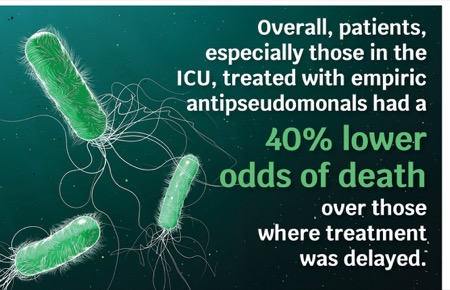
A recent study shows that the empiric use of antipseudomonal agents among ICU patients saves lives (J Antimicrob Chemother 2025;80[2]:322-333).
However, stewardship is important to assure that the agents are being prescribed to those who would benefit. Approximately 40% of patients admitted to the hospital with skin and soft tissue infections, and up to 75% of patients with urinary tract infections, are prescribed broad-spectrum antipseudomonal agents, but they have little effect on reducing mortality, the researchers found.
“The result of our meta-analysis shows that not all patients with infectious syndromes need to get antibiotics against Pseudomonas. Many infectious syndromes have either very low mortality rates or are very rarely due to Pseudomonas, and treatment with these agents yields a benefit that is not clinically meaningful and is harmful toward efforts at antibiotic stewardship,” senior study author Craig G. Gunderson, MD, an associate professor in the Department of Medicine at Yale School of Medicine, in New Haven, Conn., told Infectious Disease Special Edition.
In the analysis, Dr. Gunderson and his colleagues identified 27 studies involving 12,522 patients with infections from P. aeruginosa that reported the effect of active empiric antibiotics on mortality. Overall, patients treated with active empiric antibiotics had a 40% lower odds of death than those when antibiotics were delayed. However, the estimated absolute mortality benefit varied greatly among infection types depending on the risk for Pseudomonas and baseline mortality rate: 0.02% (95% CI, 0.02%-0.02%) for skin and soft tissue infections, 0.12% (95% CI, 0.10%-0.13%) for urinary tract infections and community-acquired pneumonia, and 0.3% (95% CI, 0.25%-0.34%) for sepsis without shock. “Even though there is a statistical reduction in mortality rates in these four cases, the difference is not clinically meaningful,” Dr. Gunderson explained. “A randomized controlled study designed to assess a difference in mortality of 0.1%, for example, would require almost 3 million patients.”
However, the researchers recorded a clinically significant benefit of empiric antipseudomonal use among more seriously ill patients. The mortality risk was reduced among study participants with septic shock by 1.1% (95% CI, 0.9%-1.4%) and 2.4% (95% CI, 1.9%-2.8%) for those with nosocomial pneumonia.
The authors concluded that when a patient is hospitalized with a condition that has an estimated 30% baseline mortality rate and there is a 5% prevalence of Pseudomonas infection, general use of broad-spectrum antipseudomonal antibiotics is more likely to have a meaningful benefit. Taylor Morrisette, PharmD, MPH, an associate professor of clinical pharmacy and outcomes sciences at the Medical University of South Carolina College of Pharmacy, in Charleston, agreed with this guidance and lauded the study for offering considerations in determining when the use of antipseudomonal antibiotics may be most warranted.
Dr. Morrisette, who was not involved in the study, added that although early drug initiation is important for all patients, “it is even more so in the critically ill population, because if you’re wrong, the patient is more likely to exhibit a higher degree of morbidity and mortality in comparison to more stable patients.”
In his own practice, Dr. Morrisette, who is also a member of the IDSE editorial advisory board, encourages the use of an antibiogram, which indicates local prevalence of pathogens and their susceptibility to different treatments, to determine the prevalence of Pseudomonas and optimal drugs for treatment. Rather than solely relying on national or international guidelines, he encourages other practitioners to determine the prevalence of Pseudomonas at their institutions and/or communities, as well as the risks and benefits, before they leap to use an antipseudomonal antibiotic for any patient admitted to the hospital. In fact, Dr. Morrisette believes that doctors have sufficient evidence to understand when and where antibiotics should be used to empirically treat Pseudomonas. Rather than refining this knowledge and guidelines, he said clinicians and health systems should work toward understanding and responding to localized risk for Pseudomonas in their communities.
Moving Toward Antibiotic Stewardship
Gregory Weston, MD, MS, an infectious disease specialist at the Albert Einstein College of Medicine, in New York City, who was not associated with the study, noted, “these data largely confirm the way that most infectious disease specialists practice.” Furthermore, he echoed Dr. Gunderson’s concern that overuse of antipseudomonal antibiotics is problematic and could lead to increasing Pseudomonas resistance.
Dr. Weston suggested that to improve patient outcomes and antibiotic stewardship simultaneously, the guidelines for these agents’ use could employ more clear and descriptive language about when a suspected infection does or does not warrant the use of antipseudomonal agents. However, he warned that “physicians need to do their part in understanding what the guidelines are trying to convey. It’s a two-way street.”
Dr. Gunderson told Infectious Disease Special Edition that he hopes this research will help reduce the overuse of antibiotics against resistant organisms, including Pseudomonas, especially for lower risk infections. He said he particularly wants this paper to inform updated Surviving Sepsis Campaign guidelines. The latest guidelines published in 2021 recommend withholding broad-spectrum antibiotics in low-risk patients. However, they don’t offer metrics for determining a patient’s risk (bit.ly/3HG1XAD-IDSE).
“I’m hoping this paper will actually fill in the blanks for that, because it has a quantitative way of estimating who’s high risk and low risk,” Dr. Gunderson said, “thus saving antibiotics for patients who are most likely to benefit from them.”
Drs. Gunderson, Morrisette and Weston reported no relevant financial disclosures.
This article is from the August 2025 print issue.