In hospitals in the United States, Candida species are one of the most common causes of bloodstream infections (BSIs), with an associated all-cause in-hospital mortality rate of 25% to 40%.1 Among the evolving trends in candidiasis, one of the most significant is the shift from Candida albicans to more drug-resistant, non-albicans species.2 This change has been driven, in part, by the widespread use of fluconazole for prophylaxis in people living with HIV/AIDS or patients undergoing chemotherapy or a transplant. As a result, infections caused by C. glabrata and C. krusei—species that are either less susceptible or intrinsically resistant to fluconazole—have become more prevalent. Over the past 10 to 15 years, the increased use of echinocandins has been paralleled by the emergence of echinocandin-resistant Candida species. According to the CDC, 7% of Candida bloodstream isolates exhibit resistance to fluconazole, with C. glabrata and C. krusei accounting for more than 70% of these resistant strains.1 Resistance to echinocandins in C. glabrata has steadily increased, with 1 US center reporting a rise from 4.9% to 12.3% between 2001 and 2010, and resistance to fluconazole growing from 18% to 30% in the same period.3
The emergence of C. auris, a multidrug-resistant species, has further complicated the treatment landscape. C. auris is resistant to all major classes of antifungal agents in certain healthcare settings, posing significant treatment challenges. Alarmingly, in 2023, the United States reported 4,514 new clinical cases of C. auris, representing a staggering 200% increase compared with 2 years earlier.4
In addition to Candida species, antifungal resistance is affecting other pathogens, such as Aspergillus fumigatus. The first azole-resistant isolates of A. fumigatus were identified in the United States in 2015, and there has been a steady increase in incidence since then.5 Over the past 2 decades, the use of triazole fungicides in agriculture has significantly contributed to resistance in environmental molds that infect humans. Resistance mutations, including TR34/L98H and TR46/Y121F/T289A, likely originate in fungicide-exposed environments and have been found in patients with no prior azole therapy, highlighting the environmental contribution to resistance.6 In a large US study, antimicrobial resistance occurred in up to 7% of Aspergillus specimens from patients with stem cell and organ transplants.5 Infections caused by azole-resistant A. fumigatus are difficult to treat, and these patients are up to 33% more likely to die than those with infections treatable with azoles.5
The rise of antifungal resistance not only affects patient outcomes but also places a significant financial burden on healthcare systems. A US study conducted in the early 2000s estimated the median hospital cost of a single Candida bloodstream infection at approximately $46,000.7 When the infecting strain is resistant, the need for second-line drugs, such as liposomal amphotericin (which can cost thousands of dollars per day), or combination therapy can drive this figure even higher. Each case of C. auris also carries a substantial economic impact, especially with respect to infection control strategies. A hospital outbreak in London, England, was estimated to have cost more than $1.3 million in additional infection control and healthcare expenses, with ongoing costs of approximately $75,000 per month in the following year.8
In this article, we discuss resistance mechanisms of various infectious fungi, the role of rapid diagnostics, and new treatments on the horizon.
Mechanisms of Resistance
Azole Resistance
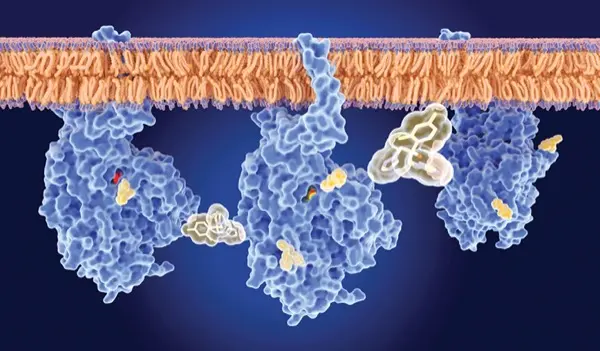
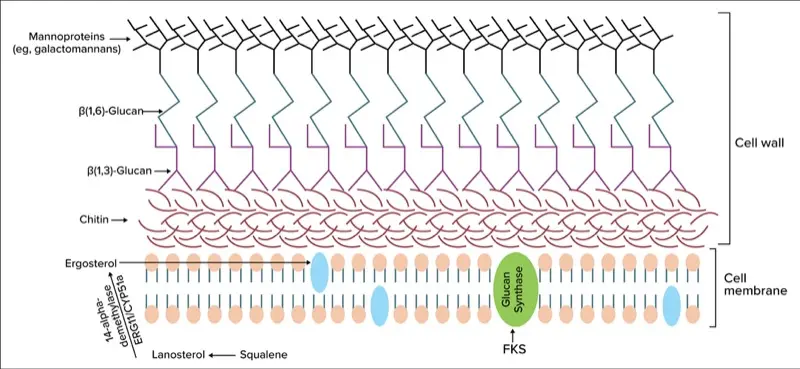
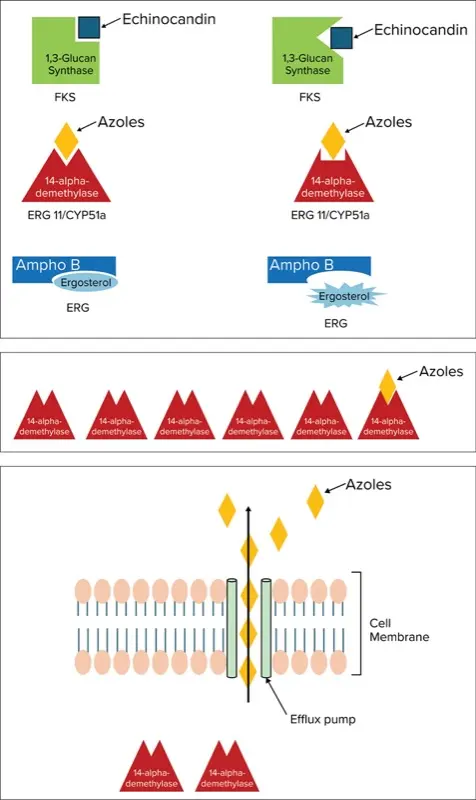
Action in Yeasts: Azole antifungals (eg, fluconazole, voriconazole) target the ergosterol biosynthesis enzyme lanosterol 14-alpha–demethylase, encoded by the ERG11 gene in yeasts (Figure 1). In Candida species, azole resistance often arises from mutations in the ERG11 gene that reduce the drug’s binding affinity (Figure 2).9 Additionally, overexpression of ERG11, sometimes due to gain-of-function mutations in its regulator UPC2, can lead to increased enzyme levels, rendering azole drugs less effective (Figure 3).9
A second major mechanism of azole resistance is active efflux of the drug from the fungal cell. Candida species upregulate membrane transport proteins—specifically ATP-binding cassette (ABC) transporters such as Cdr1p/Cdr2p and major facilitator superfamily transporters like Mdr1p—which pump azole molecules out of the cell, thus lowering the intracellular drug concentration (Figure 4).10 Efflux pump upregulation is responsible for dose-dependent susceptibility for C. glabrata. Some yeasts exhibit intrinsic azole resistance; for example, C. krusei has a naturally low affinity for ERG11 and exhibits high efflux activity, rendering it inherently resistant to fluconazole.2
Action in Molds: In A. fumigatus, azole resistance is commonly associated with mutations in the CYP51A gene (analogous to ERG11 in Candida) that reduce the drug’s binding affinity (Figure 2). Over the past decade, 2 environmental mutations—TR34/L98H and TR46/Y121F/T289A—have emerged as the dominant mechanisms. These mutations consist of tandem repeats in the promoter and point mutations in the CYP51A coding region.11 Besides target-site alterations, Aspergillus may use efflux pumps or stress responses to survive azole exposure (Figure 4). However, CYP51A mutations remain the primary proven mechanism in clinical isolates. Other molds, such as Fusarium, also exhibit azole resistance, typically via intrinsic traits or CYP51A mutations, but these mechanisms are poorly understood.
Echinocandins (eg, caspofungin, micafungin, anidulafungin) inhibit beta-1,3-D-glucan synthase, thus disrupting the formation of the fungal cell wall glucan (Figure 1). In Candida, resistance to echinocandins is usually due to point mutations in the FKS1 or FKS2 genes, which encode the glucan synthase catalytic subunits. Mutations in 2 hotspot regions of FKS1 and in parallel regions of FKS2 (in C. glabrata) reduce the enzyme’s affinity for the drug (Figure 2).9 Echinocandin resistance in Candida was first reported around 2005 and remains uncommon, largely because mutations in FKS genes often carry a fitness cost: Mutant strains grow more slowly in the absence of the drug.9 However, in patients receiving long-term echinocandin therapy (eg, for refractory candidemia or prophylaxis), resistant mutants can occur. C. glabrata is particularly notable for its echinocandin resistance, as it harbors both FKS1 and FKS2 genes and is prone to developing mutations in either gene under drug pressure.9
Although some Candida species, such as C. parapsilosis, have naturally higher echinocandin minimum inhibitory concentrations due to a polymorphism (P660A) in FKS1, they are not fully resistant.9 Of note, no Candida species is intrinsically echinocandin- resistant in the same way that C. krusei is intrinsically resistant to azoles. Instead, resistance develops primarily through mutations.
In molds, clinical echinocandin resistance is less common. Although Aspergillus can acquire FKS1 mutations in laboratory settings, echinocandins typically are used in combination with other agents or as salvage therapy, making resistance less frequently observed or tested. Cryptococcus species and Mucorales are intrinsically resistant to echinocandins due to the absence of functional beta-1,3-D-glucan synthase target, unique cell wall composition. Additionally, echinocandins have no in vitro activity against Trichosporon and exhibit only weak activity against the yeast forms of Histoplasma capsulatum, Blastocystis dermatitidis, and Coccidioides immitis, although they show potent activity against the mycelial forms.12
Polyenes (Amphotericin B) Resistance
Amphotericin B and other polyenes bind to ergosterol in the fungal cell membrane, forming pores that lead to cell death (Figure 1). Acquired resistance to amphotericin B is infrequent, but when it occurs, it is typically due to alterations in the ergosterol biosynthesis pathway, resulting in reduced ergosterol content or modifications to its structure (Figure 2).9 For instance, mutations in ERG3 (sterol-delta-5,6-desaturase) or ERG6 can cause Candida species to accumulate alternative sterols, rendering amphotericin less effective because there is less ergosterol for the drug to bind.9 These mutations have been observed in laboratory-derived strains of C. albicans and C. lusitaniae. Of note, C. lusitaniae has a higher propensity for developing amphotericin B resistance during therapy, sometimes through ERG3 loss-of-function mutations, leading to breakthrough infections.2 C. auris also exhibits variable amphotericin susceptibility, with some clades showing reduced susceptibility. The mechanisms underlying this resistance are under investigation and may involve changes in the ergosterol biosynthesis pathway or stress responses.
In filamentous fungi, intrinsic polyene resistance occurs in A. terreus, Scedosporium, Lomentospora, and some Fusarium species. The exact mechanisms of resistance differ but often involve lower ergosterol content or binding affinity. For example, A. terreus naturally produces molecules (eg, terreic acid) that interfere with amphotericin, and its cell membranes contain less ergosterol, thus explaining its relative resistance. Although polyene resistance is less common than azole or echinocandin resistance, it can evolve under drug pressure by downregulating or altering ergosterol biosynthesis. This change typically comes with a fitness cost, which is why amphotericin B resistance remains sporadic.
Other Mechanisms
Fungi respond to the cell wall stress induced by echinocandins through a salvage mechanism, such as increased chitin synthesis. In response to antifungal-induced stress, there is upregulation of HSP90 and its client proteins, which activate pathways such as drug efflux pumps to mitigate the effects of treatment. Azoles cause the accumulation of toxic sterols, but fungi counter this by downregulating the pathways responsible for their synthesis. Additionally, under stress conditions, fungi may undergo aneuploidy, altering chromosomal numbers to modulate the expression of specific pathways.13 Some fungi, such as Cryptococcus, produce titan cells, which are significantly larger and exhibit altered cell structures that impair drug efficacy by reducing the ability of antifungals to penetrate the cell wall or membrane effectively (Table 1).14
| Table 1. Mechanism of Action and Resistance to Antifungals ABC, ATP-binding cassette; MF, major facilitator. Based on reference 14. | |||
| Antifungal class | Mode of action | Resistance mechanisms | Intrinsically resistant fungi |
|---|---|---|---|
| Echinocandins | Inhibition of beta-1,3-D-glucan synthase disrupting cell wall integrity and causing cell wall stress |
|
|
| Azoles | Inhibition of lanosterol 14-alpha-demethylase blocking ergosterol biosynthesis and leading to accumulation of toxic sterols causing cell membrane stress |
|
|
| Polyenes | Forms extra-membrane aggregates extracting ergosterol from cell membrane (acting as a sterol sponge) |
|
|
2 ID Nightmares: C. auris and Trichophyton indotineae
C. auris exhibits intrinsically high tolerance to azoles and a remarkable ability to acquire further resistance. In the United States, more than 90% of C. auris isolates are fluconazole-resistant, primarily due to mutations in the ERG11 gene (eg, Y132F, K143R), in combination with the overexpression of efflux pumps.15 Approximately 30% of isolates show resistance to amphotericin B, with elevated minimum inhibitory concentrations. Echinocandins remain the last fully effective treatment, although C. auris can develop mutations in FKS1 (eg, S639F), conferring resistance to this class as well. This pathogen often demonstrates pan-resistance to azoles, amphotericin B, and echinocandins, complicating treatment and management. Its ability to persist on surfaces and skin contributes to widespread healthcare-associated outbreaks.
Trichophyton indotineae is an emerging dermatophyte fungus responsible for widespread ringworm outbreaks in South Asia and has recently been identified in the United States. It is particularly notable for its high-level resistance to terbinafine, the first-line antifungal treatment for skin infections. This resistance is primarily due to mutations in the squalene epoxidase gene (ERG1), the enzyme inhibited by terbinafine. Point mutations such as Phe397Leu in ERG1 prevent the drug from binding, rendering treatment ineffective. Strains of T. indotineae often require alternative therapies, such as itraconazole or griseofulvin, but some isolates are also developing resistance to azoles, with mutations in the ERG11 gene observed alongside terbinafine resistance. Although T. indotineae typically causes superficial infections rather than invasive disease, its rapid spread necessitates prolonged treatment with second-line agents.16
The Role of PCR-Based Diagnostics
Early diagnosis of invasive fungal infections and any associated resistance is crucial for improving patient outcomes and reducing healthcare costs. Traditional culture and susceptibility testing for fungi can take days to weeks, during which inappropriate empiric therapy may prove ineffective. In contrast, molecular diagnostics, particularly polymerase chain reaction (PCR)-based methods, allow for much faster identification of fungi and, in some cases, their resistance markers, thus enabling earlier targeted therapy. PCR tests can detect fungal DNA in clinical specimens—such as blood, tissue, and bronchoalveolar lavage—with high sensitivity, often even when cultures yield negative results. Several FDA-cleared PCR-based pathogen identification panels can identify Candida species within minutes to a few hours of growth in blood culture bottles. This rapid species identification facilitates targeted antifungal therapy, helping to avoid mismatches between the pathogen and the drug. Two FDA-cleared PCR fungal tests are the FilmArray BCID Panel and the BIOFIRE BCID 2 Panel (both bioMérieux).17
In addition to pathogen identification, advanced PCR assays can directly detect antifungal resistance genes or mutations. While there are multiplex real-time PCR kits capable of detecting different species of Aspergillus, Candida, and Mucor and identifying resistance markers (such as mutations or genes), none are currently available in the United States or FDA cleared.18
Drugs in Clinical Trials
There are several antifungal therapies currently under investigation (Table 2).16 Two are being researched for aspergillosis: opelconazole (PC945, Pulmocide), an inhaled azole in phase 3 trials,19 and BAL-2062 (Basilea), a siderophore–drug conjugate in phase 2.20,21
| Table 2. Newer Antifungals Based on reference 16. | ||||
| Drug | FDA approval | Class/mechanism | Indications/target pathogens | Activity vs resistance |
|---|---|---|---|---|
| Ibrexafungerp (Brexafemme, Glaxo- SmithKline) | 2021 | Oral triterpenoid; inhibits beta-1,3-D-glucan synthase (first-in-class “fungerp”) | Approved for vulvovaginal candidiasis; in trials for invasive candidiasis (including Candida auris). |
|
| Rezafungin (Rezzayo, Melinta) | 2023 | Echinocandin (long-acting) | Approved for candidemia and invasive candidiasis in adults (first new echinocandin in >10 y). Under study for prophylaxis in stem cell transplant patients. |
|
| Oteseconazole (Vivjoa, Mycovia) | 2022 | Azole (oral tetrazole) | Approved for recurrent vulvovaginal candidiasis in women. Narrow indication (not for invasive use). |
|
| Olorofim (F2G) | Pending | Orotomide; inhibits dihydroorotate dehydrogenase (pyrimidine synthesis) | In phase 2/3 trials for invasive mold infections (including aspergillosis, scedosporiosis). Received FDA Breakthrough Therapy designation for refractory coccidioidomycosis and invasive aspergillosis. |
|
| Fosmanogepix (Basilea) | Pending | Prodrug of manogepix; inhibits GWT1 (cell wall mannoprotein anchor synthesis) | Investigational for invasive candidiasis (including C. auris), invasive aspergillosis, and rare molds. An IV and oral broad-spectrum agent. |
|
For cryptococcal meningitis, encochleated amphotericin B (MAT2203, Matinas BioPharma), an oral nano-formulation of amphotericin B, is under investigation, currently finishing phase 2 trials.22
The investigational drugs may have benefits not currently seen on the market. For instance, inhaled opelconazole delivers high lung azole levels for azole-resistant aspergillosis.19 Cochleated amphotericin may allow oral central nervous system delivery of AmB.22 Efficacy against resistant strains is under investigation, with the hope they will address current treatment gaps.
Conclusion
As the number of immunocompromised patients continues to increase due to chemotherapy, organ transplants, and other therapies, the incidence of invasive fungal infections is expected to rise.23 It is essential to understand the mechanisms behind fungal defenses and intrinsic antifungal resistance, ensuring appropriate antifungal use. Antifungal drug use should last for as short a course as necessary because prolonged use increases selection pressure and promotes antifungal resistance. Although the judicious use of antifungals is crucial, it is equally important to use antibacterials responsibly, since unnecessary disruptions to the microbiota can contribute to the increased incidence of fungal infections.24 Minimizing such disruptions helps maintain the delicate balance between bacteria and fungi, reducing the risk for invasive fungal diseases.
Furthermore, the timely removal of indwelling catheters—both urinary and venous—is vital, as these devices can be potential sources of infections.25 Winning the battle against antifungal resistance requires careful and responsible antifungal usage, not only in clinical settings but also in nonclinical applications, such as agriculture. Preserving the effectiveness of existing antifungals and ensuring the development of new options will require a concerted effort across all sectors. With collective action, we can safeguard the antifungal treatments we have, ensuring we do not run out of viable therapeutic options in the future.
References
- Data and statistics on candidemia. CDC. Published April 24, 2024. Accessed December 18, 2025. cdc.gov/candidiasis/data-research/facts-stats/index.html
- Wiederhold NP. Antifungal resistance: current trends and future strategies to combat. Infect Drug Resist. 2017;10:249-259.
- Alexander BD, Johnson MD, Pfeiffer CD, et al. Increasing echinocandin resistance in Candida glabrata: clinical failure correlates with presence of FKS mutations and elevated minimum inhibitory concentrations. Clin Infect Dis. 2013;56(12):1724-1732.
- Tracking C. auris. CDC. Published December 9, 2024. Accessed December 18, 2025. cdc.gov/candida-auris/tracking-c-auris/index.html
- Antimicrobial-resistant Aspergillus. CDC. Published April 24, 2024. Accessed December 18, 2025. cdc.gov/aspergillosis/php/guidance/index.html
- Berger S, El Chazli Y, Babu AF, et al. Azole resistance in Aspergillus fumigatus: a consequence of antifungal use in agriculture? Front Microbiol. 2017;8:1024.
- Benedict K, Jackson BR, Chiller T, et al. Estimation of direct healthcare costs of fungal diseases in the United States. Clin Infect Dis. 2019;68(11):1791-1797.
- Taori SK, Khonyongwa K, Hayden I, et al. Candida auris outbreak: mortality, interventions and cost of sustaining control. J Infect. 2019;79(6):601-611.
- Czajka KM, Venkataraman K, Brabant-Kirwan D, et al. Molecular mechanisms associated with antifungal resistance in pathogenic Candida species. Cells. 2023;12(22):2655.
- Bhattacharya S, Sae-Tia S, Fries BC. Candidiasis and mechanisms of antifungal resistance. Antibiotics (Basel). 2020;9(6):312.
- Camps SM, Rijs AJ, Klaassen CH, et al. Molecular epidemiology of Aspergillus fumigatus isolates harboring the TR34/L98H azole resistance mechanism. J Clin Microbiol. 2012;50(8):2674-2680.
- Chandrasekar PH, Sobel JD. Micafungin: a new echinocandin. Clin Infect Dis. 2006;42(8):1171-1178.
- Lee Y, Robbins N, Cowen LE. Molecular mechanisms governing antifungal drug resistance. NPJ Antimicrob Resist. 2023;1(1):5.
- Zaragoza O, Nielsen K. Titan cells in Cryptococcus neoformans: cells with a giant impact. Curr Opin Microbiol. 2013;16(4):409-413.
- Logan A, Wolfe A, Williamson JC. Antifungal resistance and the role of new therapeutic agents. Curr Infect Dis Rep. 2022;24(9):105-116.
- Sonego B, Corio A, Mazzoletti V, et al. Trichophyton indotineae, an emerging drug-resistant dermatophyte: a review of the treatment options. J Clin Med. 2024;13(12):3558.
- Wieczorkiewicz SM. Timely detection, targeted therapy: advances in rapid diagnostics for the management of invasive fungal infections. Infectious Disease Special Edition. 2025;29:53-61.
- Gudisa R, Harchand R, Rudramurthy SM. Nucleic-acid-based molecular fungal diagnostics: a way to a better future. Diagnostics (Basel). 2024;14(5):520.
- Opelconazole. Pulmocide. Accessed December 18, 2025. pulmocide.com/opelconazole/
- Basilea announces acquisition of novel clinical-stage antifungal for treatment of Aspergillus mold infections. News release. Globe Newswire. October 19, 2023. Accessed December 18, 2025. ml-eu.globenewswire.com/Resource/Download/d66c140b-5816-4eba-a163-3686797586d2
- Basilea receives USD 25 million funding under BARDA agreement to continue to advance novel antifungals fosmanogepix and BAL2062. News release. BioSpace. September 16, 2025. Accessed December 18, 2025. biospace.com/press-releases/basilea-receives-usd-25-million-funding-under-barda-agreement-to-continue-to-advance-novel-antifungals-fosmanogepix-and-bal2062
- MAT2203. Matinas BioPharma. Accessed December 18, 2025. matinasbiopharma.com/lnc-technology/mat2203
- Giannella M, Lanternier F, Dellière S, et al. Invasive fungal disease in the immunocompromised host: changing epidemiology, new antifungal therapies, and management challenges. Clin Microbiol Infect. 2025;31(1):29-36.
- Drummond RA, Desai JV, Ricotta EE, et al. Long-term antibiotic exposure promotes mortality after systemic fungal infection by driving lymphocyte dysfunction and systemic escape of commensal bacteria. Cell Host Microbe. 2022;30(7):1020-1033.e6.
- Janum S, Afshari A. Central venous catheter (CVC) removal for patients of all ages with candidaemia. Cochrane Database Syst Rev. 2016;7(7):CD011195.
Copyright © 2026 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Resisting Resistance: The Growing Threat of Antifungal Failure
Resisting Resistance: The Growing Threat of Antifungal Failure
This article is from the February 2026 print issue.