By Marie Rosenthal, MS
We got it wrong. The acceptance was only for CHKV. We apologize for the error.
The CDC accepted the recommendation of the Advisory Committee on Immunization Practices (ACIP) for the chikungunya vaccine. It has yet to make a decision on the vaccines for respiratory syncycial virus and meningococcal diseases.
The CDC did not announce that the recommendation was accepted, but just added a two-line notice on the ACIP’s webpage, under the heading Recent Meeting Recommendations.
“ACIP approved the following recommendations by majority vote, and they have been adopted by the CDC Director. They will be published in MMWR [Morbidity and Mortality Weekly Report] and reflected in CDC’s print and digital resources in the coming months,” the statement said.
ACIP has had a rocky start under new Health and Human Services Director Robert F. Kennedy Jr. The meeting, which was supposed to take place in February, was postponed until April, and then the CDC took until May 15 to accept the recommendations.
The CDC director’s acceptance of the ACIP recommendations is the first step toward insurance coverage for vaccines; third-party payors tend to wait for that adoption before covering a vaccine, even though the FDA already approved it for marketing. The Affordable Care Act requires insurers to cover recommended vaccines in the next plan year.
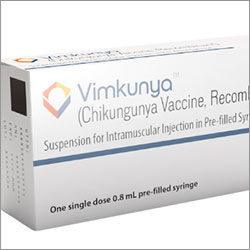
Chikungunya Vaccine
ACIP also voted to recommend the virus-like particle (VLP)-based chikungunya vaccine, recombinant (CHIK-VLP; Vimkunya, Bavarian Nordic) for certain populations. The CHIK-VLP vaccine, an adjuvanted VLP-based vaccine, is indicated for people 12 and older traveling to a country or territory where there is a chikungunya outbreak. In addition, CHIK-VLP may be considered for those traveling to or taking up residence for an extended period in a country or territory with an elevated risk for chikungunya, but is not experiencing an outbreak. Finally, ACIP recommended CHIK-VLP for laboratory workers with a potential for exposure to chikungunya virus.
Chikungunya is a mosquito-borne disease caused by the chikungunya virus, which typically presents with acute symptoms including fever, rash, fatigue, headache, and often severe and incapacitating joint pain. Most patients recover within one to two weeks, but up to 40% of those affected may develop chronic arthritis that can last for months or even years, according to the European Centre for Disease Prevention and Control.
In 2024, there were 620,000 cases of chikungunya reported worldwide, an increase of more than 20% compared with 2023, according to the European agency.
Regardless of the licensure pathway for any vaccine, they must demonstrate safety in well-controlled trials with a sample size that is large enough to determine this end point, explained Susan L. Hills, MBBS, MTH, a medical epidemiologist in the CDC’s Division of Vector-Borne Diseases, at the meeting.
In related news, the Department of Health and Human Services is expected to change the COVID-19 vaccine recommendations for pregnant women and children to remove the recommendation that they receive updated COVID-19 vaccines, according to an announcement by Leland Lehrman, the executive director of the new MAHA (Make America Healthy Again) Institute. The announcement was made during a panel event launching the institute.
The FDA’s Vaccines and Related Biological Products Advisory Committee is scheduled to meet on May 22 to discuss and make recommendations on the selection of the 2025-2026 formula for COVID-19 vaccines.