Hologic Panther Fusion Assay Demonstrates High Sensitivity
The Panther Fusion SARS-CoV-2/Flu A/B/RSV assay (Hologic) detected respiratory viruses from nasopharyngeal samples (P0170; ESCMID).
The Berlin study tested leftover positive samples of SARS-CoV-2 (n=102), influenza A (n=99), influenza B (n=43) and respiratory syncytial virus (RSV) (n=93), as well as 100 samples that were negative for the four pathogens. The results from the assay were compared with those from the Abbott Alinity m Resp-4-plex (Abbott Laboratories) and the Allplex RV Master Assay (Seegene).
Both the Panther-4plex and the Allplex assays confirmed the negative results of all 100 pretested negative samples. The Panther assay showed a specificity of 100%, and a higher sensitivity for SARS-CoV-2, influenza A, influenza B and RSV than the Allplex assay. However, the sensitivity was lower when compared with the Alinity-4plex assay.
ACC Positioned to Help More Biotech Companies Reduce Supply Chain Risk
The U.S. Pharmacopeia Microbiology Expert Committee will permit the use of reagents for endotoxin testing that are not derived from animals, according to an inclusion in USP Chapter <86> Bacterial Endotoxins Test Using Recombinant Reagents. Pharmaceutical, and medical device manufacturers can transition their bacterial endotoxin testing (BET) from naturally sourced reagents from animals to recombinant cascade (rCR) and recombinant Factor C (rFC) BET reagents.
Bacterial endotoxin testing has been based on naturally sourced Limulus Amebocyte Lysate (LAL). This traditional method has been the gold standard in Quality Control for pharmaceutical manufacturing, but it relies on horseshoe crab blood as a raw material. With ACC’s introduction of PyroSmart NextGen rCR in 2021, non-animal derived BET testing provides greater availability, reliability and robustness as a direct reagent replacement for traditional BET methods.
PyroSmart NextGen is more reliable and less costly than LAL reagents. In addition, synthetic reagents will be less vulnerable to raw material availability with a more sustainable and robust supply chain, helping pharmaceutical and medical device manufacturers respond quickly to emerging threats to human health while decreasing the BET industry’s overall impact on the U.S. horseshoe crab population.
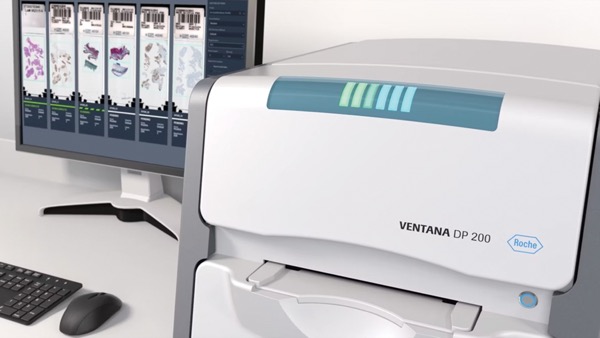
Roche Receives FDA Clearance on Its Digital Pathology Solution
The FDA recently cleared the Roche Digital Pathology Dx (VENTANA DP 200) intended to aid the pathologist in reviewing and interpreting digital images of scanned pathology slides to help diagnose patients.
Digital pathology refers to the digitalization of the traditional pathology workflow, starting from slide scanning, to visualization, to analysis. Digital pathology is transforming traditional histopathology by improving efficiency, depth of analysis and opportunity for collaboration in pathology workflows. For example, once a slide scanner captures and converts stained tissue on glass slides to digital images, these images can be managed, shared and analyzed by pathologists and can help determine a cancer patient’s treatment.
“Primary diagnosis for digital pathology streamlines the digital workflow that empowers pathologists to make a timely diagnosis from anywhere,” said Jill German, the head of Pathology Lab at Roche Diagnostics.
This article is from the October 2024 print issue.