the MIC90 for cefiderocol increased to 1 mcg/mL, “which is right at the CLSI and FDA breakpoint,” Dr. Kobic told Infectious Disease Special Edition. “While all the isolates remained susceptible to cefiderocol in this study, the higher MICs that we saw in these multidrug-resistant phenotypes may suggest selective pressure toward resistance, but the overall number is too small to make any conclusion.
“We need continued surveillance, particularly for cefiderocol use in this population since, as we do in my setting, we usually reserve it for refractory or multidrug-resistant Stenotrophomonas infection,” he said.
Dr. Kobic has received honoraria from Shionogi. Dr. Slover is employed by Shionogi.
This article is from the February 2026 print issue.