By Ethan Covey
The risks for secondary transmission of the vaccine-strain varicella-zoster virus (Oka strain; vOka) are minimal, according to a study conducted by CDC epidemiologists.
The findings support the use of varicella vaccine for routine immunization and the safety of vaccination of close contacts of people at high risk for severe varicella who have contraindications for vaccination (Pediatrics 2019;144[3]:e20191305).
vOka is a live attenuated virus vaccine, used to prevent the transmission of the wild-type varicella-zoster virus. While live attenuated vaccines usually provide more robust immunity than vaccines containing inactivated viruses, they can, in certain instances, transmit vaccine virus.
“Millions of persons are vaccinated with varicella vaccine each year in the U.S.,” explained Mona Marin, MD, of the Division of Viral Diseases, CDC’s National Center for Immunization and Respiratory Diseases. “Our review added to the body of evidence of the safety of the varicella vaccine. We should continue to remain vigilant and track the characteristics of transmission of vaccine-strain varicella-zoster virus, or vOka.”
Dr. Marin and her colleagues conducted a review of the literature on live varicella and zoster vaccines published from 1981 to 2019. Twenty-six articles met the inclusion criteria.
The team found that transmission of vOka from healthy vaccine recipients is very rare. “To date, only 13 cases from 11 vaccinated, immunocompetent persons have been reported,” Dr. Marin said. “All after receipt of the first dose of varicella vaccine and most commonly among household contacts.”
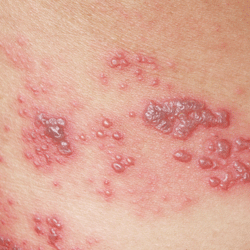
Additionally, in all instances, the vaccine recipient had a rash after vaccination.
In the 13 cases of varicella caused by the vaccine, strains were typically mild. “Healthy, varicella-vaccinated persons have minimal risk for transmitting vaccine strain to contacts, and only if a rash is present,” Dr. Marin said.
She noted that the study reinforces the use, and safety, of the varicella vaccine. “The findings support the existing recommendations for routine varicella vaccination and the guidance that persons with vaccine-related rash avoid contact for the duration of the rash with persons not immune to varicella who are at high risk for severe complications,” she said.