By IDSE News Staff
The FDA has approved a new COVID-19 vaccine from Moderna, mNEXSPIKE (mRNA-1283).
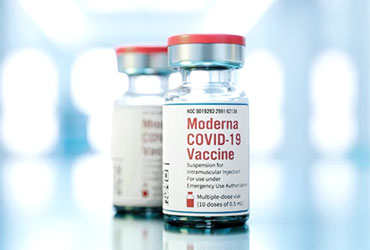
The vaccine is indicated for people older than 65 years of age or those 12 between 64 years who have one underlying risk factor as described by the CDC.
The approval follows positive results of the vaccination’s randomized, observer-blind, active-controlled phase 3 trial (ClinicalTrials.gov Identifier: NCT05815498) of 11,400 participants aged 12 years and older. The trial—which had an active comparator arm to Moderna’s previously approved COVID-19 vaccine, mRNA-1273 (Spikevax)—produced data showing the new shot had higher efficacy than the original vaccination, with a similar safety profile. In the trial, participants received either a 10-mcg dose of mRNA-1283 or a 50-mcg dose of mRNA-1273. The new shot showed a 9.3% higher relative vaccine efficacy compared with the original shot in participants aged 12 years and older, and a subgroup analysis found a 13.5% higher relative vaccine efficacy in favor of the new shot in those aged 65 and older.
Safety was similar for mRNA-1283, with fewer local reactions and comparable systemic reactions. Most common side effects were injection site pain, fatigue, headache and myalgia. The shot, like other COVID-19 vaccines, comes with a warning for severe allergic reaction, and includes a warning that myocarditis and pericarditis has occurred in some individuals after mRNA COVID-19 vaccination, particularly in males between the ages of 12 and 24 years.
The company expects to have mRNA-1283 available in the United States for the 2025-2026 respiratory virus season, along with its original COVID-19 vaccine and its FDA-approved respiratory syncytial virus vaccine.
Based on a Moderna press release.