Since the publication of the REPRIEVE trial last summer in The New England Journal of Medicine,1 there has been renewed interest and enthusiasm for cardiovascular disease (CVD) prevention for people living with HIV (PLWH). Regardless of how you feel about the benefits of statins, we can all agree that helping our patients live long and healthy lives requires us to look beyond the HIV care cascade to consider the prevention of non-AIDS comorbidities, including CVD.2
Taking a whole health approach requires us to continually reexamine how we provide care in our HIV clinics and consider how we can do better to address the top 3 risk factors for heart disease in PLWH: smoking, hypertension, and high cholesterol.3 It is encouraging to see smoking rates among PLWH decline in many places. Where smoking is still common, smoking cessation programs are needed urgently, but advice about smoking cessation for PLWH is beyond the scope of this review. Instead, this review focuses on new (and old) strategies to address hypertension and cholesterol. Statins are certainly our mainstay of CVD prevention, but we now have a range of new evidence-based options to treat residual lipid, metabolic, and inflammatory risk. If properly implemented in HIV clinics, we might achieve health outcomes for our patients that rival or even surpass those of the general population.
Opportunity to Provide High-Quality Primary Care
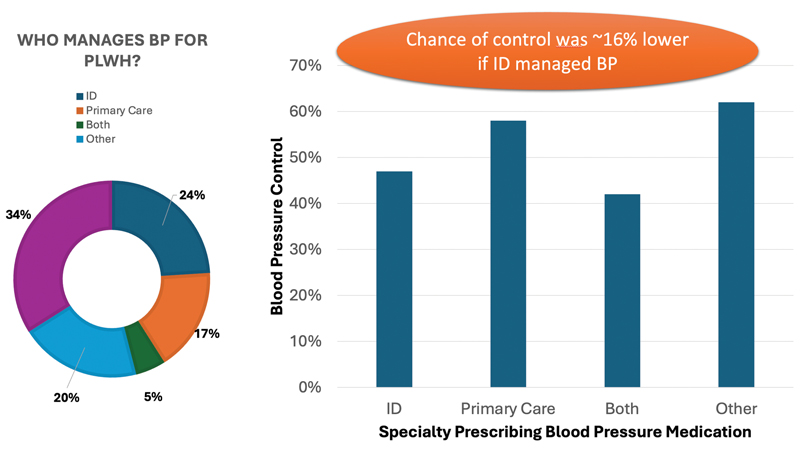
HIV care in the United States presents several barriers but also many opportunities for high-quality CVD prevention care. In some parts of the country, HIV providers—many of whom are trained as infectious disease specialists—prefer to focus on HIV and ID care, while leaving the rest to a primary care provider (PCP). Elsewhere, HIV clinicians practice a model of comprehensive primary care for their patients. The relative benefits of each approach can be debated, but in either model, it is important for someone to take responsibility for CVD prevention care. Does it matter who? In an interesting study examining this question,4 Okeke and colleagues suggest that if an ID specialist is managing your blood pressure (BP), your chances of control are lower than if a PCP is doing it (Figure). Although I know many amazing HIV providers who do a great job with hypertension, it is a reminder that life is not just about antiretroviral drug resistance and syphilis.
Even if intentions are good, we need care structures in place to help us provide high-quality care. In the United States, the Ryan White Comprehensive AIDS Resources Emergency Act provides additional tools that could be used to augment CVD prevention care.5 One effective model may be to empower nurses to provide medical case management alongside social workers. In the EXTRA-CVD trial, we demonstrated that a nurse-led approach with home BP monitoring overcame clinical inertia to lower systolic BP by more than 4 mm Hg and cholesterol by almost 17 mg/dL.6 Implementation of this model is possible in clinics where nurses are resourced adequately to play a prominent role in care.
Another cornerstone of CVD prevention is estimating and tailoring risk reduction to those who are most likely to benefit. Most HIV clinicians are familiar with the 2013 pooled cohort equations that are recommended by guidelines to estimate 10-year atherosclerotic CVD (ASCVD) risk.7 Most also know that PLWH are at higher risk not only for ASCVD but also heart failure (HF), arrhythmias, and sudden cardiac death.8 Newer risk equations called PREVENT have been introduced by the American Heart Association, and they now incorporate risk for HF and ASCVD.9 The new equations also extend the age range for 10-year risk down to 30 years. (The pooled cohort equations range is 40-79 years.) We will see implementation of the new PREVENT equations in primary and HIV care over the next couple of years.
Hypertension Care Is Not Rocket Science
Preventing ASCVD and HF begins with BP control. Sometimes in the face of competing priorities in the clinic, BP doesn’t make it to the top of the list. How often have you seen someone in the clinic with a BP of 143 mm Hg systolic and thought: “It’s not that high. We can address that at the next visit.” Clinical inertia is real, but hypertension management is truly not rocket science. We have decades of evidence supporting algorithmic guidelines-based BP management,10 typically starting with a thiazide diuretic or dihydropyridine calcium channel blocker and then adding an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker. The SPRINT trial provided evidence that treating to a target systolic BP of less than 130 mm Hg reduced the risk for major adverse cardiovascular events, particularly HF.11 If you can’t get there with the first-line therapies above, adding a mineralocorticoid receptor antagonist (MRA) such as spironolactone is typically effective. Also, in PLWH there is emerging evidence that MRAs may reduce inflammation12 and myocardial perfusion,13 which is likely to improve both ASCVD and HF outcomes long term.
New Enthusiasm for Statins
Along with hypertension, cholesterol is the other modifiable risk factor that is critical to address, especially with all the awareness of the REPRIEVE trial results. Statins have been around for decades, and there is solid evidence for their use in primary prevention. Because absolute benefit depends on absolute risk, the guidelines have always advocated for a risk-based approach.7 The 2018 joint cholesterol guidelines recommended considering a statin for all PLWH with less than 5% 10-year risk for ASCVD.
The REPRIEVE trial went further to test the effectiveness of pitavastatin calcium (a moderate-intensity statin that does not have any drug interactions with antiretroviral therapy [ART]) to reduce major adverse cardiovascular events in lower risk individuals, including PLWH with less than a 5% 10-year risk.1 The trial was stopped early for clear evidence of benefit—a 35% reduction in events, which was somewhat higher than would be expected for the modest amount of low-density lipoprotein (LDL) lowering seen in the trial. Although most of the absolute benefit in REPRIEVE was seen in participants with less than 5% risk, new guidelines by the Department of Health and Human Services about implementing the REPRIEVE trial findings in clinical practice now recommend considering statins in low-risk patients aged 40 to 75 as well, especially those with other risk enhancers such as a low CD4+ count or long duration of HIV. Of note, REPRIEVE confirmed the excellent safety profile of statins, with low rates of statin-associated muscle toxicity and incident diabetes comparable with other statin trials in the general population. Regardless of patient risk, the decision to initiate a statin is always a shared decision between patients and their providers. It is hoped, in the post-REPRIEVE era, HIV providers who strive to provide high-quality comprehensive primary care will be better equipped to have that discussion.
New Cholesterol Drugs to Address Residual Lipid Risk
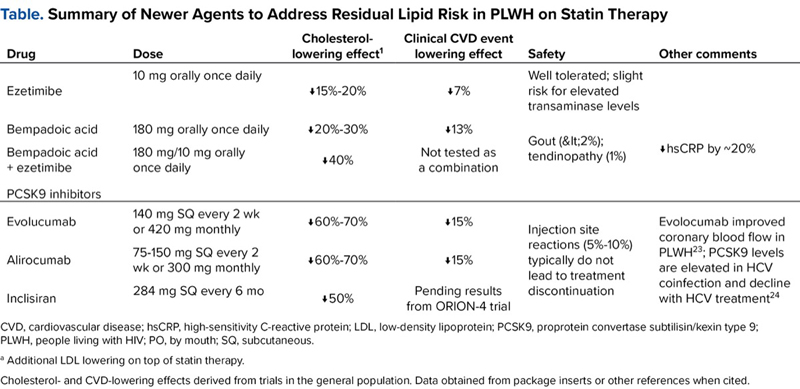
Once someone is on a statin, it is important to monitor the LDL response. My target for a PLWH without known ASCVD in my clinic is generally an LDL less than 100 mg/dL or non-HDL (high-density lipoprotein) cholesterol less than 130 mg/dL (simply total cholesterol minus HDL). Conveniently, that “130” number is also the same as the target systolic BP for patients with hypertension. For patients with established ASCVD, including those with prior myocardial infarction or stroke and subclinical atherosclerosis on a CT scan, I aim for an LDL less than 50 mg/dL in line with European guidelines14 and the standard practice of many preventive cardiologists in the United States. For patients who can’t get to goal on a statin alone, we have many powerful new agents in the arsenal with randomized clinical trial evidence of benefit in a general population (Table). We will likely never have a randomized trial of these drugs in PLWH, but we don’t need them. Just as REPRIEVE showed us statins also work in PLWH, these drugs will as well.
| Table. Summary of Newer Agents to Address Residual Lipid Risk in PLWH on Statin Therapy | |||||
| Drug | Dose | Cholesterol-lowering effect1 | Clinical CVD event lowering effect | Safety | Other comments |
|---|---|---|---|---|---|
| Ezetimibe | 10 mg orally once daily | ?15%-20% | ?7% | Well tolerated; slight risk for elevated transaminase levels | |
| Bempadoic acid | 180 mg orally once daily | ?20%-30% | ?13% | Gout (<2%); tendinopathy (1%) | ?hsCRP by ~20% |
| Bempadoic acid + ezetimibe | 180 mg/10 mg orally once daily | ?40% | Not tested as a combination | ||
| PCSK9 inhibitors | |||||
| Evolucumab | 140 mg SQ every 2 wk or 420 mg monthly | ?60%-70% | ?15% | Injection site reactions (5%-10%) typically do not lead to treatment discontinuation | Evolocumab improved coronary blood flow in PLWH23; PCSK9 levels are elevated in HCV coinfection and decline with HCV treatment24 |
| Alirocumab | 75-150 mg SQ every 2 wk or 300 mg monthly | ?60%-70% | ?15% | ||
| Inclisiran | 284 mg SQ every 6 mo | ?50% | Pending results from ORION-4 trial | ||
| CVD, cardiovascular disease; hsCRP, high-sensitivity C-reactive protein; LDL, low-density lipoprotein; PCSK9, proprotein convertase subtilisin/kexin type 9; PLWH, people living with HIV; PO, by mouth; SQ, subcutaneous. a Additional LDL lowering on top of statin therapy. Cholesterol- and CVD-lowering effects derived from trials in the general population. Data obtained from package inserts or other references when cited. | |||||
Ezetimibe lowers LDL by about 15% to 20% and provides a small additional hard clinical CVD event reduction (~6%-7% resistive reserve ratio [RRR]) for patients on statin therapy.15 It is recommended as a cost-effective strategy for secondary prevention in the joint cholesterol guidelines.7 Bempedoic acid is a newer drug that can be given alone (Nexletol, Esperion) or with ezetimibe (Nexlizet, Esperion) on top of the patient's highest tolerated statin. Because it is a prodrug that only becomes active when taken up by the liver, it is not associated with muscle side effects. In the CLEAR Outcomes trial, bempedoic acid reduced clinical CVD events by 15% compared with placebo.16 Of interest to PLWH, it also has consistently lowered inflammation as measured by high-sensitivity C-reactive protein, including a reduction of more than 20% in CLEAR Outcomes. Inhibiting proprotein convertase subtilisin/kexin type 9 can provide up to 70% additional LDL lowering and 15% RRR in CVD events, but these drugs are more expensive and less cost-effective.17 The monoclonal antibody drugs alirocumab (Praluent, Regeneron) and evolocumab (Repatha, Amgen) have been on the market for several years, and patients give themselves injections every 2 to 4 weeks. The newer small interfering RNA drug inclisiran (Leqvio, Novartis) is given in the clinic with an injection every 6 months. HIV clinics that are implementing long-acting ART might easily add inclisiran to their menu of long-acting therapeutics.
New Drugs to Address Residual Metabolic Risk
In the current era of ART with widespread use of integrase inhibitors and tenofovir alafenamide (TAF), there has been a lot of consternation about weight gain and metabolic complications, particularly among women of African descent. Whether that’s due to the new drugs themselves or more a function of the weight-suppressive effects of prior regimens (eg, efavirenz and tenofovir disoproxil fumarate) is not the subject of this article. But regardless of the cause, we have new options to address weight and CVD risk together. First, it is paramount that we stress the cardiovascular and weight management benefits of a healthy diet and exercise, particularly for PLWH.18 Recognizing that not all patients will achieve their desired weight-loss goals, the glucagon-like peptide-1 receptor agonists (GLP-1 RAs) and other incretin-based therapies clearly are effective at lowering hemoglobin A1c and reducing weight and are rapidly accumulating evidence for CVD prevention.19 Even for patients with obesity but without diabetes, semaglutide reduced cardiovascular events by 20% in the SELECT trial.20 In addition, the sodium glucose transporter 2 (SGLT2) inhibitors now have almost 10 years of clinical trial data demonstrating cardiovascular benefit for people with diabetes since the initial EMPA-REG OUTCOME trial was published in 2015.21 They have become mainstays of guideline-directed medical therapy for HF with reduced and preserved ejection fraction.22 GLP-1 RAs and SGLT2 inhibitors already are indicated for PLWH with diabetes, obesity, and clinical HF; however, there may be selected high-risk PLWH without these conditions for which the drugs may be indicated to prevent progression of metabolic disease and prevent incident CVD events. For example, semaglutide improved fatty liver disease in the SLIM LIVER study (ACTG A5371; ClinicalTrials.gov Identifier: NCT04216589), a marker of visceral adiposopathy, which has become a vexing problem for some PLWH.
Future of CVD Prevention for PLWH
Regarding CVD risk in PLWH, we’ve done the epidemiology, and we have the clinical trial evidence. What we need now is to implement what we know works. For our PLWH patients engaged in HIV care, we have a unique opportunity to provide high-quality CVD prevention care if we design our clinic models with CVD and other comorbidities in mind. Many people without a chronic disease fall between the cracks in our US healthcare system, with many people not seeking care in midlife when CVD prevention is so important. But because PLWH are engaged in care, we have an opportunity to address risk factors before they lead to clinical CVD. We just need the vision and motivation to achieve it.
References
- Grinspoon SK, Fitch KV, Zanni MV, et al. Pitavastatin to prevent cardiovascular disease in HIV infection. N Engl J Med. Published online July 23, 2023. doi:10.1056/NEJMoa2304146
- Longenecker CT, Webel AR, Lloyd-Jones DM. Prevention as treatment: a bold vision for improving the cardiovascular health of people living with HIV. Prog Cardiovasc Dis. 2020;63(2):77-78.
- Althoff KN, Gebo KA, Moore RD, et al. Contributions of traditional and HIV-related risk factors on non-AIDS-defining cancer, myocardial infarction, and end-stage liver and renal diseases in adults with HIV in the USA and Canada: a collaboration of cohort studies. Lancet HIV. 2019;6(2):e93-e104.
- Okeke NL, Schafer KR, Meissner EG, et al. Cardiovascular disease risk management in persons with HIV: does clinician specialty matter? Open Forum Infect Dis. 2020;7(9):ofaa361.
- Webel AR, Schexnayder J, Rentrope CR, et al. The influence of healthcare financing on cardiovascular disease prevention in people living with HIV. BMC Public Health. 2020;20(1):1768.
- Longenecker CT, Jones KA, Hileman CO, et al. Nurse-led strategy to improve blood pressure and cholesterol level among people with HIV: a randomized clinical trial. JAMA Netw Open. 2024;7(3):e2356445.
- Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139(25):e1082-e1143.
- Feinstein MJ, Hsue PY, Benjamin LA, et al. Characteristics, Prevention, and Management of Cardiovascular Disease in People Living With HIV: a Scientific Statement From the American Heart Association. Circulation. 2019;140(2):e98-e124.
- Khan SS, Coresh J, Pencina MJ, et al. Novel Prediction Equations for Absolute Risk Assessment of Total Cardiovascular Disease Incorporating Cardiovascular-Kidney-Metabolic Health: A Scientific Statement From the American Heart Association. Circulation. 2023;148(24):1982-2004.
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71(6):e13-e115.
- Group SR, Wright JT Jr, Williamson JD, et al. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373(22):2103-2116.
- Srinivasa S, Abohashem S, Walpert AR, et al. Mineralocorticoid receptor antagonism by eplerenone and arterial inflammation in HIV: the MIRABELLA HIV study. JAMA Cardiol. 2024;9(2):189-194.
- Srinivasa S, Walpert AR, Thomas TS, et al. Randomized placebo-controlled trial to evaluate effects of eplerenone on myocardial perfusion and function among persons with human immunodeficiency virus (HIV)-results from the MIRACLE HIV study. Clin Infect Dis. 2023;77(8):1166-1175.
- Authors/Task Force Members; ESC Committee for Practice Guidelines (CPG); ESC National Cardiac Societies. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Atherosclerosis. 2019;290:140-205.
- Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372(25):2387-2397.
- Abrahams T, Nelson AJ, Nicholls SJ. How will our practice change after the CLEAR outcomes trial? Curr Atheroscler Rep. 2024;26(3):83-89.
- Cao Zhang AM, Ziogos E, Harb T, et al. Emerging clinical role of proprotein convertase subtilisin/kexin type 9 inhibition-part two: current and emerging concepts in the clinical use of PCSK9 inhibition. Eur J Clin Invest. Published online June 26, 2024. doi:10.1111/eci.14272
- Jones R, Robinson AT, Beach LB, et al. Exercise to prevent accelerated vascular aging in people living with HIV. Circ Res. 2024;134(11):1607-1635.
- Gami A, Blumenthal RS, McGuire DK, et al. New perspectives in management of cardiovascular risk among people with diabetes. J Am Heart Assoc. 2024;13(12):e034053.
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med. 2023;389(24):2221-2232.
- Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117-2128.
- Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022;79(17):1757-1780.
- Leucker TM, Gerstenblith G, Schar M, et al. Evolocumab, a PCSK9-monoclonal antibody, rapidly reverses coronary artery endothelial dysfunction in people living with HIV and people with dyslipidemia. J Am Heart Assoc. 2020;9(14):e016263.
- Gandhi MM, Nguyen KL, Lake JE, et al. Proprotein convertase subtisilin/kexin 9 levels decline with hepatitis C virus therapy in people with HIV/hepatitis C virus and correlate with inflammation. AIDS. 2024;38(3):317-327.
Copyright © 2024 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() New Strategies to Extend the HIV Treatment Cascade for CVD Prevention
New Strategies to Extend the HIV Treatment Cascade for CVD Prevention