Originally published by our sister publication Pharmacy Practice News
By Gina Shaw
A pharmacist-led penicillin allergy delabeling program for pediatric patients is safe, feasible and effective, with nearly all of the patients deemed eligible for the antibiotic class after oral challenge testing, according to new data reported at the ASHP Pharmacy Futures 2025 meeting, in Charlotte, N.C.
“There is now data showing that you can safely test low-risk pediatric patients, those who did not have SCAR [severe cutaneous adverse reaction] or severe anaphylactic reactions,” said Candice Mercadel, PharmD, a penicillin allergy and emergency department transition of care pharmacist at Parkland Hospital, in Dallas. “Our findings confirm that, with a 95% removal rate and no serious adverse reactions.”

Parkland’s initial delabeling program was started in 2014 for inpatient adults only, Dr. Mercadel noted. In 2022, the program was expanded to pediatric patients in the hospital’s community-oriented primary care clinic. But that initiative was suspended due to staff turnover and lack of interest, she said. In 2023, Dr. Mercadel launched a second pediatric delabeling clinic, which is ongoing.
In the study presented at the ASHP meeting, Dr. Mercadel summarized key data from that first effort (Clinic 1) and the ongoing program (Clinic 2).
In Clinic 1, 30 of 32 tested patients were negative for sensitivity to amoxicillin, a penicillin-class antibiotic. There was one immediate reaction, and follow-up calls revealed one delayed reaction, she reported. In Clinic 2, the deprescribing team so far has tested 41 patients, 39 of whom tested negative; there was one immediate reaction and one delayed reaction. All reactions were mild, involving pruritus, nonspecific rash or delayed rash, and did not require medications.
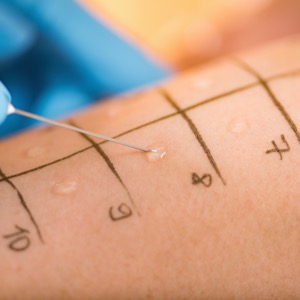
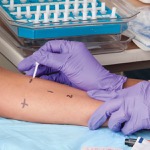
As for the specifics of the delabeling program, patients (aged 2-17 years) at Parkland are prescreened for delabeling eligibility during well-child visits; cold calls are also made by a medical practice assistant from an established list. The lead pharmacist is trained on allergy assessment, testing protocols and emergency preparedness, and required to complete annual continuing education on antimicrobial stewardship, drug hypersensitivity, and allergy and immunology.
Allergy tests are conducted in a once-monthly group visit, with five patients per session. Testing involves a graded oral challenge using amoxicillin, and patients are monitored for 65 minutes. If there is no reaction, the patient is discharged, with a follow-up call to assess for delayed reactions after one week. The patient’s local pharmacy is then called to remove the allergy from the patient’s records.
The group testing model is efficient and scalable, Dr. Mercadel suggested, with the most challenging element being parent counseling. “They may have been very upset by what happened the first time that led to their child being labeled as allergic to penicillin. Maybe they went to the emergency room,” she said. “So we need to talk them through the process and explain that it is very low risk, and that the child probably had a viral exanthem rather than a true IgE [immunoglobin E]–mediated anaphylactic reaction.
“The fact that we can get these patients delabeled at an earlier age helps their quality of life over the long term,” she added. “I’m seeing adult patients, both inpatient and outpatient, who have always believed that they were allergic to penicillin based on something they were told as a child. These labels have stuck with these patients for so long. Getting them delabeled in childhood helps to decrease adverse events, Clostridioides difficile infections, resistant pathogens and hospital lengths of stay, and allows us to provide patients with the most appropriate antibiotics.”
That reduced rate of C. difficile—a particularly lethal nosocomial infection—has been documented in previous research. For example, in a study by Turner et al (JAMA Netw Open 2021;4[5]:e219820), a pharmacist-led allergy assessment and penicillin testing program in a tertiary care hospital yielded significant reductions in hospital-acquired C. difficile infections (rate ratio, 0.61; 95% CI, 0.43-0.86).
The sources reported no relevant financial disclosures.