From surveillance to stewardship, rapid diagnostic testing is becoming an integral tool for managing infectious diseases. These platforms identify pathogens more quickly, improving diagnosis and management to optimize patient care while curbing inappropriate antimicrobial use. Here is a quick roundup of some of the latest data from IDWeek 2023. There were quite a few interesting studies, so we split them into two parts. Part 2 will appear in our next issue.
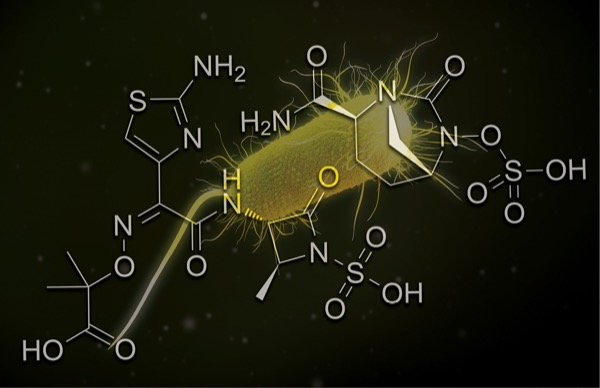
Aztreonam-Avibactam Effective Against Drug-Resistant P. aeruginosa Isolates
Aztreonam-avibactam (ATM-AVI) demonstrated in vitro activity against highly drug-resistant Pseudomonas aeruginosa isolated from contaminated artificial tears products (poster 289).
The drug-resistant P. aeruginosa were identified in samples from artificial tears products, which resulted in a large outbreak that affected people in 16 U.S. states during March 2023. The strain carried Verona integron–encoded metallo-beta-lactamase and Guiana extended-spectrum beta-lactamase, a combination that had not been identified in the United States.
ATM-AVI is in development for the treatment of infections caused by drug-resistant gram-negative pathogens, especially those coproducing metallo-beta-lactamases and other beta-lactamases.
Samples of the P. aeruginosa isolates were tested for susceptibility to ATM and ATM-AVI using broth microdilution and disk-diffusion methods manufactured by BD Biosciences and the United Kingdom’s Mast Group, as well as for susceptibility to cefiderocol (Fetroja, Shionogi) and ATM using Sensi-Discs by Hardy Diagnostics and ATM-AVI with E-test strips by Liofilchem Inc.
All tested isolates were susceptible to ATM-AVI based on proposed pharmacokinetic and pharmacodynamic breakpoints.
PCR Testing for RSV May Underestimate Prevalence Compared With Swab Testing With Luminex
Testing for respiratory syncytial virus (RSV) using sputum and/or saliva increases diagnostic yield, and results in more confirmed infections than the use of nasopharyngeal (NP) swabs alone (session 1019).
The findings likely illustrate that standard-of-care PCR testing of NP swabs for RSV underestimates RSV prevalence.
The prospective cohort study focused on patients at least 40 years old and hospitalized for acute respiratory infection in four hospitals in Louisville, Ky., during two seasons: Dec. 27, 2021 through April 1, 2022, and Aug. 22, 2022 through March 3, 2023.
Nasopharyngeal swabs, saliva and sputum samples were obtained and tested using the Luminex ARIES System.
A total of 2,798 patients were enrolled in the study. Nasopharyngeal swabs were collected in 100%, saliva in 99% and sputum in 31%. RSV was diagnosed by any specimen in 4.8% of patients, whereas 2.9% were diagnosed by NP swabs alone. This was consistent with a 66% increase in prevalence.
In the cohort of 856 patients who had all three specimens obtained, 7.2% were diagnosed by any specimen versus 4.3% diagnosed by NP swabs alone.
“Standard-of-care PCR testing of NP swabs for RSV underestimates RSV prevalence in adult patients hospitalized with ARI,” the authors wrote. “Hospitalized RSV ARI burden estimates in adults based solely on NP swab RT-PCR should be adjusted for underestimation.”
bioMÉrieux Automated Blood Culture Systems May Enhance Microbiological Diagnosis of Empyema
More than half of patients who underwent thoracentesis or percutaneous drainage, and whose pleural fluid cultures were positive when tested with an automated blood culture system (ABCS), had negative conventional culture results, according to data presented during session 1106.
The findings, the presenters wrote, suggest that ABCS may enhance microbiological diagnosis of empyema.
The single-center, retrospective study focused on data from patients whose pleural fluid was tested using both conventional culture and ABCS (BACT/ALERT 3D and VIRTUO, bioMÉrieux) from 2001 through 2021 at Samsung Medical Center, in Seoul, Republic of Korea.
A total of 9,020 patients were included. Of these, 819 patients had positive results—other than coagulase-negative staphylococci with ABCS.
While conventional culture was also positive in 44% of patients, 56% received positive results only from ABCS testing. Viridans group Streptococcus, Candida and Staphylococcus aureus were the most common species isolated only with ABCS, therefore, resulting in the discrepancy.
“Our results suggest that ABCS may enhance microbiologic diagnosis of empyema,” wrote the presenting author, Young Ho Lee, MD, a fellow in the Division of Infectious Diseases, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, in Seoul.
Use of IMMY Immunoassay May Change Management of Talaromycosis
An Mp1p lateral flow assay (LFA) is easy to use and effective in diagnosing talaromycosis, according to data presented during session 988.
Talaromycosis is an invasive mycosis caused by Talaromyces marneffei, and is a leading cause of AIDS-associated death in Southeast Asia. The infection also is diagnosed among people who do not have HIV infection, and incidence of the disease is expected to increase 35% by 2025.
However, diagnosis can be slow, the study authors wrote, with blood cultures taking four weeks to return results, and positive cultures observed in only 70% of patients.
“Lack of non-culture diagnostics is the greatest hurdle to reducing mortality,” the researchers added.
A previous study, published during 2021, found that a novel Mp1p antigen enzyme immunoassay (EIA) showed superior sensitivity compared with blood culture (Clin Infect Dis 2021;73[2]:e330-e336). In this nested case–control cohort study, the researchers analyzed use of the EIA and LFA, which was developed by IMMY.
A total of 132 hospitalized patients with advanced HIV and culture-proven talaromycosis were included, and compared with 160 controls, randomly selected patients who did not develop culture-proven talaromycosis over a six-month follow-up.
The Mp1p LFA and EIA had similar diagnostic performance. Sensitivity in plasma was 82% versus 84%, and specificity was 99% versus 98%. In urine, sensitivity was 90% versus 92%, and specificity was 99% versus 100%.
In addition, sensitivity was higher in urine versus plasma in both assays, and was higher still when testing plasma and urine concurrently.
Microbiocide Resistance Common Among Acinetobacter Isolates With Qiagen Test
Microbicide resistance genes are common in both carbapenem-susceptible and -resistant clinical isolates of A. baumannii (poster 713).
Commonly used hospital disinfectants include quaternary ammonium compounds (QACs) and the continuous use of these disinfectants, coupled with the ability of some micro- organisms such as Acinetobacter to persist on surfaces, may lead to reduced susceptibility of these organisms to disinfectants.
To clarify the effect of such cleaners on microbial resistance, the study focused on 139 Acinetobacter patient isolates taken from two hospitals in Detroit between 2017 and 2021. DNA isolation was conducted using the Qiagen DNA extraction kit, and the Nextera Flex kit was used for library preparation of Acinetobacter clinical isolates.
Half (50.4%) of the isolates were carbapenem-resistant A. baumannii (CRAb), and 49.6% were carbapenem-susceptible A. baumannii (CSAb).
Among the CRAb isolates, 56% had qacE genes, compared with 39% of CSAb that had qacE genes. About 42% of CRAb isolates were multidrug-resistant (MDR), indicating a prevalence of qacE genes among the MDR isolates.
“Our data suggest there is a prevalence of the qacE genes in the CRAb isolates versus the CSAb isolates,” said lead author, Piyali Chatterjee, MS, PhD, a senior research scientist at the Central Texas Veterans Health Care System, in Temple. “Among the CRAb isolates, some of them were MDR, indicating a prevalence of qacE genes even among the MDR isolates.
“Future studies are needed to determine the reduced susceptibility to other commonly used hospital disinfectants,” she added.
This article is from the December 2023 print issue.