This article was updated on Oct. 2. There was an error in a quote that referenced "chloromycetin," instead of "clarithromycin." It was erroneous in press materials from the University of Michigan. We regret the error.
By IDSE News Staff
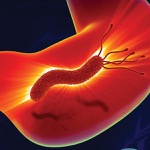
The American College of Gastroenterology updated its 2017 guideline for treating Helicobacter pylori (Am J Gastroenterol 2024;119[9]:1730 10.14309/ajg.0000000000002968).
Despite a decreasing prevalence, up to 40% of people in the United States have H. pylori; many of the infections are asymptomatic.
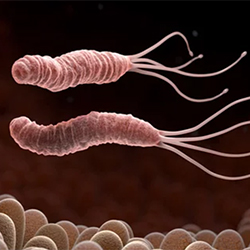
In the new guideline, the No. 1 recommendation for treatment-naive patients is bismuth quadruple therapy. That treatment typically includes a proton pump inhibitor (PPI), tetracycline, bismuth and a nitroimidazole for 14 days. The 2017 guideline maintained the recommendation of a PPI-clarithromycin triple therapy as an option.
“We were already recommending that healthcare providers move away from PPI triple therapy in 2017 because of increasing problems with chloromycetin resistance among H. pylori strains in the United States," said William Chey, MD, of the Division of Gastroenterology, Department of Internal Medicine, Michigan Medicine, in Ann Arbor.
“Despite that recommendation, PPI triple therapy still dominates first-line therapy prescriptions for H. pylori patients in the United States. In this newest iteration of the guideline, we are very clear to say that in essentially all circumstances you should not be prescribing PPI triple therapy, and you should be instead using bismuth quadruple therapy or one of the other suggested treatment options.”
The guideline makes 12 total treatment suggestions for patients in a variety of different situations.
The No. 2 recommendation for treatment-naive patients—after bismuth quadruple therapy—is rifabutin triple therapy (a PPI, rifabutin and amoxicillin).
A third option consists of a new, highly potent drug vonoprazan (Voquezna, Phathom Pharmaceuticals), which blocks stomach acid production, combined with the antibiotic amoxicillin.
Besides the move away from PPI triple therapy, another change from the 2017 guideline is the discussion of increasingly available molecular testing for antibiotic susceptibility.
“Molecular testing really opens the door to the possibility of more liberally utilizing antibiotic sensitivity testing as a mechanism of tailoring therapy to the antibiotics that H. pylori or a person infected with H. pylori is sensitive to,” Dr. Chey said.
The guideline also outlines future research priorities, such as identifying which individuals would most benefit from H. pylori testing to prevent gastric cancer and evaluating newly FDA-approved regimens for persistent infections.