Antibiotic-resistant pathogens stemming from antibiotic overuse are estimated to be responsible for 2.8 million antibiotic-resistant infections and 35,000 deaths in the United States in 2019.1,2 The CDC established the Core Elements of Hospital Antibiotic Stewardship Programs in 2014, and subsequently the Infectious Diseases Society of America (IDSA), the Society for Healthcare Epidemiology of America (SHEA), and the Pediatric Infectious Diseases Society (PIDS) created guidelines to “improve and measure the appropriate use of antimicrobial agents by promoting the selection of the optimal antimicrobial drug regimen including dosing, duration of therapy (DOT), and route of administration.”3,4
IDSA and SHEA instituted guidelines in 2016 and jointly published a manuscript in 2018, indicating the use of rapid diagnostic testing (RDT) for bloodstream infections (BSI) and respiratory infections as an appropriate antimicrobial stewardship (AMS) measure.3,5 This intersects with the CDC push toward more awareness and implementation of diagnostic stewardship, or the role that test ordering and reporting plays in AMS.6 There is strong evidence that the use of RDT for BSIs can reduce mortality, time to effective and optimal therapy, hospital length of stay (LOS), and healthcare costs among a wide variety of hospitals and populations.7 RDT can also be used to strengthen AMS programs under other syndromes, such as central nervous system (CNS), gastrointestinal (GI), and sexually transmitted infections (STIs), as well as in solid organ transplants.8-12 This article discusses some of the newest RDT technology to receive FDA clearance, technology still awaiting approval, and the reported effects on RDT integration with AMS programs (ASPs).
Respiratory Syndromes
Discerning between viral and bacterial etiologies is a major difficulty in diagnosing respiratory infections. Misdiagnosis or late diagnosis can lead to the misuse of antibiotics in viral respiratory infections and is a major cause of inappropriate antibiotic prescribing.7 Multiple studies reviewed in meta-analyses have suggested decreases in time to diagnosis, antibiotic use, and LOS in both emergency and pediatric populations when RDT is used properly.7,13-16 The BioFire FilmArray system has been used extensively to help diagnose respiratory infections. The FDA recently approved the BioFire FilmArray Pneumonia Panel plus (bioMérieux), intended to run on the BioFire FilmArray Torch System. This panel can detect 27 microbial targets (18 bacterial and 9 viral) and 7 antimicrobial resistance genes in 1 hour.17
Two recent studies suggest the BioFire FilmArray Pneumonia and Pneumonia Plus panels have strong capacity to aid in AMS goals. The first was a randomized controlled trial that showed significantly reduced time to results-directed antibiotic de-escalation (from 46.5 to 4.8 hours) in lower respiratory tract infections (LRTIs) in the ICU.18 The second study investigated potential interventions based on timely, accurate results from the pneumonia panel and suggested that 48.2% of patients enrolled in this retrospective study would have been eligible for antibiotic de-escalation or discontinuation.19 The Unyvero LRT BAL (Fisher Scientific) has FDA approval for diagnosis of LRTIs; however, no current studies have evaluated its ability to aid in ASPs. A similar panel, the Unyvero Hospitalized Pneumonia panel, was evaluated in a randomized study and reduced time on inappropriate antibiotic treatment by 38.6 hours.20 Other rapid diagnostic tests for respiratory syndromes that offer panels are also available, such as the Cobas Influenza A/B & RSV and ePlex Respiratory Pathogen Panel (both by Roche).10,21 Differences between patient populations were also noted. Pediatric patients (ages 5-6 months) still received antibiotic prescriptions in 20% of influenza-positive cases reviewed, but it was also found that duration of usage was decreased (from 3.4 to 3.2 days) often with the use of diagnostic testing.13,22 While some reviews failed to find any statistically significant reduction in antibiotic use, most did highlight a decrease in DOT, suggesting that RDT can aid in the AMS mission as it relates to respiratory syndromes.
Gastrointestinal Syndromes
Detection of Clostridioides difficile is often factored into metrics within ASPs.23 This alone necessitates investigating the efficacy of RDT in assisting stewardship, but detection of other organisms can also be valuable. There are not as many rapid diagnostic multipanel tests available for GI syndromes as for respiratory syndromes, and subsequently very few studies exist investigating the effects that these tests have on ASPs.
One test that received FDA clearance is the QIAstat-Dx Gastrointestinal Panel 2 (Qiagen), which has the capability of 16 bacterial, parasitic, and viral targets.24 At the time of this publication, only 1 study has been conducted evaluating the effectiveness for appropriate antimicrobial usage in conjunction with the QIAstat-Dx Gastrointestinal Panel. The authors determined that compared with standard of care, this panel led to informed antibiotic treatment changes in 14.4% of patients in a pediatric hospital.25 A few older studies looked at the BioFire FilmArray GI panel, which can currently detect up to 22 pathogens.26 One prospective study did not find statistically significant differences between antibiotic days when the panel was compared with historical laboratory testing (1.73 vs 2.12 days), although LOS following collection was reduced (3.6 vs 4.3 days) and the number of additional stool tests required for identification was reduced (0.58 vs 3.02).27
Although the authors did not provide direct data, these latter factors can aid in appropriate antibiotic use and promote AMS goals. The other 2 retrospective studies did find that antibiotic usage was changed significantly by implementation of the molecular rapid diagnostic test. One study showed a reduction in the percentage of patients prescribed antibiotics within 14 days of RDT results as opposed to culture methods (36.2% vs 40.9%).28 Another study looked more closely at the relationship with AMS and the use of the BioFire FilmArray GI panel among hospitalized patients, comparing the outcomes and antibiotic usage before and after AMS implementation. Inappropriate antibiotic use was reduced compared with pre-AMS measures when used with RDT (25.8% vs 42.9%).29
There continues to be a lack of literature about the effectiveness of using GI RDT in support of AMS. Two other FDA-cleared rapid diagnostic tests also have been mentioned—Luminex xTAG Gastrointestinal Pathogen Panel and Verigene Enteric Pathogens Test (both Diasorin)—but no studies have determined their effect on AMS.15 The gap in literature covering AMS in GI syndromes highlights the need for further studies.
Bloodstream Infections
Bloodstream infections (BSIs) have a strong association with increased morbidity and mortality.30 Accurate identification of the pathogen is important for appropriate antibiotic use and improving patient outcomes. Correct identification of BSI pathogens requires a positive blood culture and a subsequent diagnostic test. Even with RDT in place, results could take 1 to 5 days before the most optimal antibiotic is prescribed for the best possible patient outcome.31 Multiple categories of RDT are cleared by the FDA for identifying BSIs, including peptide nucleic acid fluorescent in situ hybridization (PNA-FISH), polymerase chain reaction (PCR) and multiplex or panel PCR, matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS), and a nuclear magnetic resonance assay.23,31 The BioFire Blood Culture Identification 2 (BCID2) Panel is the most recent to receive FDA approval at the time of this publication. The panel can identify 43 targets (26 bacterial, 7 yeast, and 10 antimicrobial resistance genes) in around 1 hour.32
There have been several new studies conducted on RDT in BSI identification and the impact on ASPs. Two of these studies specifically investigated the effect that the BCID2 panel had on appropriateness of antimicrobials and time to optimal treatment. Time to effective therapy was reduced from 10 to 3.75 hours in healthcare settings that had ASPs that subsequently added BCID2 to their diagnostic testing.33 The second study reported rapid identification of methicillin-susceptible Staphylococcus aureus in BSI, wherein the authors observed reductions in antibiotic initiation, antibiotic durations (44.2 vs 20.9 hours), and even in duration of bacteremia (68.6 vs 47.3 hours).34 Tate et al reported similar results related to streptococcal bacteremia, showing time to de-escalation decreasing significantly (88 vs 42 hours).35 Another study compared time of inpatient antibiotic therapy, among other patient outcomes, before and after implementation of both RDT and AMS. The authors saw a significant reduction in length of inpatient therapy (6.7 vs 8.8 days) when the programs were implemented.36 Compared with 3 traditional methods for identifying antibiotic nonsusceptibility, Verigene multiplex PCR was faster and more accurate for detecting third-generation cephalosporin resistance (3GCR) by screening for CTX-M. Verigene correctly predicted ceftriaxone (a third-generation cephalosporin) susceptibility 97.3% of the time as opposed to a mean of 79.9% with 3 separate traditional scoring methods. This led to only 0.3% overtreatment and 15.1% undertreatment by antibiotics with Verigene compared with the means of 10.9% and 67.6%, respectively, for traditional methods.37
Another retrospective study indicated statistically significant improvements in early de-escalation (38.1% vs 34.6%), appropriate antimicrobial therapy (93.8% vs 91.7%), and time to appropriate therapy (8 vs 9 h) when blood culture multiplex PCR was used for identification.38 Walsh et al looked at the Accelerate Pheno FISH system (Accelerate Diagnostics) that evaluates minimum inhibitory concentrations for antibiotic susceptibility testing (AST), with a hospital ASP to determine whether rapid AST and identification would improve outcomes in BSI patients. They found a strong association with decreased DOT (9.5 vs 14.2 days), duration of effective therapy (9.3 vs 14.2 days), and overall LOS (5.3 vs 7.9 days).39 Rapid AST performed with MALDI-TOF analysis compared with conventional identification and AST was examined in another recent study. The authors found a significant reduction in time to narrowest therapy (68 vs 89 hours) when patients already on the most appropriate therapy at time of organism identification were excluded. Table 1 summarizes the findings of the entire cohort.40 Timbrook et al cited multiple studies determining that only through AMS programs does RDT for BSI improve antibiotic use, thus testing should only be integrated when proper AMS guidance is available.23 The current literature supports that statement, but when implemented with AMS, the benefits to stewardship and to the patient are evident.
| Table 1. Median Time for Therapeutic Milestones Between Conventional AST and Rapid AST | ||||
| Test type | Susceptibilities | Narrowest therapy | Oral therapy | Discharge |
|---|---|---|---|---|
| Conventional AST | 62 h | 73 h | 126 h | 7 d |
| Rapid AST | 39 h | 64 h | 91 h | 5 d |
| AST, antimicrobial susceptibility testing; d, days; h, hours. Adapted from reference 40. | ||||
Central Nervous System Infections
CNS infections are associated with increased morbidity and long-term neurologic complications, so quick administration of empiric antibiotics is often considered paramount in improving patient outcomes.41 Because time is a critical factor and empiric antibiotic treatment is likely to be administered, DOT is going to be investigated for this syndrome. The BioFire FilmArray Meningitis Encephalitis (ME) is the only FDA-cleared multipanel rapid diagnostic test for CNS syndromes available at the time of this publication and can detect up to 14 different targets.15,42 There are a few studies comparing the effectiveness of this assay in reducing antibiotic usage with mixed results.
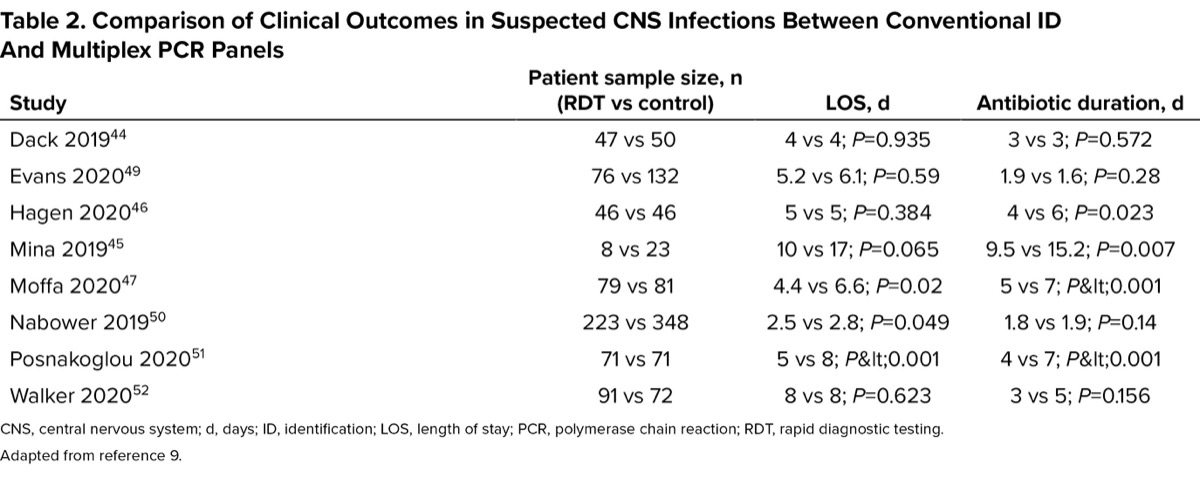
Early studies indicated that the implementation of the BioFire FilmArray ME did little to alter DOT when a negative bacterial result was obtained: a study from 2018, stating the median DOT was 3 days43 and another from 2019, showing a reduction in DOT that was not statistically significant (3.6 vs 3.1 days).44 A review by Goodlet et al indicated that 4 out of 8 studies measuring DOT showed statistically significant antibiotic duration reductions, and the findings are summarized in Table 2.9 These data showed a range from 5.7-day45 to 2-day46 reductions, but all sample sizes were relatively small, with a control group of 81 patients being the largest among statistically significant findings.47 The most recent study published was an analysis of the BioFire FilmArray ME being implemented alongside a diagnostic stewardship program in a pediatric ward. The post-implementation cohort had reduced time to optimal antimicrobial (28 vs 18 hours) and reduced IV antimicrobial durations (36 vs 24 hours).48 While older studies struggled to show clear improvement in ASPs with CNS RDT, the most recent literature is promising. Unfortunately, these benefits are understudied, and more work is required to set best-practice guidelines for ASPs.
| Table 2. Comparison of Clinical Outcomes in Suspected CNS Infections Between Conventional ID And Multiplex PCR Panels | |||
| Study | Patient sample size, n (RDT vs control) | LOS, d | Antibiotic duration, d |
|---|---|---|---|
| Dack 201944 | 47 vs 50 | 4 vs 4; P=0.935 | 3 vs 3; P=0.572 |
| Evans 202049 | 76 vs 132 | 5.2 vs 6.1; P=0.59 | 1.9 vs 1.6; P=0.28 |
| Hagen 202046 | 46 vs 46 | 5 vs 5; P=0.384 | 4 vs 6; P=0.023 |
| Mina 201945 | 8 vs 23 | 10 vs 17; P=0.065 | 9.5 vs 15.2; P=0.007 |
| Moffa 202047 | 79 vs 81 | 4.4 vs 6.6; P=0.02 | 5 vs 7; P<0.001 |
| Nabower 201950 | 223 vs 348 | 2.5 vs 2.8; P=0.049 | 1.8 vs 1.9; P=0.14 |
| Posnakoglou 202051 | 71 vs 71 | 5 vs 8; P<0.001 | 4 vs 7; P<0.001 |
| Walker 202052 | 91 vs 72 | 8 vs 8; P=0.623 | 3 vs 5; P=0.156 |
| CNS, central nervous system; d, days; ID, identification; LOS, length of stay; PCR, polymerase chain reaction; RDT, rapid diagnostic testing. Adapted from reference 9. | |||
Additional Rapid Diagnostic Platforms
There are numerous RDT technologies that have not been evaluated regarding their potential to aid in stewardship efforts. However, they may add to AMS once clinical evaluations have been completed.
The BD MAX systems (Becton Dickinson) have assays for enteric syndromes, hospital-acquired infections, women’s health, and STIs.53 Thermo Fisher Scientific’s clinical PCR testing with TaqPath technology is available for the detection of COVID-19, influenza A and B, and mpox.54,55 Beckman Coulter provides identification technology using the Bruker MALDI-TOF platform56 for several applications, including identification of infectious agents in addition to the presence of carbapenemases or extended-spectrum beta-lactamases.
Bruker also offers a sepsis solution with the MALDI Biotyper CA System and the MBT Sepsityper Kit US IVD, which can identify over 500 organisms in about 20-30 minutes.
The Quidel Lyra respiratory PCR panel was covered earlier. However, the Quidel Solana instrument uses a proprietary helicase-dependent amplification and fluorescence detection to deliver rapid identification of several targets, including a respiratory panel, streptococcal assay, and C. difficile.57 The Qvella FAST system can identify positive blood cultures in around 2 minutes, although it has not been approved for any downstream applications.58 Sekisui Diagnostics Acucy Influenza A&B reader can provide results within 15 minutes of testing.59 IMMY offers several diagnostic tests for single targets with a focus on fungal pathogens, including rapid lateral flow assays and enzyme-linked immunoassays.60 Qiagen has multiple PCR diagnostic platforms, including the multiplex instrument QIAstat-Dx, which currently has a respiratory panel, with GI and meningitis panels in development.24 Streck has developed several rapid antibiotic resistance gene detection kits with its ARM-D line capable of discerning between multiple classes and alleles of beta-lactamases.61,62 Although Streck has obtained clearance for the ARM-D detection kits for research purposes, none have been approved for diagnostic purposes in the United States. Copan Diagnostics is pioneering automated lab technology, requiring minimal technician interaction, with the fully automated WASP DT: Walk-Away Specimen Processor platform that handles plate culture, broth inoculation, and Gram stain slide preparation.63
Conclusion and a Caveat
As shown in the breadth of studies for BSIs and respiratory syndromes, using RDT with ASPs benefits the patient, hospital, and wider community through decreased antibiotic usage, more appropriate antibiotic use, and decreased LOS. In addition, benefits were also seen in studies examining GI and CNS infections, although more studies are needed. There is also a need for an updated full literature review on the effectiveness of integrated RDT with ASPs, since the last comprehensive review was published by Rader et al, in 2021.15
This review focused on RDT and ASPs, but overall cost was another factor investigated and found to be lower for the patient when RDT and AMS were used. However, these benefits to both patient and hospital cannot be realized without the hospital’s ASP. The studies reviewed here indicate that integration of RDT into ASPs can have a positive impact on both stewardship and healthcare at large.
References
- CDC. Antibiotic Resistance Threats in the United States, 2019. Accessed April 7, 2025. stacks.cdc.gov/view/cdc/82532
- CDC. The Core Elements of Hospital Antibiotic Stewardship Programs. Accessed April 7, 2025. www.cdc.gov/antibiotic-use/hcp/core-elements/hospital.html
- Barlam TF, Cosgrove SE, Abbo LM, et al. Implementing an antibiotic stewardship program: guidelines by the Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America. Clin Infect Dis. 2016;62(10):e51-e77.
- Society for Healthcare Epidemiology of America, Infectious Diseases Society of America, and Pediatric Infectious Diseases Society. Policy statement on antimicrobial stewardship by the Society for Healthcare Epidemiology of America (SHEA), the Infectious Diseases Society of America (IDSA), and the Pediatric Infectious Diseases Society (PIDS). Infect Control Hosp Epidemiol. 2012;33(4):322-327.
- Doernberg SB, Abbo LM, Burdette SD, et al. Essential resources and strategies for antibiotic stewardship programs in the acute care setting. Clin Infect Dis. 2018;67(8):1168-1174.
- Claeys KC, Johnson MD. Leveraging diagnostic stewardship within antimicrobial stewardship programmes. Drugs Context. 2023;12:2022-9-5.
- Beganovic M, McCreary EK, Mahoney MV, et al. Interplay between rapid diagnostic tests and antimicrobial stewardship programs among patients with bloodstream and other severe infections. J Appl Lab Med. 2019;3(4):601-616.
- Gradisteanu Pircalabioru G, Iliescu FS, Mihaescu G, et al. Advances in the rapid diagnostic of viral respiratory tract infections. Front Cell Infect Microbiol. 2022 Feb 10;12:807253.
- Goodlet KJ, Tan E, Knutson L, Nailor MD. Impact of the FilmArray meningitis/encephalitis panel on antimicrobial duration among patients with suspected central nervous system infection. Diagn Microbiol Infect Dis. 2021;100(4):115394.
- Bouzid D, Zanella MC, Kerneis S, et al. Rapid diagnostic tests for infectious diseases in the emergency department. Clin Microbiol Infect. 2021;27(2):182-191.
- Meyer T, Eberle J, Roß RS, et al. Schnelltestdiagnostik sexuell übertragbarer Infektionen. Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz. 2020;63(10):1271-1286.
- Vega AD, Abbo LM. Rapid molecular testing for antimicrobial stewardship and solid organ transplantation. Transpl Infect Dis. 2022;24(5):e13913.
- Weragama K, Mudgil P, Whitehall J. Diagnostic stewardship—the impact of rapid diagnostic testing for paediatric respiratory presentations in the emergency setting: a systematic review. Children. 2022;9(8):1226.
- Dessajan J, Timsit JF. Impact of multiplex PCR in the therapeutic management of severe bacterial pneumonia. Antibiotics. 2024;13(1):95.
- Rader TS, Stevens MP, Bearman G. Syndromic multiplex polymerase chain reaction (mPCR) testing and antimicrobial stewardship: current practice and future directions. Curr Infect Dis Rep. 2021;23(4):5.
- Clark TW, Lindsley K, Wigmosta TB, et al. Rapid multiplex PCR for respiratory viruses reduces time to result and improves clinical care: results of a systematic review and meta-analysis. J Infect. 2023;86(5):462-475.
- BioMérieux. BioFire FilmArray Pneumonia & Pneumonia plus Panels. Accessed March 6, 2025. biomerieux.com/tr/en/our-offer/clinical-products/biofire-filmarray-pneumonia-panels.html
- Poole S, Tanner AR, Naidu VV, et al. Molecular point-of-care testing for lower respiratory tract pathogens improves safe antibiotic de-escalation in patients with pneumonia in the ICU: results of a randomised controlled trial. J Infect. 2022;85(6):625-633.
- Buchan BW, Windham S, Balada-Llasat JM, et al. Practical comparison of the BioFire FilmArray Pneumonia Panel to routine diagnostic methods and potential impact on antimicrobial stewardship in adult hospitalized patients with lower respiratory tract infections. J Clin Microbiol. 2020;58(7):10.1128/jcm.00135-20.
- Darie AM, Khanna N, Jahn K, et al. Fast multiplex bacterial PCR of bronchoalveolar lavage for antibiotic stewardship in hospitalised patients with pneumonia at risk of gram-negative bacterial infection (Flagship II): a multicentre, randomised controlled trial. Lancet Respir Med. 2022;10(9):877-887.
- Hua HX, Wagner JL. RDTs for antimicrobial stewardship: focus on bloodstream and acute respiratory infections. Infectious Disease Special Edition. 2022 Dec 10. Accessed April 9, 2025. https://bit.ly/4bBhyvH-IDSE
- Rogers BB, Shankar P, Jerris RC, et al. Impact of a rapid respiratory panel test on patient outcomes. Arch Pathol Lab Med. 2015;139(5):636-641.
- Timbrook TT, Spivak ES, Hanson KE. Current and future opportunities for rapid diagnostics in antimicrobial stewardship. Med Clin North Am. 2018;102(5):899-911.
- Qiagen. QIAstat-Dx Automated Molecular Panel Tests | QIAGEN. Accessed March 7, 2025. http://www.qiagen.com/us/applications/syndromic-testing/products
- Castany-Feixas M, Simo S, Garcia-Garcia S, et al. Rapid molecular syndromic testing for aetiological diagnosis of gastrointestinal infections and targeted antimicrobial prescription: experience from a reference paediatric hospital in Spain. Eur J Clin Microbiol Infect Dis. 2021;40(10):2153-2160.
- BioMérieux. BioFire FilmArray GI Panel. BioMérieux Clinical Diagnostics. Accessed March 7, 2025. https://www.biomerieux-diagnostics.com/filmarrayr-gi-panel
- Beal SG, Tremblay EE, Toffel S, et al. A gastrointestinal PCR panel improves clinical management and lowers health care costs. J Clin Microbiol. 2017;56(1):e01457-17.
- Axelrad JE, Freedberg DE, Whittier S, et al. Impact of gastrointestinal panel implementation on health care utilization and outcomes. J Clin Microbiol. 2019;57(3):e01775-18.
- Keske S, Zabun B, Aksoy K, et al. Rapid molecular detection of gastrointestinal pathogens and its role in antimicrobial stewardship. J Clin Microbiol. 2018;56(5):e00148-18.
- Schechner V, Wulffhart L, Temkin E, et al. One-year mortality and years of potential life lost following bloodstream infection among adults: a nation-wide population based study. Lancet Reg Health Eur. 2022;23:100511.
- Briggs N, Campbell S, Gupta S. Advances in rapid diagnostics for bloodstream infections. Diagn Microbiol Infect Dis. 2021;99(1):115219.
- BioMérieux. BioFire Blood Culture Identification 2 Panel. Accessed March 7, 2025. www.biomerieux.com/us/en/our-offer/clinical-products/biofire-blood-culture-identification-2-panel.html
- Pérez-Lazo G, del Valle-Mendoza J, Sandoval-Ahumada R, et al. Impact of adding a rapid PCR-based blood culture identification panel to the antimicrobial stewardship program of patients with febrile neutropenia in a Peruvian referral hospital. Antibiotics. 2023;12(4):648.
- Yetukuri J, Patel D, Bandali A, et al. Impact of rapid blood culture identification PCR panel on optimal antibiotic use in methicillin-susceptible Staphylococcus aureus bacteremia. Microbiol Spectr. 2024;12(12):e00629-24.
- Tate T, Viehman JA, Shields RK. Rapid molecular testing is associated with decreased broad-spectrum antibiotic use among patients with streptococcal bloodstream infections. Antimicrob Steward Healthc Epidemiol ASHE. 2024;4(1):e170.
- Cavalieri SJ, Kwon S, Vivekanandan R, et al. Effect of antimicrobial stewardship with rapid MALDI-TOF identification and Vitek 2 antimicrobial susceptibility testing on hospitalization outcome. Diagn Microbiol Infect Dis. 2019;95(2):208-211.
- Cwengros LN, Mynatt RP, Timbrook TT, et al. Minimizing time to optimal antimicrobial therapy for Enterobacteriaceae bloodstream infections: a retrospective, hypothetical application of predictive scoring tools vs rapid diagnostics tests. Open Forum Infect Dis. 2020;7(8):ofaa278.
- Britt NS, Khader K, He T, et al. Examining the clinical impact of rapid multiplex polymerase chain reaction-based diagnostic testing for bloodstream infections in a national cohort of the Veterans Health Administration. Pharmacother J Hum Pharmacol Drug Ther. 2023;43(1):24-34.
- Walsh TL, Bremmer DN, Moffa MA, et al. Impact of an antimicrobial stewardship program-bundled initiative utilizing Accelerate Pheno system in the management of patients with aerobic gram-negative bacilli bacteremia. Infection. 2021;49(3):511-519.
- Christensen AB, Footer B, Pusch T, et al. Impact of a laboratory-developed phenotypic rapid susceptibility test directly from positive blood cultures on time to narrowest effective therapy in patients with gram-negative bacteremia: a prospective randomized trial. Open Forum Infect Dis. 2022;9(7):ofac347.
- Nauclér P, Huttner A, van Werkhoven CH, et al. Impact of time to antibiotic therapy on clinical outcome in patients with bacterial infections in the emergency department: implications for antimicrobial stewardship. Clin Microbiol Infect. 2021;27(2):175-181.
- BioMérieux. The BioFire FilmArray Meningitis/Encephalitis (ME) Panel. BioFire Diagnostics. Accessed April 9, 2025. www.biofiredx.com/products/the-filmarray-panels/filmarrayme/
- Chang D, Okulicz JF, Nielsen LE, et al. A tertiary care center’s experience with novel molecular meningitis/encephalitis diagnostics and implementation with antimicrobial stewardship. Mil Med. 2018;183(12):e24-e27.
- Dack K, Pankow S, Ablah E, et al. Contribution of the BioFire FilmArray Meningitis/Encephalitis Panel: assessing antimicrobial duration and length of stay. Kans J Med. 2019;12(1):1-3.
- Mina Y, Schechner V, Savion M, et al. Clinical benefits of FilmArray meningitis-encephalitis PCR assay in partially-treated bacterial meningitis in Israel. BMC Infect Dis. 2019;19(1):713.
- Hagen A, Eichinger A, Meyer-Buehn M, et al. Comparison of antibiotic and acyclovir usage before and after the implementation of an on-site FilmArray meningitis/encephalitis panel in an academic tertiary pediatric hospital: a retrospective observational study. BMC Pediatr. 2020;20(1):56.
- Moffa MA, Bremmer DN, Carr D, et al. Impact of a multiplex polymerase chain reaction assay on the clinical management of adults undergoing a lumbar puncture for suspected community-onset central nervous system infections. Antibiotics. 2020;9(6):282.
- Messacar K, Palmer C, Gregoire L, et al. Clinical and financial impact of a diagnostic stewardship program for children with suspected central nervous system infection. J Pediatr. 2022;244:161-168.e1.
- Evans M, Merkel KG, Harder J, et al. Impact of the implementation of a rapid meningitis/encephalitis multiplex polymerase chain reaction panel on IV acyclovir duration: multicenter, retrospective cohort of adult and pediatric patients. Diagn Microbiol Infect Dis. 2020;96(2):114935.
- Nabower AM, Miller S, Biewen B, et al. Association of the FilmArray Meningitis/Encephalitis Panel with clinical management. Hosp Pediatr. 2019;9(10):763-769.
- Posnakoglou L, Siahanidou T, Syriopoulou V, et al. Impact of cerebrospinal fluid syndromic testing in the management of children with suspected central nervous system infection. Eur J Clin Microbiol Infect Dis. 2020;39(12):2379-2386.
- Walker M, Taylor K, Beaulieu R, et al. The clinical impact of the FilmArray Meningitis/Encephalitis (ME) Panel on antimicrobial utilization in a tertiary hospital setting. Am J Med Sci. 2021;361(4):550-552.
- Becton Dickinson. BD MAX system. Accessed March 7, 2025. www.bd.com/en-ca/offerings/capabilities/molecular-diagnostics/molecular-systems/bd-max-system
- Thermo Fisher Scientific. TaqPath COVID-19, Flu A, Flu B Multiplex Diagnostic Solution - US. Accessed March 7, 2025. www.thermofisher.com/us/en/home/clinical/clinical-genomics/pathogen-detection-solutions/covid-19-sars-cov-2/influenza-a-b-multiplex.html
- Thermo Fisher Scientific. Quality Monkeypox Virus Testing Workflow Solutions | Thermo Fisher Scientific - US. Accessed March 7, 2025. www.thermofisher.com/us/en/home/clinical/clinical-genomics/pathogen-detection-solutions/monkeypox.html
- Beckman Coulter. Bruker MALDI Biotyper Microbiology Systems. Accessed March 7, 2025. www.beckmancoulter.com/products/microbiology/bruker-maldi-biotyper-system
- Quidel. Solana | Quidel. Accessed March 7, 2025. www.quidel.com/molecular-diagnostics/solana-products
- Qvella. FAST System. qvella.com. Accessed March 7, 2025. www.qvella.com/fast-prep-pbc-2/
- Sekisui Diagnostics. Acucy Influenza A&B. Accessed March 7, 2025. sekisuidiagnostics.com/product/acucy-influenza-ab/
- IMMY. Accessed March 7, 2025. www.immy.com/
- Streck. ARM-D Kit, ß-Lactamase. Accessed March 7, 2025. www.streck.com/products/molecular/antibiotic-resistance-kits/arm-d-kit-b-lactamase/
- Streck. ARM-D Kit, ampC. Accessed March 7, 2025. www.streck.com/products/molecular/antibiotic-resistance-kits/arm-d-kit-ampc/
- Copan Diagnostics. WASP DT Accessed March 7, 2025. www.copanusa.com/full-lab-automation-and-artificial-intelligence/wasp/
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Advancing Antibiotic Stewardship With Rapid Diagnostic Testing
Advancing Antibiotic Stewardship With Rapid Diagnostic Testing