Saline “flush” bags used to prepare anesthetic medications could be an underappreciated source of bacterial contamination.
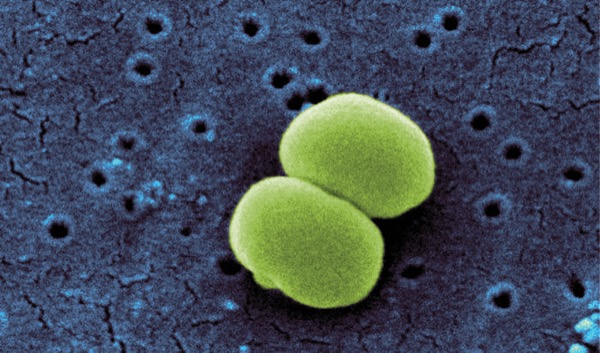
Researchers found that Staphylococcus epidermidis, a common skin microbe that can contaminate medications during preparation, may persist in saline for more than 24 hours.
“Contamination most often occurs at touchpoints, so that would mean injection ports on bags of fluids that we would prepare the medications in or at the hubs we inject into, but the reason in the first place that it happens is we’re not preparing these medications in the same environment as, say, pharmacy would be,” said Brian Woytowich, MD, FRCPC, BSc, an anesthesiologist at the University of Saskatchewan, in Saskatoon, and lead study author who presented his findings at the 2024 meeting of the Canadian Anesthesiologists' Society. “We use aseptic technique to minimize the risk for contamination, but, over the week that medications would be kept before they’re discarded and then re-prepared, this is potentially a source of pathogen exposure to patients.”
When asked to comment on the study, Jonathan Schmitz, MD, PhD, DABMM, a clinical microbiologist, was not surprised by the results. “From a basic microbiologic perspective, the fact that S. epidermidis can persist in saline is quite consistent with its biology. Staphylococcus species, in general, are known for their salt tolerance. In fact, selective media for isolating staphylococci can include elevated salt concentrations for exactly this purpose,” said Dr. Schmitz, who is also an investigator of microbial pathogenesis at Vanderbilt University Medical Center and School of Medicine, in Nashville, Tenn., and a member of the Infectious Disease Special Edition editorial advisory board.
“Overall, as this work represents a contrived in vitro study, it shouldn’t necessarily raise any concerns that saline infusions represent an overlooked cause of iatrogenic infections. But it does reinforce the fact that—when unusual healthcare-associated infections do arise—managing physicians and hospital epidemiologists have to keep an open mind about potential atypical sources.”
Another Thought
In addition to normal saline, the researchers tested the viability of S. epidermidis in two medications: norepinephrine prepared in 5% dextrose and phenylephrine prepared in saline. These were selected because they represent commonly used anesthetic vasopressors and diluents. Dr. Woytowich “thought dextrose could be a potential energy source for the bacteria” and was thus expecting S. epidermidis viability in norepinephrine.
After preparing the two medications and saline control with aseptic technique and inoculating each with S. epidermidis, aliquots were collected at 24-hour intervals for seven days. In the saline solution, viable bacteria were seen at 24 hours, but samples at 48 and 72 hours did not yield any viable bacteria. In the phenylephrine and norepinephrine solutions, viability did not persist beyond time zero, and there was no evidence of viability at 24 hours, Dr. Woytowich told IDSE.
“It’s interesting that it appears the bacteria persist longer in saline, and that might mean that maybe the phenylephrine or norepinephrine actually have some activity against the bacteria themselves,” Dr. Woytowich said.
However, another study found that phenylephrine may increase susceptibility for infection (Br J Anaesth 2021;126[3]:652-664).
Even in the event of contamination with skin flora during preparation of medications, S. epidermidis does not appear to be persist in some commonly pre-prepared medications, according to Christine Patterson, MD, FRCPC, BSc, an anesthesiologist at the University of Saskatchewan. “This suggests that there is limited risk from S. epidermidis when we pre-prepare and store these medications for up to a week, even if the medications do experience some contamination during preparation.”
Because there are limited data determining viability of bacteria through CFU over time and the antimicrobial effects of medications against virulent pathogens, the researchers hope their work inspires future research.
“This project is really meant to be a stepping stone to other projects looking into viability and CFU counts over time for various pathogens, like the more virulent pathogen Staphylococcus aureus, and various solutions,” Dr. Woytowich explained.
The sources reported no relevant financial disclosures.
This article is from the February 2025 print issue.