The CDC’s Advisory Committee on Immunization Practices (ACIP) recently changed its recommendations for respiratory syncytial virus (RSV) vaccination suggesting that everyone ages 75 years and older receive the vaccine, but those 60 to 74 should be vaccinated only if they are at increased risk for severe RSV.
This recommendation replaces the previous guidance that called for shared clinical decision making for adults who are 60 and older to receive the vaccine.
“Under the new recommendation CDC isn’t just encouraging adults to talk to their providers to determine whether they should get an RSV vaccine, but instead actively recommending that all adults 75 and older and adults 60 to 74 with risk factors get an RSV vaccine if they haven’t already had one,” explained Amadea Britton, MD, the co-lead of the ACIP Adults RSV Work Group.
The impetus for updating the guidelines was due to feedback from providers indicating that under the shared clinical decision-making recommendation it was not clear which patients should be vaccinated, and the process of discussing the vaccine with patients was confusing and complicated. “The updated recommendations are intended to maximize RSV vaccination coverage among persons most likely to benefit, by clarifying who is at highest risk and by reducing implementation barriers,” Dr. Britton explained.
Only about 24% of those older than 60 were vaccinated by May 2024, despite the vaccines being available for a little more than a year, according to the CDC. Vaccine coverage was highest in white adults (26.8%) while lower in Blacks (21%) and Hispanics (14.7%).
The vaccines Arexvy (GSK) and Abrysvo (Pfizer) were first recommended for use in June 2023. Both are made of a single surface protein from the RSV virus, called the F protein. There are two subtypes of RSV: RSV A and RSV B. Abrysvo contains the protein from both subtypes, while Arexvy contains a single F protein. “However, since the F protein in both types tends to be the same and because both vaccines have the same quantity of the F protein, this difference is not anticipated to make one vaccine better than the other,” explained Hannah Nam, MD, an assistant professor of medicine at the University of California, Irvine, who is not part of ACIP.
Another difference between Arexvy and Abrysvo is the former is an adjuvanted vaccine that contains the same adjuvant as the shingles vaccine.
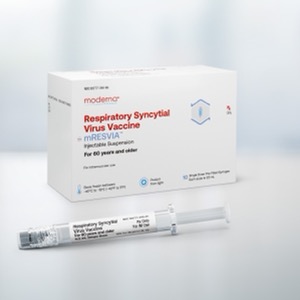
The updated guidelines include recommending the use of mRESVIA (Moderna) an mRNA-based vaccine for RSV that was approved in May 2024.
“For most nonpregnant adults over 60 years of age, the choice of the vaccine is determined by availability. There are no data to support the use of one vaccine over the other as the trials were each done with different end points and different periods of follow-up following vaccination. However, for immunocompromised patients, there may be a theoretical benefit to utilizing the adjuvanted vaccine. These patients generally have a worse immunological response to vaccination, and adjuvanted vaccines may be more immunogenic,” Dr. Nam explained.
Dr. Britton underscored that providers should be flexible in offering RSV vaccine to patients at increased risk for severe disease, even if they do not fall into an explicitly named risk category. However, Arexvy is the only vaccine approved for at-risk adults who are 50 to 59.
Although it may appear that the new recommendations imply that fewer people will get the vaccine, “these new vaccination criteria may qualify more people to receive the vaccines. Many patients do not have access to healthcare providers who can conduct meaningful shared decision making. Several patients will receive vaccines at pharmacies rather than through physician or healthcare provider visits. Therefore, the recommendation for all patients over 75 to receive the RSV vaccines would potentially increase the number of patients receiving the vaccines,” Dr. Nam concluded.
Drs. Britton and Nam reported no relevant financial disclosures.
This article is from the October 2024 print issue.