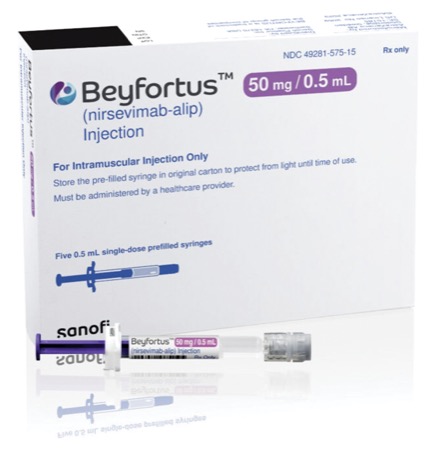
Early real-world data show that a single injection of the monoclonal antibody (mAb) nirsevimab (Beyfortus, Sanofi/AstraZeneca) was 83% effective in preventing babies from being hospitalized with lower respiratory tract disease caused by respiratory syncytial virus (RSV), according to French researchers (N Engl J Med 2024;391[2]:144-154).
Protection for a Vulnerable Population
RSV often causes severe illness, particularly in infants and young children. The previous RSV prophylaxis for infants required monthly doses of palivizumab (Synagis, Swedish Orphan Biovitrum AB) for the entire season to protect against RSV bronchiolitis.
“Three randomized trials showed that a single injection of the monoclonal antibody nirsevimab could prevent RSV lower respiratory tract infection in young children, leading to FDA and EMA [European Medicines Agency] approval,” Naïm Ouldali, MD, PhD, who was involved in the study, told Infectious Disease Special Edition.
The French study included 1,035 infants, of whom 690 were case patients (median age, 3.1 months; interquartile range [IQR], 1.8-5.3 months) and 345 were matched control patients (median age, 3.4 months; IQR, 1.6-5.6 months).
In this prospective case-control study, infants younger than 12 months were admitted to six French hospitals with RSV bronchiolitis (cases) and compared with infants who presented with unrelated complaints (controls). Sixty of the patients with RSV (8.7%) and 97 control patients (28.1%) had received nirsevimab as prophylaxis. The estimated adjusted effectiveness of nirsevimab therapy against hospitalization for RSV-associated bronchiolitis was 83.0% (95% CI, 73.4%-89.2%).
The researchers determined nirsevimab was 69.6% effective in preventing RSV that resulted in ICU care (95% CI, 42.9%-83.8%) and 67.2% effective in preventing RSV-associated bronchiolitis that required ventilator support (95% CI, 38.6%-82.5%), the researchers added.
Of note, mAbs such as nirsevimab are laboratory-made proteins that mimic the immune system’s ability to fight off harmful pathogens. Nirsevimab targets the RSV fusion (F) protein, a key component the virus uses to enter human cells. By binding to this protein, nirsevimab prevents the virus from infecting cells, reducing the severity of the infection (MMWR Morb Mortal Wkly Rep 2023;72[34]:920-925).
Nirsevimab is specifically designed to provide passive immunity against RSV, making it a valuable tool in reducing the burden of RSV-related hospitalizations and severe illnesses, according to the CDC.
Unlike vaccines that stimulate the body’s immune system to produce antibodies, nirsevimab directly supplies the body with antibodies that can neutralize the virus, according to the CDC. This approach is particularly beneficial for infants and young children who are at high risk for severe RSV infection (www.cdc.gov/vaccines/ vpd/ rsv/ hcp/ child-faqs.html).
“Respiratory syncytial virus is the leading cause of hospitalization for babies in the United States,” said Jefferson Jones, MD, MPH, the Maternal-Pediatrics Work Group co-lead for the Advisory Committee on Immunization Practices.
Although other mAb injections are available, they require more frequent injections because palivizumab has a short half-life and limited efficacy.
“Palivizumab requires repeated injections every month and was restricted to populations at highest risk for severe RSV bronchiolitis,” Dr. Ouldali said. Taking advantage of a longer half-life and stronger efficacy, nirsevimab can now be recommended to all children younger than 12 months of age, regardless of their risk factors for RSV disease, he noted.
Nirsevimab was implemented in the general pediatric population in just a few countries in winter 2023-2024, and real-world data to assess its effectiveness were missing, Dr. Ouldali explained.
“By showing the huge effect of nirsevimab in preventing severe forms of RSV bronchiolitis, this study may help policymakers estimate the potential benefit of implementing this product in their national immunization programs to reduce the burden associated with this pathogen in children,” Dr. Ouldali said.
Robert H. Hopkins Jr., MD, the medical director of the National Foundation for Infectious Diseases, who was not involved in the study, echoed Dr. Ouldali’s sentiment that the injection offers a new layer of protection for infants.
“Nirsevimab is a long-acting monoclonal antibody that, when given to infants in the study and the clinical trials that achieved FDA approval, is effective at preventing severe RSV-associated bronchiolitis resulting in hospitalization and the need for respiratory support,” said Dr. Hopkins, who is also a professor of internal medicine and pediatrics at the University of Arkansas for Medical Sciences (UAMS) and the director of the UAMS Division of General Internal Medicine.
In addition to one injection of nirsevimab being associated with strong protection against severe RSV bronchiolitis during an epidemic season, this important effectiveness was also found in children with risk factors for severe RSV bronchiolitis, such as prematurity or chronic lung or cardiac diseases, Dr. Oulaldi said.
U.S. Nirsevimab Use
According to the CDC, nirsevimab is recommended for infants who were born to mothers who did not receive the RSV vaccine during pregnancy.
“For infants born during October through March, nirsevimab should be administered in the first week of life—ideally during the birth hospitalization. For infants born before October and eligible children 8 through 19 months at increased risk for severe disease, nirsevimab should ideally be administered just before or at the start of the RSV season,” she said. The CDC recommends RSV immunizations to protect all babies from severe RSV.
The sources reported no relevant financial disclosures.
This article is from the October 2024 print issue.