The COVID-19 pandemic may be over, but SARS-CoV-2 is still circulating. While not a reboot, this season’s sequel is likely to bring more hospitalizations and death among unvaccinated people due to decreased uptake of boosters, although admittedly it won’t be the megahit we saw during the height of the pandemic. So, how should we be managing “COVID-25”?
Although annual boosters of COVID-19 vaccination have been recommended, uptake has been declining, and the intensified controversy over vaccine guidelines this year is bound to reduce uptake even more, according to experts, which will affect those at higher risk.
“There will be preventable hospitalizations occurring because high-risk people are not vaccinated. We won’t see pandemic numbers, but we are likely to see more cases of COVID in the hospital because of this lack of vaccination,” said Amesh Adalja, MD, FIDSA, a senior scholar at the Johns Hopkins Center for Health Security, and an adjunct assistant professor at the Johns Hopkins Bloomberg School of Public Health, in Baltimore.
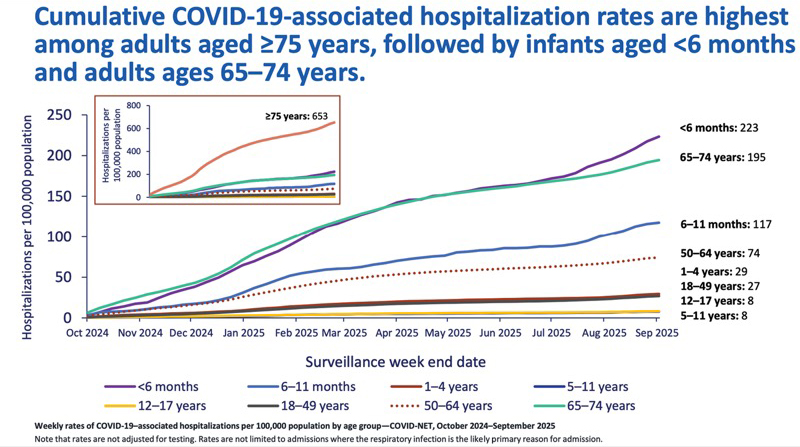
The effect of vaccination on hospitalization has been borne out by the data. Most COVID-19 hospitalizations are among unvaccinated or not fully vaccinated people, with the 12-month risk being highest among the youngest and oldest Americans, according to Arjun Srinivasan, MD, the acting chief medical officer of the CDC’s National Center for Immunization and Respiratory Diseases, who presented the data at the September Advisory Committee on Immunization Practices meeting.
This has also been the experience of healthcare professionals. “People who are being hospitalized in my neck of the woods tend to be the people in the high-risk groups—older, younger with chronic underlying conditions—who also are not up to date with their vaccines,” said William Schaffner, MD, a professor of infectious diseases, preventive medicine and health policy in the Department of Health Policy at Vanderbilt University, in Nashville, Tenn.
The primary goal of managing COVID-19 in 2025 is keeping people well enough that they don’t require hospitalization, which is why vaccination is still so important, experts said, but if they are hospitalized, the importance shifts to getting them stabilized and improved quickly and avoiding a stay in the ICU. Those goals also facilitate shorter lengths of stays and successful discharge, Dr. Gottlieb explained.
Prophylaxis
Outside of vaccination, most options today are treatments rather than prevention. However, pemivibart (Pemgarda, Invivyd) has an emergency use authorization (EUA) for the pre-exposure prophylaxis (PrEP) of COVID-19 in moderately to severely immunocompromised people 12 years of age and older. (The FDA declined to expand the EUA to include mild to moderately immunocompromised people.)
Immunobridging was used to grant the EUA, looking at the relationship between neutralizing antibody titers and clinical efficacy with other monoclonal antibodies (mAbs) against SARS-CoV-2. The titers achieved against the JN.1 variant for 90 days after administration of pemivibart were consistent with the titer levels associated with efficacy of other SARS-CoV-2–targeting mAbs (Nat Commun 2023;14[1]:4545). Invivyd is also working with the FDA for full approval of its novel mAb, VYD2311.
However, pemivibart is not a replacement for vaccination, according to Dr. Adalja. “Pemgarda should be used in tandem with vaccination: It is not an either-or,” Dr. Adalja explained, and elaborated that these are complementary products meant to boost immunity among people who will not mount a sufficient immune response to vaccination alone.
These are patients with the highest risk for hospitalization and death, according to Robert L. Gottlieb, MD, PhD, FACC, FAST, FIDSA, a transplant cardiologist with expertise in virology and genetics on the medical staff at Baylor Scott & White The Heart Hospital in Dallas and Plano, and Baylor University Medical Center in Dallas.
However, if a patient declines to be vaccinated, even after counselling, would they prescribe pemivibart? That is tough to answer because pemivibart is available under an EUA, and is not authorized for all patients.
“We want to be patient-centric,” Dr. Gottlieb said. “But pemivibart is focused on patients that are moderately to severely immunocompromised or unlikely to generate an adequate immune response as their risk factor. Because it has an emergency use authorization without a full label, it may only be used according to the terms of the EUA.”
Adarsh Bhimraj, MD, FIDSA, the director of the Infectious Diseases Fellowship and Education Programs at Houston Methodist Hospital, agreed with Dr. Gottlieb’s assessment. “If a drug has full FDA approval, physicians may sometimes use it off-label. But when a medication is authorized only under an Emergency Use Authorization, it must be prescribed strictly within the terms of that authorization,” said Dr. Bhimraj, the lead author of the Infectious Diseases Society of America (IDSA) COVID-19 treatment guidelines.
Receiving pemivibart every three months is costly and inconvenient, underscoring the importance of using it only as authorized under the EUA, Dr. Bhimraj added.
“In general, I target it to my more high-risk patients, especially for the first few seasons. I work in a heart and lung transplant program, and so lung transplant patients are at a higher risk, for example,” Dr. Gottlieb said. “Additionally, recipients of recent stem cell transplant are at particularly high risk as they have to redevelop an immunologically naive immune system.”
An oral option in the pipeline that excites experts is ensitrelvir (Shionogi), a 3CL protease inhibitor that suppresses replication of SARS-CoV-2 and, if approved, would be used as post-exposure prophylaxis. The FDA has accepted the New Drug Application for ensitrelvir with a decision deadline of June 16, 2026.
Shionogi evaluated the safety and efficacy of ensitrelvir through SCORPIO-SR, a phase 3 study that showed both clinical and symptomatic efficacy for five typical omicron-related symptoms (primary end point) and antiviral efficacy (key secondary end point) in a predominantly vaccinated population of patients with mild to moderate SARS-CoV-2 infection, regardless of risk factors (JAMA Netw Open 2024;7[2]:e2354991).
In addition, the global double-blind, randomized, placebo-controlled phase 3 SCORPIO-PEP study indicated that ensitrelvir prevented symptomatic COVID-19 through day 101. There were no hospitalizations or deaths related to COVID-19.
“I think one of the exciting aspects of this antiviral—one that really differentiates it from others—is they use it in a post-exposure prophylaxis study. Someone gets exposed to COVID, they take the antiviral and they’ve been able to show a very significant decrease in people catching COVID if they take this antiviral,” Dr. Adalja said. “This will be another tool we can use.”
Treatment Options
There are important differences in the way COVID-19 is treated today compared with during the height of the pandemic, the experts who spoke with Infectious Disease Special Edition explained. Healthcare providers know more about the virus, more people have some immunity through vaccination and/or previous infection, and the circulating variants interact with immune experienced hosts, resulting in less virulence.
“In hospitalized COVID-19, the key is the stage of illness,” Dr. Bhimraj said. “Patients who do not need oxygen or only need low-flow support are treated differently from those requiring high-flow oxygen or mechanical ventilation, because their risks and outcomes are very different. “There is evidence for dexamethasone in severe and critical COVID-19, and for Janus kinase or interleukin-6 inhibitors in addition to dexamethasone in patients who are worsening or critically ill,” he added.
Of the drugs with a specific indication for COVID-19, remdesivir (Veklury, Gilead) was the first medication to receive an FDA approval versus an EUA.
The approval was supported by an analysis of data from three randomized, controlled clinical trials that included patients hospitalized with mild to severe COVID-19. The studies found a reduction in time to recovery, which was defined as discharge from the hospital or continued hospitalization, but not requiring supplemental oxygen, compared with placebo (N Engl J Med 2020;383[19]:1813-1826).
“For patients hospitalized with severe COVID-19 who require supplemental oxygen by nasal cannula, remdesivir can be given for five days,” Dr. Bhimraj said. “For outpatients with mild to moderate COVID-19 who are at high risk for progression to severe disease, a three-day course may be used, and there may still be benefit in these high-risk groups even in the current phase of the pandemic.” Dr. Gottlieb said he would consider remdesivir in the outpatient setting for patients with risk factors for progression who are not good candidates for nirmatrelvir+ritonavir (NMV+RTV; Paxlovid, Pfizer) (N Engl J Med 2022;386[4]:305-315).
After being used for several years under an EUA, the oral antiviral combination NMV+RTV was approved by the FDA to treat mild to moderate COVID-19 in adults at high risk for progression to severe disease. In studies, NMV+RTV had an 89% reduction in risk for hospitalization and death among people who were unvaccinated. That risk reduction was high enough to prompt the National Institutes of Health to prioritize it over other COVID-19 treatments, according to the FDA. It is also available under the EUA for people 12 to 18 years of age.
The efficacy of NMV+RTV was supported primarily by the final results of the EPIC-HR clinical trial, a randomized, double-blind, placebo-controlled clinical trial studying NMV+RTV for the treatment of nonhospitalized symptomatic adults with a laboratory confirmed diagnosis of SARS-CoV-2 infection. All patients were unvaccinated with no history of previous SARS-CoV-2 infection. NMV+RTV significantly reduced the proportion of people with hospitalization or death related to COVID-19 by 86% compared with placebo, if given within five days of symptom onset (N Engl J Med 2022;386[15]:1397-1408).
Because of drug–drug interactions, however, NMV+RTV is not appropriate for all patients.
“For patients that have a risk factor for progression and do not have a drug–drug interaction or have a manageable drug–drug interaction, those remain the patients that would be appropriate for ritonavir-boosted nirmatrelvir,” Dr. Gottlieb said. “Now, unfortunately, many of my patients can’t receive Paxlovid because they do have unmanageable drug–drug interactions … and those are the patients that would benefit from remdesivir.”
Another option is molnupiravir (Lagevrio, Merck/Ridgeback), which in clinical trials showed a risk reduction of hospitalization or death of about 50%, but is still only available under an EUA.
Patients who cannot take NMV+RTV or receive outpatient remdesivir could be candidates for molnupiravir.
Dr. Gottlieb, however, disagrees. Monupirivir is a special class because it is a viral mutagen, instead of antiviral in the classic sense, and in a Nature study was shown to accelerate viral evolution (2023;623[7987]:594-600) including the risk for promoting escape mutations. “For this reason, I have never prescribed it,” he told IDSE.
Another option that Vanderbilt’s Dr. Schaffner called “provocative” is the use of azelastine nasal spray. As IDSE was going to press, a study was published using the over-the-counter spray to prevent SARS-CoV-2 infection. In the phase 2, double-blind, placebo-controlled, single-center trial, participants were randomly assigned to use azelastine or placebo three times per day for 56 days. There was a significant decrease in the incidence of confirmed COVID-19 infection in the azelastine group than the placebo arm (JAMA Intern Med 2025 Sep 2:e254283).
This is an early study in just one institution, so the findings need to be confirmed, but Dr. Schaffner said there is a biological reason it could be effective. “These viruses implant on the respiratory mucosa,” he explained; therefore, a nasal spray may keep the virus off the mucosal surface preventing the virus from establishing itself. “I think it’s very interesting,” he said.
It is reassuring that researchers have not turned their back on this virus, and people are still looking for more effective prophylaxis and treatment.
Be Prepared
Regardless of patients’ vaccination status, Dr. Gottlieb advised physicians to ensure immunocompromised patients understand their risks. “It is absolutely critical that patients with significant risk factors for progression, such as a solid-organ or stem cell transplant recipient, know that there are options for them—that they are informed ahead of time and have an action plan about what they can and cannot take if and when they turn positive,” he said.
And more help is coming for clinicians. Dr. Bhimraj said updated IDSA guidelines will be released, which he and Rajesh Gandhi, MD, a professor of medicine at Harvard Medical School in Boston, will discuss at IDWeek 2025 on Oct. 22.
Finally, don’t forget nonpharmaceutical options, Dr. Schaffner concluded. Respiratory season is a time to dust off those masks and remember the importance of hand hygiene and social distancing.
Dr. Adalja serves as a consultant to Invivyd and Shionogi. Dr. Gottlieb reported grants or contracts to his institution from AstraZeneca, Eli Lilly, Gilead Sciences, Johnson & Johnson, Kinevant Sciences, Pfizer, and Regeneron; participation on advisory boards and/or consulting with AbbVie, AstraZeneca, Eli Lilly, Gilead, GSK Pharmaceuticals, Invivyd and Roche; honoraria for lectures from Gilead and Pfizer (the latter unrelated to infectious diseases); prior de minimis investment in AbCellera; and a gift in kind to his institution from Gilead Sciences to facilitate an unrelated academic-sponsored clinical trial (NCT03383419). Drs. Bhimraj and Schaffner reported no relevant financial disclosures.
This article is from the October 2025 print issue.