By Ethan Covey
Cell-based and adjuvanted influenza vaccines can reduce the burden of seasonal influenza on hospitals and health systems, according to data presented at the 11th congress in the OPTIONS series, held in Belfast, Northern Ireland.
In the first study, a retrospective cohort design (poster P-328), researchers used electronic health records (EHRs) to estimate the relative effectiveness of adjuvanted trivalent vaccines (aTIV) (FLUAD, CSL Seqirus) compared with high-dose trivalent vaccine (HD-TIV) and standard-dose egg-based vaccine (QIVe) in preventing cardiorespiratory-related hospitalization among adults 65 years of age and older during the 2019-2020 influenza season.
Because influenza can be associated with adverse cardiorespiratory events, the outcomes evaluated were cardiorespiratory-related hospitalizations and respiratory-related hospitalizations, specifically influenza-related and pneumonia-related hospitalizations, as well as hospitalizations related to myocardial infarction and ischemic stroke.
Among vulnerable populations, such as adults at least 65 years old, influenza vaccine effectiveness is a challenge because of age-related immune decline, which makes it more difficult for the body to protect against the influenza virus. This immunosenescence may result in a reduced immune response to antigens in the vaccine.
“Adjuvant vaccines may provide a benefit to combating immunosenescence,” Gregg Sylvester, MD, the chief health officer of CSL Seqirus, in Summit, N.J., told Infectious Disease Special Edition. “An adjuvant is a component used in vaccines to help enhance the body’s immune response for effective protection against influenza.”
A total of 4,299,594 individuals met the selection criteria for the study, of whom 25.2% received FLUAD, 56.9% received an HD-TIV and 17.9% received a QIVe.
The researchers found that immunization with FLUAD was more effective than the QIVe and the HD-TIV in preventing cardiorespiratory-, respiratory- and influenza-related hospitalizations. Vaccine effectiveness was consistently higher for FLUAD versus the QIVe, rather than FLUAD versus the HD-TIV.
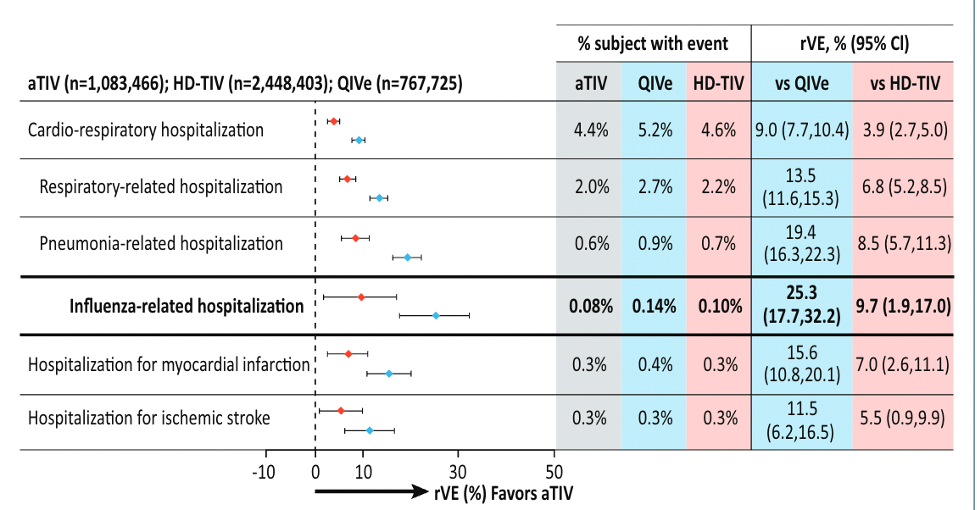
They concluded that FLUAD offered improved protection against severe influenza disease in older adults.
The second study aimed to estimate the relative vaccine effectiveness of a mammalian cell-based quadrivalent influenza vaccine (QIVc), compared with the QIVe, in preventing cardiorespiratory-related hospitalization among U.S. adults 18 to 64 years of age during the 2019-2020 influenza season (poster P-329).
The EHRs of these adults with a record of influenza vaccination with the QIVc or the QIVe were linked to claims data for the 2019-2020 influenza season. Of a total of 5,905,855 patients, 25.2% received the QIVc and 74.8% received the QIVe.
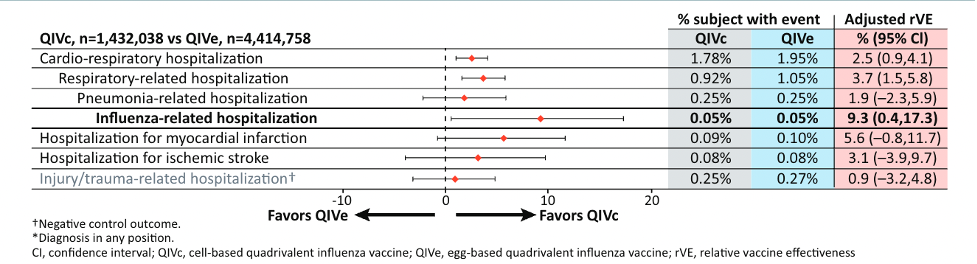
The results showed that the QIVc was more effective than the QIVe in preventing cardiorespiratory-, respiratory- and influenza-related hospitalizations. No difference was observed between the two types of vaccines in hospitalizations related to pneumonia, myocardial infarction or ischemic stroke.
“The ever-changing nature of influenza poses unique challenges to the development of seasonal influenza vaccines, impacting vaccine effectiveness from season to season,” Dr. Sylvester said. “These studies demonstrate the benefits of [FLUAD] and QIVc compared to QIVe and HD-TIV.”
The data from these two studies showcased that cell-based and adjuvanted vaccine technologies are designed to address influenza prevention challenges and can reduce the burden of seasonal influenza on hospitals and health systems, Dr. Sylvester added. “As new technology drives vaccine innovation, it improves our ability to develop more precise, effective vaccines to address seasonal influenza.”
Dr. Sylvester is employed by CSL Seqirus.