Treating invasive fungal disease (IFD) for patients with acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS) before confirmatory tests is more effective than waiting for a test to confirm breakthrough disease (Microorganisms 2024;12[1]:117).
For example, one study that was part of the literature review found that among 549 AML/MDS patients undergoing induction chemotherapy or allogeneic stem cell transplantation, those who were treated with fluconazole prophylaxis preemptively had higher overall survival and lower IFD rates than patients treated empirically for IFD (96.7% vs. 93.1% and 7.7% vs. 6.6%, respectively) (Clin Infect Dis 2023;76[4]:674-682).
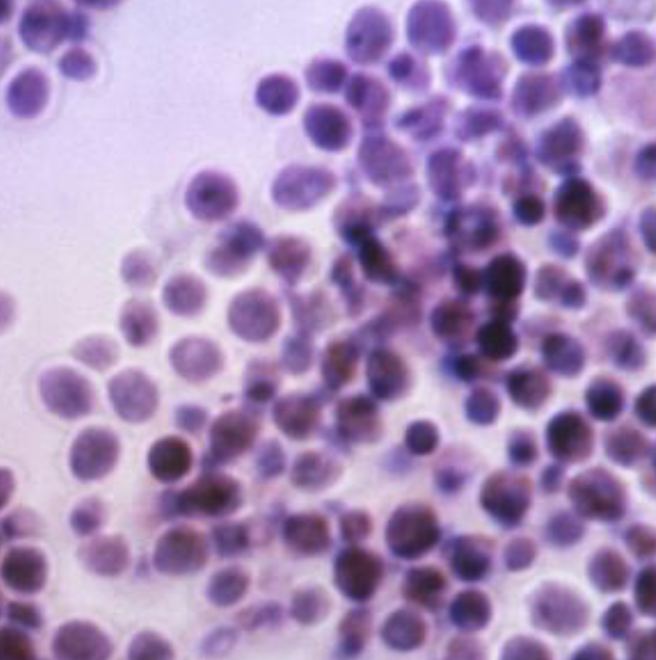
The researchers compared these findings with their own retrospective study of 184 adults with AML/MDS treated with posaconazole at San Raffaele Hospital, in Milan, between 2010 and June 2023. All participants underwent high-dose induction, consolidation and/or salvage chemotherapy with remission or cure as a goal. They analyzed 339 chemotherapy treatments: 153 induction treatments, 126 consolidation treatments and 60 salvage treatments. Six cases of breakthrough IFDs were recorded in six patients, with an overall incidence rate of 1.7% among all treatments. The incidence rates of breakthrough IFDs (per 100 person-years follow-up) were 5.04 among induction (n=2), 3.25 in consolidation (n=1) and 18.38 in salvage chemotherapy (n=3). At week 12, all patients with breakthrough IFDs achieved treatment success and continued their hematologic therapy. Researchers concluded that results suggest that “broad-spectrum AFP [alpha-fetoprotein] may reduce the burden of IFDs, resulting in less severe forms [of disease].”
Tom Chiller, MD, MPHTM, the chief of the CDC’s Mycotic Diseases Branch, emphasized the importance of prophylaxis in this fragile patient population. “These are incredibly sick people that can get sick very fast with these mold infections. They can die quickly, and mortality can be very high because we don’t have good ways to diagnose them. They need prophylaxis and this study confirms that,” said Dr. Chiller, who was not associated with either study.
Despite the efficacy of prophylaxis identified by the study’s authors, they warned that because of “some limitations of current antifungals (e.g., route of administration, toxicity or drug-to-drug interactions) and the emergence of resistant fungi, there is still an urgent need to expand our armamentarium, and several compounds are reaching the stage of phase 2 or 3 clinical assessment. These include new drugs within the existing antifungal classes or displaying a similar mechanism of activity with improved pharmacologic properties (rezafungin [Rezzayo, Melinta] and ibrexafungerp [Brexafemme, Scynexis]) or first-in-class drugs (olorofim and fosmanogepix).”
Dr. Chiller also said more diagnostics are needed, emphasizing the importance of rapid diagnostics to enable physicians to use a screen-and-treat strategy that would allow the sparing use of antifungals to lower the chances for resistance developing.
“It certainly would be helpful if the U.S. and other governments help industry to develop these assays. This is particularly important because the number of people who receive organ transplants [or] take biologics and other immunosuppressive therapies is growing every year.
“Essentially, the risk group is growing, and one of the best ways to help mitigate that risk is to screen these patients, diagnose them as early as possible and have the ability to use the drugs we do have in a targeted way,” Dr. Chiller concluded.
The sources reported no relevant financial disclosures.
This article is from the December 2024 print issue.