When sheep are in the field grazing on grass, they are at risk for being attacked by wolves. When attacked, it usually results in the death of the sheep. Farmers use herding dogs to guard and protect their sheep. If the dog sees a wolf lurking in the distance, the dog begins to bark and herds the sheep in a direction away from the wolf. This farming example of a dog protecting its flock of sheep from harm’s way is analogous to introducing new antibiotics and the role of antimicrobial stewardship (AMS) programs.
The antibiotic is the “sheep,” and the pharmacist and physician stewards are the “dogs.” The role of the antibiotic steward is to protect new antibiotics from the “wolf” (antimicrobial resistance [AMR]) and harm’s way (inappropriate use) by overseeing the new antibiotic to make sure it is prescribed appropriately, so the medication’s effectiveness is preserved as long as possible.
Increasing AMR Due to Lack of Stewardship
What happens when new antibiotics are introduced in a country without an effective AMS strategy? The introduction of 2 new beta-lactam/beta-lactamase inhibitor combinations, ceftazidime-avibactam (CA) and ceftolozane-tazobactam (CT), in South Africa provides an answer.1 The emergence of antibiotic resistance after short courses of therapy with CT and CA highlights the importance of AMS stewards establishing strict criteria for the use of new antibiotics.
However, as physicians in the private sector continued to prescribe these new antibiotics without adhering to AMS guidance, increasing levels of resistance were observed, jeopardizing these agents’ long-term viability as treatment options of last resort. In essence, they let the sheep graze in the grass without the guard dogs watching for the wolves. Appropriate stewardship practices are critical to maximize the efficacy and longevity of all new antibiotics that enter clinical practices. Ideally, no hospital should allow physicians to prescribe a new antibiotic without the oversight of AMS stewards.
In the United States, AMS programs are required in all hospitals. The Infectious Diseases Society of America (IDSA) recommends that AMS teams should be co-led by an infectious diseases (ID) physician and ID pharmacists, and should include a microbiologist, infection control nurse, and information technology manager. This is a great recommendation, but in reality, it is not possible in every US hospital due to shortages of ID physicians and ID pharmacists. When local resources are not available, the CDC Core Elements of Hospital Antibiotic Stewardship suggest the use of off-site or remote ID-trained providers.2 Telehealth is a promising tool for AMS, especially in resource-limited hospitals.3 The requirement for AMS programs in US hospitals ensures that new antibiotics are only prescribed with AMS oversight and guidance.
In contrast to the United States and other high-income countries (HICs), many low- and middle-income countries (LMICs) have few, if any, ID-trained pharmacists and physicians; therefore, other AMS models are necessary.4 Most LMICs’ National Action Plans include AMS, but based on my personal in-country experiences, several countries have not been able to put the “action” into the plan. The Tripartite country self-assessment reports show LMICs lag behind HICs in all indicators of the implementation and financing of National Action Plans.5
Much work remains to be done. Lack of funding, lack of AMS champions, weak health systems, and post-pandemic burnout are just a few reasons for the slow implementation of multidisciplinary AMS programs. While LMICs are working to implement AMS programs, they face challenges accessing new antibiotics that target infections caused by multidrug-resistant (MDR) organisms. The rate of antibiotic resistance in LMICs is often much higher than the rate in HICs. Therefore, these countries desperately need access to new antibiotics.6 For multiple reasons, such as the lack of infrastructure, poor healthcare systems, and regulatory hurdles, in most cases, pharmaceutical companies will not register a new antibiotic in LMICs. Bold thinking and meaningful collaboration are needed to address this gap. Sadly, the lack of access to new antibiotics means many patients with MDR infections die.
First-of-Its-Kind Partnership Ensuring Proper Use Of a New Antibiotic
Cefiderocol is one example of a new antibiotic developed by Shionogi & Co, Ltd for treating MDR gram-negative infections. It has been available for use in the United States since 2019. AMS stewards developed guidelines for appropriate use prior to authorization.7 How will this sheep be protected from the wolves and harm’s way in LMICs without effective enforcement of AMS programs?
To address this problem, a bold first-of-its-kind partnership was formed in 2021 between three organizations:
- the Global Antibiotic Research & Development Partnership (GARDP);
- the Clinton Health Access Initiative (CHAI); and
- Shionogi & Co, Ltd.
Their goal is to accelerate access to cefiderocol for MDR gram-negative infections in patients with limited treatment options in LMICs. Importantly, the collaboration aims to work with governments and partners to introduce cefiderocol into health systems across 135 countries, together with measures that support appropriate use.
While I applaud this strategy, I believe it will require the additional collaboration of AMS experts with hands-on AMS implementation experience in LMICs. We already know that National Action Plans and free AMS tool kits are insufficient to implement successful AMS programs.
With global AMS collaboration, we will protect the sheep from the wolves and harm’s way. If we can succeed, we may see new antibiotics used more appropriately in the near future.
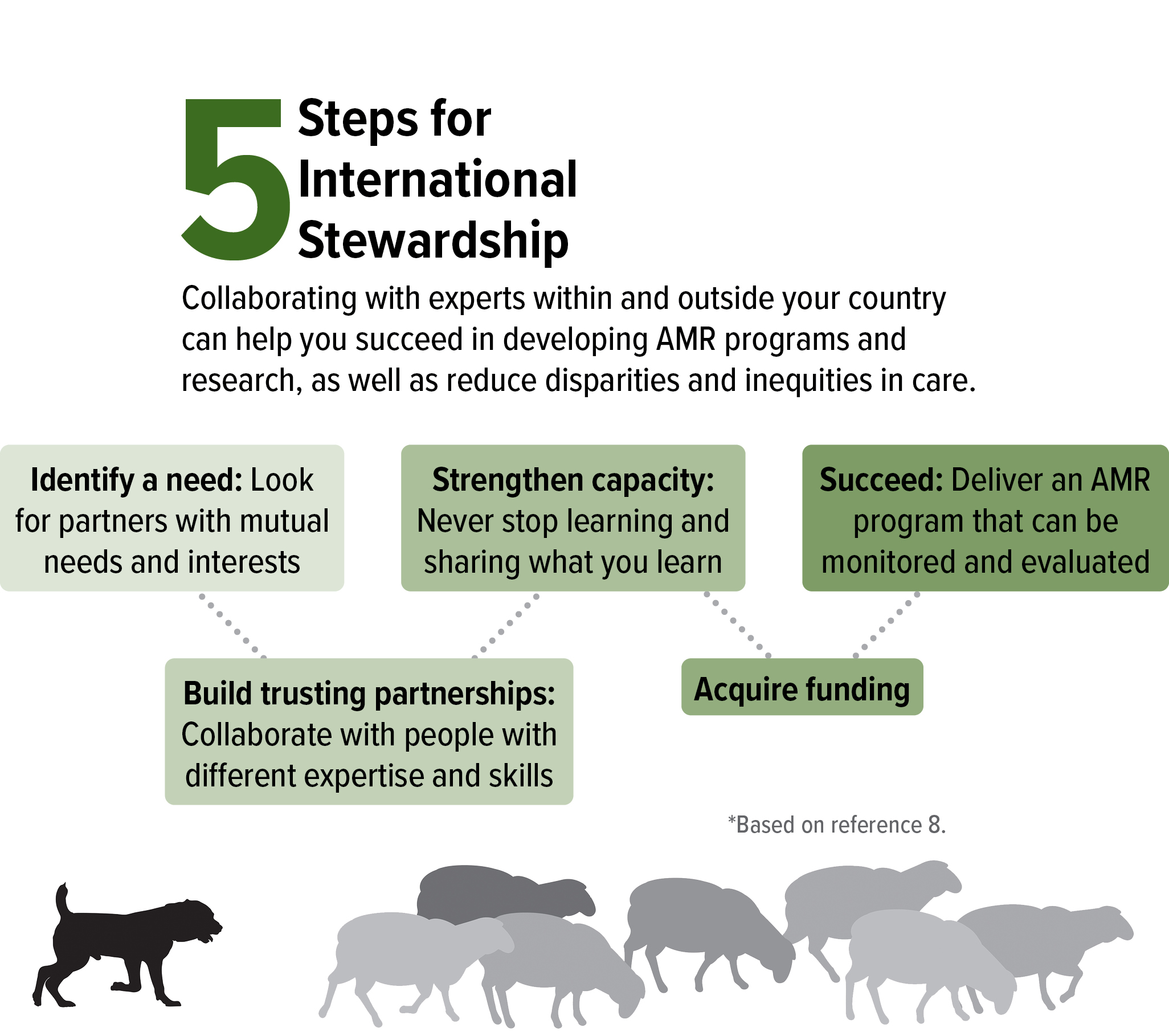
The missing element is knowledge of applying behavior change science, multidisciplinary intervention-implementation research, and having a helping hand from AMS experts. The current understanding of AMR, antibiotic use, access, and AMS implementation comes mainly from hospitals in HICs. An international group of AMS experts from across resource settings recently described how antimicrobial stewards can provide support to colleagues in LMICs struggling to get AMS started (Figure 1).8 We urgently need more dogs in the fight to generate new strategies and programs like this partnership to preserve these precious resources and slow AMR.
A Call to Action
We have had enough advocacy. Let’s put some action behind the words and just do it. Let’s form more stewardship partnerships across resource settings to implement AMS in these 132 countries.9 Antimicrobial stewards have the opportunity to develop the global road map for introducing new antibiotics in LMICs with context-appropriate AMS. With global AMS collaboration, we will protect the sheep from the wolves and harm’s way. If we can succeed, we may see a “flock” of new antibiotics used more appropriately in the near future.
References
- Brink AJ, Coetzee J, Richards GA, et al. Best practices: appropriate use of the new beta-lactam/beta-lactamase inhibitor combinations, ceftazidime-avibactam and ceftolozane-tazobactam in South Africa. S Afr J Infect Dis. 2022;37(1):453.
- CDC. Implementation of antibiotic stewardship core elements at small and critical access hospitals. 2020. Accessed April 9, 2025. www.cdc.gov/antibiotic-use/hcp/core-elements/small-and-critical-access-hospitals.html
- Pierce J, Stevens MP. The emerging role of telehealth in antimicrobial stewardship: a systematic review and perspective.Curr Treat Options Infect Dis. 2021;13(4):175-191.
- Goff DA, Bauer KA, Brink A, et al. International Train the Trainer antibiotic stewardship program for pharmacists: implementation, sustainability, and outcomes. J Am Coll Clin Pharm. 2020;3(5):869-876.
- Khurana MP, Essack S, Zoubiane G, et al. Mitigating antimicrobial resistance (AMR) using implementation research: a development funder’s approach. JAC Antimicrob Resist. 2023;5(2):dlad031.
- Shamas N, Stokle E, Ashiru-Oredope D, et al. Challenges of implementing antimicrobial stewardship tools in low to middle income countries (LMICs). Infect Prev Pract. 2023;5(4):100315.
- Chou A, Ramsey D, Amenta E, et al. Real-world experience with cefiderocol therapy for Pseudomonas aeruginosa and other multidrug resistant gram-negative infections within the Veterans Health Administration, 2019-2022. Antimicrob Steward Healthc Epidemiol. 2023;3(1):e90.
- Ashiru-Oredope D, Langford BJ, Bonaconsa C, et al. Global collaborations in antimicrobial stewardship: all hands on deck. Antimicrob Steward Healthc Epidemiol. 2023;3(1):e66.
- Goff D, Prusakov P, Mangino JE, et al. International Train the Trainer neonatal antibiotic stewardship program for South African pharmacists. J Am Coll Clin Pharm. Published online August 26, 2021. doi.org/10.1002/jac5.1517
This viewpoint was originally published on the REVIVE website revive.gardp.org, an activity of the Global Antibiotic Research & Development Partnership. It has been edited for style and length.
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Debra A. Goff, PharmD
Debra A. Goff, PharmD