Originally published by our sister publication Pharmacy Practice News
By Karen Blum
Since cefotaxime—the preferred cephalosporin in neonates—was discontinued in 2018, many hospital neonatal ICUs (NICUs) have switched to ceftazidime or cefepime. But a recent study of more than 50,000 neonates indicates that these alternatives could lead to increased adverse events, with a significantly higher proportion of babies developing sepsis, necrotizing enterocolitis (NEC) or dying, compared with those given cefotaxime.
The study “added to some of my worrisome hypotheses” about this therapeutic switch, said lead study author Deborah Bondi, PharmD, FCCP, FPPA, BCPS, BCPPS, a clinical pharmacy specialist with the NICU at the University of Chicago Comer Children’s Hospital.
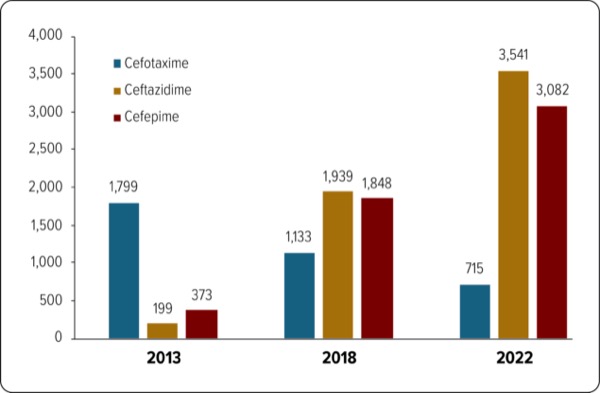
In a retrospective study presented at 2024 PediatRxCon33, sponsored by the Pediatric Pharmacy Association, Dr. Bondi and colleagues pulled data from the Vizient Clinical Data Base from more than 1,100 hospitals, including 97% of academic medical centers, from January 2013 to June 2023. They reviewed records from 50,618 neonates admitted to a NICU who received cefotaxime, ceftazidime or cefepime in the first two weeks of life for at least 24 hours, with 16,189 receiving cefotaxime, 17,398 ceftazidime and 17,031 cefepime. The babies were 28.3 to 29.4 weeks’ gestational age with an average birth weight of 1,749 to 2,280 g. The investigators also performed a subgroup analysis in those with very low birth weight (VLBW), averaging 800 to 870 g. There were no statistically significant differences in baseline demographics including gestational age, birth weight or duration of study antibiotic, according to Dr. Bondi.
Several outcomes were worse for the babies receiving ceftazidime and cefepime, even after adjusting for baseline demographics, she said. For example, 2,621 babies (15.1% of the total) given ceftazidime and 2,288 (13.4%) given cefepime developed culture-positive sepsis, compared with 808 (5%) of those who received cefotaxime. Some 6,113 babies (35%) given ceftazidime and 5,081 (29.8%) given cefepime developed culture-negative sepsis, compared with 2,492 (15.4%) of those who received cefotaxime.
Of note, 1,506 neonates (8.7%) given ceftazidime and 2,049 (12%) given cefepime had stage 2 to 3 NEC compared with 675 (6%) in the cefotaxime group. About 1,594 infants (9.2%) given ceftazidime and 2,093 (12.3%) given cefepime developed urinary tract infections compared with 1,387 (8.5%) who received cefotaxime. Death rates and hospital length of stay (LOS) also were lower in the cefotaxime group, with 1,722 babies (9.9%) given ceftazidime and 2,015 (11.8%) given cefepime dying compared with 1,424 (8.8%) of those who received cefotaxime. Number of days hospitalized averaged 47.5 among the ceftazidime group and 60.1 in the cefepime group compared with 41.4 in the cefotaxime group.
Similar results were seen in the VLBW neonates, although death rates were only numerically higher for ceftazidime, Dr. Bondi said.
The study suggests that the broader gram-negative empiric coverage from ceftazidime or cefepime “could alter the gut microbiome, which may predispose the child to later NEC or culture-positive infections,” she said. “NEC is associated with a higher rate of mortality, so these are the kind of concerns that we have with using these antibiotics from the get-go.”
There was a U.S. shortage of cefotaxime in 2015, and the drug was discontinued in February 2018, Dr. Bondi said. Her hospital and others, per FDA allowance, continue to use the agent by importing it from Canada, but other NICUs have switched to ceftazidime or cefepime. Guidelines from the American Academy of Pediatrics list ceftazidime as a “reasonable option,” Dr. Bondi said.
Some of the concerns posted in online discussion groups about importing cefotaxime relate to expense or periodic shortages, rather than side effects, added study co-author Gretchen Brummel, PharmD, BCPS, who worked as a pharmacy executive director for Vizient’s Center for Pharmacy Practice Excellence when the study was conducted.
A previous study by Dr. Bondi and colleagues (J Pediatr Pharmacol Ther 2020;25[2]:117-123) comparing use of ceftazidime and cefotaxime in neonates at the University of Chicago found similar results. That study prompted Dr. Brummel to explore whether the same issues exist across medical centers, and to determine whether alternative antibiotics are good substitutes. “We know that with a retrospective database, we cannot determine causality,” Dr. Brummel said. “But the association between those factors is certainly significant, and I think that warrants further analysis and attention.”
The new results “strengthened the concerns that I have about the challenges our providers face in accessing this drug,” Dr. Brummel said. “It’s a vulnerable population that we’re working with when we talk about NICU patients, so it’s really important for [clinicians] to have every available tool in their toolbox when they’re caring for these patients.” She also said she hoped that the data will help encourage a U.S. manufacturer to step forward and resume production of cefotaxime.
Notable Differences Among Infants
Although the study data “are certainly concerning,” the baseline characteristics of the neonates in the three groups receiving the antibiotics “suggests there are some major underlying differences,” which could affect the outcomes, said James Lewis, PharmD, FIDSA, a clinical supervisor for infectious disease, Department of Pharmacy Services, at Oregon Health & Science University, in Portland.
Babies in the cefotaxime group “are clearly bigger, which, when you’re talking about preterm neonates, is a big deal,” explained Dr. Lewis, who was not involved with the study. The babies receiving cefotaxime had an average birth weight of 2,280 g compared with 2,080 g in the ceftazidime group and 1,749 g in the cefepime group. There also was considerably more NEC in the ceftazidime and cefepime groups.
Moreover, the days of study antibiotics varied. For example, days of study antibiotics in the first course averaged four for the cefotaxime group, six for the ceftazidime group and eight for the cefepime group. “What this says to me is that you’ve got sicker kids going into the ceftazidime and cefepime arms,” Dr. Lewis said. “When you put all of this together, I think that’s what’s driving [the divergent results].”
The hospital LOS in the infants given cefepime was also longer, demonstrating another important variable in the groups.
“While I do think it’s concerning and potentially hypothesis-generating, … I am not sure that what you’re seeing here is a switch in antibiotics driving some of these [results],” he said. “The types of spectrum differences that we’re talking about among these three drugs are not massive, and I would be hard-pressed to believe that [variations] in microbiome activity would be driving this.”
Dr. Bondi told Pharmacy Practice News that the group calculated the adjusted odds ratios and adjusted P values for all baseline demographics to account for the differences and still saw statistically significant outcomes in both analyses.
The Clinical Value of Addressing Shortage
As for the issue of cefotaxime supplies, “it was unusual from my perspective for a drug that still has clinical utility to be discontinued,” Dr. Bondi said, noting that it may not have been financially viable for the manufacturers to continue producing the antibiotic. However, “I would love to see advocacy” surrounding cefotaxime’s potential for significantly improving clinical outcomes in neonates, she noted. Such efforts “[could help] get it back into the U.S. as an FDA-approved product that’s commercially marketed.”
Erin Fox, PharmD, MHA, BCPS, FASHP, the associate chief pharmacy officer for shared services at the University of Utah, in Salt Lake City, said that ceftazidime and cefepime are the closest substitutes to cefotaxime. But she said she had heard an unconfimed rumor that the cost of cefotaxime raw materials became too high for companies to profitably produce the antibiotic. Regardless, “when a product is gone for so long, it’s highly unlikely that it will come back,” said Dr. Fox, who was not involved with the study.
The sources reported no relevant financial disclosures beyond their stated employment.