By IDSE News Staff
Sanofi said it would ship nirsevimab (Beyfortus) starting in the beginning of the third quarter to ensure broad availability well ahead of the 2025-2026 respiratory syncytial virus (RSV) season, which typically starts in November and runs through March.
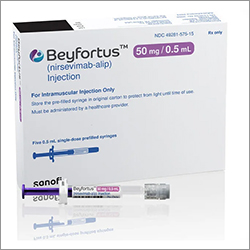
The immunotherapy should begin in early fall, and these advance shipments will support the efforts of healthcare professionals in reaching this deadline. Demand has continued to increase since the launch of nirsevimab, the only option that protects infants from RSV protection.
Sanofi, in collaboration with its partner AstraZeneca, has tripled production capacity and doubled the number of manufacturing sites since the launch of Beyfortus in 2023. Current supply for the upcoming season already matches the total doses distributed last year and production continues. Last season in the United States, enough doses were produced to cover every infant—a benchmark that reflects both Sanofi’s and AstraZeneca’s global commitment to ensuring timely access for all infants.
“Our third year of providing RSV protection marks a transformative era in infant health. Backed by more than 40 real-world studies involving 250,000 immunized infants, Beyfortus has consistently proven itself as an effective RSV immunization. To meet growing demand since launch, we have meticulously primed our global distribution network to ensure Beyfortus will continue to deliver significant public health impact and offer high, sustained efficacy for infants regardless of underlying health conditions or the timing of when they are born,” said Thomas Triomphe, the executive vice president of Vaccines at Sanofi.
This year’s European Union shipments are marked by an important change to the nirsevimab EU label extending the duration of protection through 6 months of age, so nirsevimab can continue to offer season-long protection for all infants, including older babies immunized just before the RSV season begins. More than 6 million babies have been immunized worldwide, and millions more will be immunized this season in more than 40 countries to help protect them from the potentially devastating impact of RSV disease.
RSV is a highly contagious virus infecting two out of three infants in their first year of life and nearly all children by their second birthday. RSV is the main cause of lower respiratory tract disease and a leading cause of hospitalization in all infants worldwide, with most hospitalizations occurring in healthy infants born at term.
With an extended half-life of 71 days, nirsevimab stands apart as the longest-acting monoclonal antibody for the prevention of RSV lower respiratory tract disease in infants, and the only RSV immunization designed to protect all infants born before or during the RSV season, at term or preterm, and healthy or with underlying conditions. Administration can be timed to coincide with the RSV season and provided directly to newborns and infants as a single dose. Nirsevimab offers rapid protection without requiring activation of the immune system. Nirsevimab is also designed to protect children up to 24 months of age who remain vulnerable to severe RSV disease through their second RSV season.