For more on avian flu, check out The Avian Flu Outbreak That Just Won't Die Out.
If H5N1 were on a soccer team, as one expert described it, it just might be able to score a goal: human-to-human transmission.
With each mammal that becomes infected with H5N1 avian influenza virus, the concern for a human pandemic grows, especially since it has appeared in dairy cows because these animals do not normally get influenza (Viruses 2019;11[6]:561).
In fact, it was an astute veterinarian, Barb Petersen, DVM, who identified H5N1 in U.S. dairy cows in 2024 after ruling out other causes of the signs she was seeing in her patients. Dr. Petersen owns and operates Sunrise Veterinary Service, in the Texas Panhandle, which serves about 40,000 dairy cattle.
“With the virus now present in the cows, the potential number of contacts between the H5N1 and humans increases, [which increases] the potential number of infections in humans,” explained Adolfo Garcia-Sastre, PhD, a professor in the Department of Microbiology and the director of the Global Health and Emerging Pathogens Institute of the Icahn School of Medicine at Mount Sinai, in New York City.
There is concern that dairy cows may serve as the “mixing vessel” where avian influenza viruses could produce novel strains that are more adaptable to human-to-human transmission, he said. This process, called reassortment, could also occur in a person infected with a virus from the cow and a seasonal human flu virus. “We don’t know how many mutations the virus needs to get to transmitting from human to human, but certainly it already has some mammalian adaptations,” Dr. Garcia-Sastre said during a webinar sponsored by the WHO, which brought together international experts to share current scientific data and figure out knowledge gaps and research priorities to prepare for the possibility that H5N1 could cause human outbreaks.
At this point, “it is not a matter of if, but when,” said Maria Van Kerkhove, PhD, an infectious disease epidemiologist who serves as the head of the WHO Emerging Diseases and Zoonoses Unit.
The genome for influenza has eight discrete RNA segments, and if an animal or human is infected with two different lineages of the virus at the same time, and a cell ends up with these two different viruses, a “grab bag of different combinations” could occur to these segments, explained Michael Worobey, DPhil, a professor and the head of the Department of Ecology and Evolutionary Biology at the University of Arizona, Tucson.
“H5N1 is pretty dramatically expanding its host range, and in doing so, is picking up mutations that make the virus work better in mammals,” Dr. Worobey said. This development gives the virus more opportunities to score a goal, he added. “This is how pandemics get started. They start with a shot at a goal that actually makes it into the net.”
Since the goose/Guangdong-lineage of H5N1 avian flu virus was first detected in domestic geese in 1996, it has been near impossible to extinguish it (bit.ly/4ckGI21-IDSE). Since 2020, a variant of these viruses has led to an unprecedented number of deaths among wild birds and domestic poultry. And Europe and North America, including the United States, have observed an extended epidemic of avian flu with unusual persistence among wild bird populations since 2021, according to the WHO.
Reports among mammals, including seals, sea lions, foxes, bears, otters, raccoons, cats, dogs, cows, goats and others, also have increased (bit.ly/42no1q8-IDSE).
Continued Surveillance
The consensus of the meeting was that the world can expect more outbreaks from avian influenza viruses, certainly among animals, but it is becoming more likely in humans, too. Therefore, every country needs to continue to strengthen surveillance, as well as look for better ways to prevent and to treat zoonotic influenza.
The WHO’s Dr. Van Kerkhove said there should be a strategic shift from working on individual priority pathogens to including entire viral families, which would be important “because there is uncertainty about which pathogen in each individual family will cause the next phase of the pandemic.”
Moving forward, surveillance will be key. Continued surveillance of avian flu is critical in wild birds, other wild animals, and domestic animals, livestock and poultry, according to Dr. Worobey.
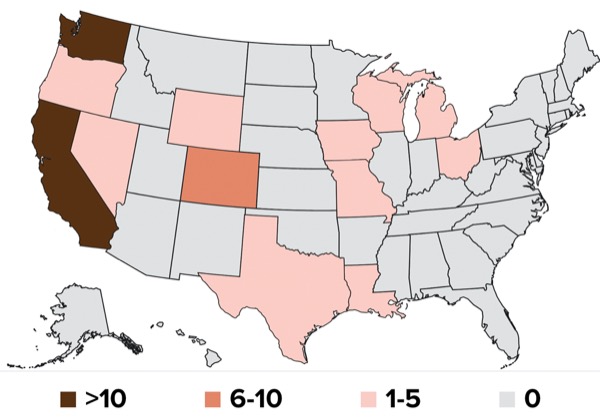
“If you don’t have surveillance in the wild bird population and mammals other than dairy cattle, you wouldn’t be able to understand anything about how this virus has gotten into these animals, and when, where and how it’s then moved across time,” Dr. Worobey said. “We need real-time monitoring of dairy cattle and other domestic animals with sensitive rapid diagnostics that are easy to deploy in the field.”
Possibilities for increased surveillance include wastewater surveillance, bioformatics and meta-genomic sequencing.
So far, high levels of resistance do not appear to have developed against the available antivirals, particularly the polymerase acidic endonuclease inhibitors like baloxavir marboxil (Xofluza, Roche/Shionogi) and neuraminidase inhibitors such as laninamivir, oseltamivir, peramivir (Rapivab, BioCryst) and zanamivir (Relenza, GSK). However, there is a high level of resistance in a lot of circulating flu viruses to the M2 proton channel antagonists such as amantadine and rimantadine, which is why they’re not widely used, according to Richard J. Webby, PhD, the director of the WHO Collaborating Centre for Studies on the Ecology of Influenza in Animals and Birds, and a member of the staff at St. Jude’s Children’s Research Hospital, in Memphis, Tenn.
However, mutations with resistance, even to baloxavir, are found sporadically, he said. Therefore, it is imperative that surveillance continues and pheno- typing is also done of viruses containing less described mutations, he suggested.
Cidara Therapeutics is one company developing a new antiviral against seasonal and pandemic influenza. CD388 is a drug–Fc conjugate (DFC) comprising multiple copies of a small molecule neuraminidase inhibitor that is conjugated to a proprietary Fc fragment of a human antibody. DFCs are low–molecular-weight biologics designed to function as long-acting inhibitors. The hope is it will provide universal protection against all known strains of seasonal and pandemic influenza by directly inhibiting viral proliferation.
Jaap Goudsmit, MD, PhD, a professor of epidemiology and immunology at Harvard T.H. Chan School of Public Health, in Boston, shared lessons from his 50-plus years in the discovery and development of monoclonal antibodies across rabies, SARS, SARS-CoV-2 and influenza. He discussed the importance of maximizing antigenic coverage and reducing the risk for viral resistance through the use of broadly neutralizing antibodies and antibody regimens.
Dr. Goudsmit warned of vaccine interference seen with some systemically administered antibodies, and highlighted the lack of tools to provide fast protection against infection, rather than severe disease, following spillover events.
Looking ahead toward pandemic preparedness against H5, he sees autonomously administered antivirals as a complement to clinician-administered vaccination. He highlighted research into intranasal administration of antibodies like CR9114 (Leyden Labs). “Broadly protective nasal sprays could enable stockpiling and fast rollout in case of spillover of H5N1, but also of other pre-pandemic zoonotic subtypes like H5N8, H3N8, H7N7 or H7N9,” he said.
Another problem with antivirals is they usually need to be prescribed early in the course of disease.
Frederick G. Hayden, MD, a professor emeritus at the University of Virginia School of Medicine, in Charlottesville, said that in many ways the readiness of the world against a new pandemic is more advanced than it was 20 years ago, citing direct-acting antivirals and monoclonal antibodies. “But there are a lot of unanswered questions in the event of a new emergency pandemic, such as: Are we going to need to have different use of drugs in terms of higher doses or prolonged therapy for both prophylaxis and treatment? We don’t know the susceptibility of that virus in treated patients,” he said. In addition, they don’t know this: “How fast will resistant variants come up?”
Dr. Hayden also highlighted the importance of therapeutics during the current interpandemic period, characterized by occasional spillovers of H5N1 into humans. ”Interpandemic” preparedness would also be key in preventing the next pandemic.
Vaccination
Vaccines are always an important player when discussing influenza. The United States has licensed and is stockpiling three vaccines against H5. The European Medical Association also has authorized vaccines against H5 viruses. Most of these vaccines are indicated for those with the highest risk for exposure, such as people who work on dairy or poultry farms, rather than for the general population.
It is likely that mRNA technology also will play an important role in creating an avian flu vaccine, if needed, because it can be modified quickly against any mutations. However, mRNA vaccines were not embraced by everyone during COVID-19, so a pressing question that needs answering is: Are we ready for the next generation of vaccines?
“The mRNA platform has an advantage over currently licensed influenza vaccines in terms of the potential speed of production of the vaccines to combat both seasonal as well as pandemic influenza,” explained Kelly Lindert, MD, the vice president of Clinical Research and Development at Pfizer, which was working on an mRNA influenza vaccine before developing its COVID-19 vaccines.
The duration of protection, best type of immunity and best possible correlates of protection are all questions requiring research for future vaccines, the experts said.
The United States is also stockpiling antigens and adjuvants to enable faster production of H5 vaccines that better match circulating strains (bit.ly/3G0Ig5p-IDSE). There is a list of all candidate vaccines for zoonotic influenza viruses, including H5N1, which was developed by the WHO and is updated regularly (www.who.int/teams/global-influenza-programme/vaccines).
As shown by the veterinarian who discovered avian flu in dairy cows, Dr. Petersen, a One Health approach will be crucial for all aspects of preparedness, they all agreed.
“I think the lesson across influenzas, including H5N1, is that they are difficult to durably treat or prevent with any modality because of the capacity for rapid antigenic drift,” said Les Tari, PhD, the chief scientific officer at Cidara Therapeutics. “That is why vaccine efficacy is so variable.”
The sources reported no relevant financial disclosures outside their places of employment. Dr. Garcia-Sastre is a co-inventors of influenza vaccine technologies owned by the Icahn School of Medicine at Mount Sinai. Dr. Goudsmit is the chief science officer of Leyden Laboratories with stock options in this company.
This article is from the April 2025 print issue.