By IDSE News Staff
The FDA cleared the BioFire Spotfire Respiratory/Sore Throat (R/ST) Panel. It also received a Clinical Laboratory Improvement Amendments (CLIA) waiver approval.
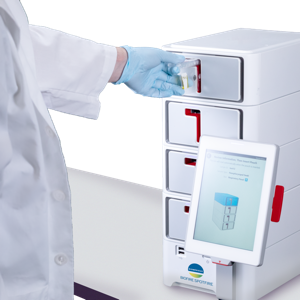
The COVID-19 pandemic has demonstrated the need for healthcare professionals to have diagnostic tests available as close as possible to the patient, providing actionable results quickly. A fast and innovative syndromic testing range, BioFire Spotfire matches these new medical needs worldwide.
