By IDsE News Staff
Interruptions in the supply of BD Bactec blood culture media bottles because of recent supplier issues could affect patient diagnoses, follow-up management and antimicrobial stewardship efforts, according to a recent letter the FDA sent to laboratories and healthcare providers (https://www.fda.gov/medical-devices/letters-health-care-providers/disruptions-availability-bd-bactec-blood-culture-media-bottles-letter-health-care-providers).
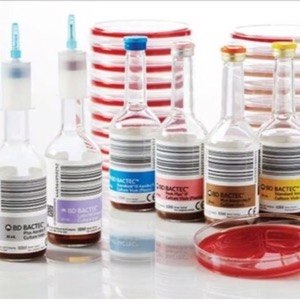
On July 10, 2024, the FDA updated the Medical Device Shortages List to include blood culture media bottles (product code MDB).
BD said in a statement that the company is working to meet the global demand and taking measures to address the shortage by allocating the resource manually. More information on BD Bactec blood culture vial supply can be found at BDBACTEC-update.com.
The FDA recommended these healthcare staff consider conservation strategies to prioritize the use of blood culture media bottles, preserving the supply for patients at highest risk. In developing strategies to preserve the supply for patients at highest risk, the agency suggested:
- performing blood culture collections when medically necessary, following clinical guidelines, such as those provided below;
- prioritizing use for patients with clinical signs and symptoms of a bloodstream infection;
- performing routine disinfection of skin protocols before collection to minimize the risk for contamination of the blood culture;
- ensuring proper blood volume collection to avoid a need to re-collect additional samples; and
- utilizing safe blood collection and transfer devices to minimize the risk for damage to blood culture media bottles.
Refer to the following guidelines for best practices for blood collection and potential considerations for prioritization for use of blood culture media bottles:
Guide to Utilization of the Microbiology Laboratory for Diagnosis of Infectious Diseases: 2024 Update by the Infectious Diseases Society of America (IDSA) and the American Society for Microbiology (ASM) (Clin Infect Dis 2024 Mar 5. https://doi.org/10.1093/cid/ciae104).
WHO guidelines on drawing blood: best practices in phlebotomy
The CDC also has resources:
- Preventing Adult Blood Culture Contamination: A Quality Tool for Clinical Laboratory Professionals
- Blood Culture Contamination: An Overview for Infection Control and Antibiotic Stewardship Programs Working with the Clinical Laboratory
The agency said it would continue to monitor the situation.
“BD is experiencing reduced availability of blood culture vials from our supplier. We understand the critical role that blood culture testing plays in diagnosing and treating infections and are taking all available measures to address this important issue, including providing the supplier our manufacturing expertise, using air shipments, modifying BD manufacturing schedules for rapid production, and collaborating with the U.S. Food and Drug Administration to review all potential options to mitigate delays in supply,” said Nikos Pavlidis, the worldwide president of BD Diagnostic Solutions, in a statement. “As an additional stopgap measure, our former supplier of glass vials will restart production to help fill the intermittent gap in supply.
“While this supplier issue is not expected to have a material financial impact on BD, we are fully focused on returning the supply of blood culture vials to normal levels,” Mr. Pavlidis said.
This shortage does not affect BD Vacutainer blood collection tubes.
From FDA and company materials.