Significant advancements have been and continue to be made in the realm of rapid diagnostic tests (RDTs) for the diagnosis of various diseases. RDTs are highly integrated into infection management, and the field of diagnostic stewardship has emerged as an important component to antimicrobial stewardship (AMS). Diagnostic stewardship in the realm of infectious diseases encompasses the process of modifying the ordering, collection, performance, and/or reporting of diagnostic tests to improve the diagnosis and treatment of infections.1,2 With more than 3,500 RDTs to choose from, it is imperative to combine the implementation of RDTs with AMS efforts to achieve favorable and beneficial results, while maximizing the cost-effectiveness of the test.
The goal in using RDTs is to expedite the diagnosis to provide appropriate initial therapeutic management and facilitation of antimicrobial optimization, thus allowing for improved patient outcomes and reduced antimicrobial consumption. However, RDTs must still be combined with culture and susceptibility results to facilitate appropriate optimization of antimicrobial therapy, especially in the era of advanced multidrug resistance.3 This article provides a clinical overview of the multiplex polymerase chain reaction (PCR) RDTs, as well as 2 microbial cell-free DNA metagenomic sequencing technologies, used for suspected bacterial bloodstream and acute bacterial or viral respiratory infections. These tests are used in the United States, and have recent data that were published within the last 3 years.
Bloodstream RDTs
Bloodstream infections (BSIs) represent 40% of cases of community- and hospital-acquired sepsis and septic shock and approximately 20% of ICU-acquired cases.4 BSIs are associated with poor outcomes and require quick and adequate antimicrobial therapy and urgent source control. The gold standard for diagnosing BSIs is through blood cultures, which typically require 1 to 5 days for organism identification, followed by an additional 1 to 2 days for antibiotic susceptibility testing.5 Most patients are initiated on overly broad antibiotic therapy regimens that are based on patient presentation and risk factors for multidrug-resistant organisms. However, using RDTs can quickly optimize therapy and limit collateral damage from increased duration of broad-spectrum exposure, such as renal and hepatic toxicity, antibiotic resistance development, opportunistic infections, and mortality.5
In a systematic review and meta-analysis of 31 studies and 5,920 patients with BSIs, molecular RDT (mRDT) was associated with a decreased mortality risk and hospital length of stay (LOS), as well as improved time to effective therapy compared with conventional microbiological methods.6 The same researchers found that mortality risk decreased significantly with mRDT in the presence of an AMS program, confirming the need to pair diagnosis and AMS.
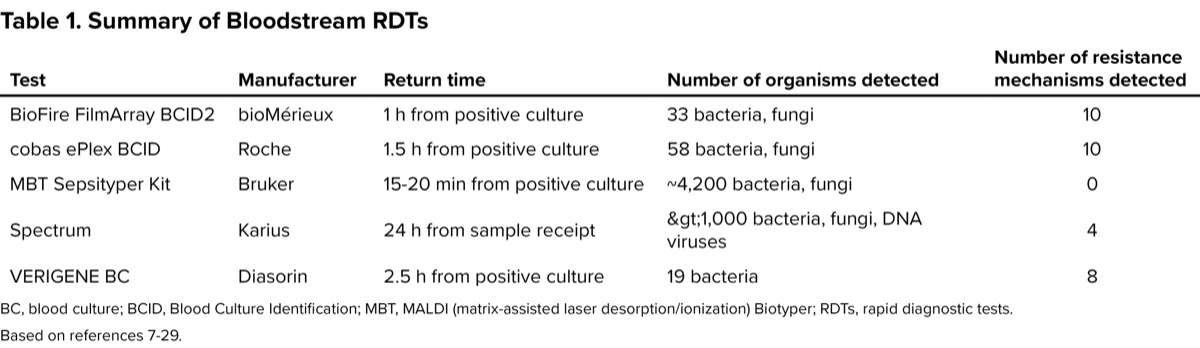
Although there are many advantages, disadvantages coexist with rapid blood culture assessment, such as limited panel sizes and reduced diagnostic quality in the presence of polymicrobial cultures. While these technologies can reduce overall hospital LOS and costs, they can be expensive to implement without the guidance of an AMS program, which allows patients to fully benefit from its advantages.5 There are 4 FDA-approved RDTs and 1 RDT not requiring FDA approval to detect organisms in the blood (Table 1).
| Table 1. Summary of Bloodstream RDTs | ||||
| Test | Manufacturer | Return time | Number of organisms detected | Number of resistance mechanisms detected |
|---|---|---|---|---|
| BioFire FilmArray BCID2 | bioMérieux | 1 h from positive culture | 33 bacteria, fungi | 10 |
| cobas ePlex BCID | Roche | 1.5 h from positive culture | 58 bacteria, fungi | 10 |
| MBT Sepsityper Kit | Bruker | 15-20 min from positive culture | ~4,200 bacteria, fungi | 0 |
| Spectrum | Karius | 24 h from sample receipt | >1,000 bacteria, fungi, DNA viruses, parasites | 4 |
| VERIGENE BC | Diasorin | 2.5 h from positive culture | 19 bacteria | 8 |
| BC, blood culture; BCID, Blood Culture Identification; MBT, MALDI (matrix-assisted laser desorption/ionization) Biotyper; RDTs, rapid diagnostic tests. Based on references 7-29. | ||||
PCR Tests
BioFire FilmArray BCID2 Panel
By extracting all nucleic acids and amplifying them by PCR techniques from positive blood cultures, the BioFire Blood Culture Identification 2 (BCID2) Panel (bioMérieux) can identify 43 targets within 1 hour.5,7,8 The BCID2 panel includes AMS program intervention, and has been shown to reduce the time to optimal therapy by almost 34 hours9 and hospital LOS, demonstrating more than $3,000 savings per patient tested.10 Time to identification of bacteria was also 14 hours faster with the BCID2 panel compared with traditional methods.11 A recent study examined the BCID2 panel results compared with culture results and found that 87.2% of the 117 positive cultures matched, with the concordance increasing to 98.1% when off-panel pathogen samples were removed.12 One study evaluated the performance of the BCID2 panel compared with 2 published diagnostic scoring tools for predicting ceftriaxone resistance in BSIs caused by Enterobacterales.13 They found ceftriaxone resistance in 41 of 356 (11.5%) isolates, which the BCID2 panel accurately predicted 99.2% of isolates, with a sensitivity of 92.7% and specificity of 100% (vs <90% for the 2 scoring tools; P<0.05). The authors concluded that the BCID2 panel outperformed both extended-spectrum beta-lactamase (ESBL) scoring tools.
cobas ePlex BCID Panels
The Roche cobas ePlex BCID Panels detect 20 gram-positive bacteria plus 4 resistance genes, 21 gram-negative bacilli plus 6 resistance genes, 15 fungal species, and 2 pan targets (gram-negative and Candida) using multiplex PCR (mPCR) technology from positive blood cultures within 1.5 hours.7,14-16 A benefit to this technology is the presence of pan-target probes, which help to identify gram-variable organisms or polymicrobial infections compared with other mRDT options.17 In one study, the ePlex BCID gram-positive panel reduced the time to pathogen identification by 32 hours and time to susceptibility by 70 hours compared with conventional methods.18 It also showed a decrease in total LOS (P<0.001) and infection-related LOS (P=0.001). Another study examined 88 positive body fluids from blood cultures and found a positive percent agreement of 96.5% and negative percent agreement of 99.8% compared with traditional cultures, showcasing the utility of the test in predicting what will grow in a culture.19
| Table 2. Summary of Respiratory RDTs | ||||
| Test | Manufacturer | Return time | Number of organisms detected | Number of resistance mechanisms detected |
|---|---|---|---|---|
| BioFire Respiratory 2.1 Panel | bioMérieux | ~45 min from positive culture | 22 bacteria, viruses | 0 |
| cobas ePlex RP2 | Roche | 1.5 h from positive culture | 24 bacteria, viruses | 0 |
| Focus | Karius | 24 h from sample receipt | >500 bacteria, fungi, DNA viruses, parasites | 0 |
| NxTAG Respiratory Pathogen Panel | Diasorin | ~3 h from positive culture | 21 bacteria, viruses | 0 |
| VERIGENE Respiratory Panel Flex Test | Diasorin | 2.5 h from positive culture | 16 bacteria, viruses | 0 |
| RDTs, rapid diagnostic tests; RP2, Respiratory Pathogen 2. Based on references 30-42. | ||||
MBT Sepsityper Kit
In 2020, the FDA cleared the matrix-assisted laser desorption/ionization (MALDI) Biotyper (MBT) Sepsityper Kit (Bruker). It uses mPCR technology on positive blood cultures to identify approximately 4,200 species of bacteria and yeast, including Candida auris, within 15 to 20 minutes, which is up to 48 hours faster than traditional methods.7,20 In patients with monomicrobial BSIs, the MBT Sepsityper Kit US IVD correctly identified 93.5% of microorganisms to the species level, with a higher identification rate in gram-negative bacilli (95.6%) compared with gram-positive cocci (93.3%) and yeast (75%).21 Compared with short subculturing (to get a pure culture for identification), the kit was approximately 98% accurate for gram-negative bacteria, approximately 85% for gram-positive bacteria, roughly 72% for anaerobes, and about 60% for yeast versus approximately 98%, 51%, 3%, and 7%, respectively, for the subculture.20
Microarray-Based Assay
VERIGENE Gram-Positive BC and Gram-Negative BC Tests
Diasorin distributes the VERIGENEa Gram-Positive and Gram-Negative BC tests to detect 19 bacteria and 8 antimicrobial resistance marker genes using magnetic beads to capture bacterial DNA into separate DNA microarrays within 2.5 hours.5,7,22,23 Compared with traditional methods, VERIGENE was able to provide results from positive blood cultures from pediatric patients within 2.9 hours compared with 44 hours for a time savings of almost 42 hours (P<0.0001).24 In adult patients, this technology has been shown to reduce time to positive bacterial identification by more than 24 hours.25 VERIGENE, when combined with AMS intervention,26 also significantly reduced the duration of inappropriate therapy and decreased the time to optimal therapy,25 especially in patients with ESBL-producing organisms.27
Cell-Free DNA Test
Karius Spectrum
The Karius Spectrum (Karius) is a laboratory-developed RDT that is both Clinical Laboratory Improvement Amendments (CLIA)-certified and accredited by the College of American Pathologists (CAP). It uses metagenomic sequencing of microbial cell-free DNA to detect more than 1,000 fungi, bacteria, and DNA viruses associated with deep-seated and difficult-to-diagnose systemic infections.28 The platform is also able to detect 4 different resistance mechanisms: 2 for gram-positive organisms (mecA, mecC, SCCmec, vanA, vanB) and 2 for gram-negative organisms (KPC, CTX-M).29 To take advantage of this testing platform, samples are required to be collected and sent to the Karius laboratory, with results returning within 24 hours after receipt of the sample. Despite being a blood test, the sample can detect very small quantities of microbial DNA from organisms infecting other parts of the body, including the lungs, liver, spleen, brain, heart, and bones and joints.28,29 Data published within the last 2 years have focused on the use of this technology in pediatric, immunocompromised, or rare/unique organism populations.29
Respiratory RDTs
Even though acute respiratory infections are more likely to be caused by a virus, differentiating between viral and bacterial infections can be a challenge. Without the assistance of RDTs to detect viral respiratory infections, they are rarely implicated in hospitalized patients, leading to antibiotic overuse.30 Determining an active viral infection not only helps reduce the unnecessary use of antibiotics but also alleviates the increased risk for an adverse drug event, new-onset organ dysfunction, or Clostridioides difficile infection. There are 4 major FDA-approved respiratory mPCR assays: NxTAG (Diasorin), BioFire FilmArray (bioMérieux), cobas ePlex (Roche), and VERIGENE (Diasorin). There is 1 CLIA-approved, CAP-accredited test: Karius Focus (Karius).
PCR Tests
BioFire Respiratory 2.1 Panel
The BioFire Respiratory 2.1 Panel (bioMérieux), FDA cleared in 2011, is a low-throughput, moderate- to low-complexity, multiplex nested PCR assay automated from extraction to results. The panel can identify 22 targets (18 viruses, including COVID-19, and 4 bacteria) with a run time of about 45 minutes. A nasopharyngeal swab in transport media or saline can detect specimens with 97.1% sensitivity and 99.3% specificity.31 A prospective twin-center cohort study found that combining the BioFire FilmArray system and procalcitonin test is a useful strategy for early detection of potential viral epidemics and assisting antibiotic stewardship efforts.32 The mPCR respiratory panel and procalcitonin test can help guide decisions on withholding antibiotics or de-escalating antibiotics.
cobas ePlex RP2
Roche’s cobas ePlex Respiratory Pathogen 2 Panel can identify 21 viral and 3 bacterial respiratory pathogens within a single cartridge.33 The ePlex system uses innovative technology to detect the organisms. First, the target DNA is mixed with the signal probe solution, where the signal probe hybridizes to the DNA. Next, the solution travels through a microfluidic chamber where the hybridized target completes the reaction with a preassembled capture probe. Finally, voltage is swept across each electrode, which allows the target DNA to be analyzed by electrochemical detection.34 This whole process takes about 90 minutes to complete.35 In a study by Nijhuis et al, a total of 343 clinical specimens were assessed.35 The RPP showed an agreement of 97.4% with the real-time PCR assay. The study concluded that the ePlex system performed well compared with real-time PCR assays for the detection of respiratory pathogens in a laboratory or decentralized setting and provided a large amount of diagnostic data quickly with minimal hands-on time. The ePlex RP2 also decreased emergency department admissions by 8.4%,36 was able to more quickly identify patients with coinfections who were also at risk for severe disease,37 and verified that 41% of antibiotic prescriptions for respiratory tract infections were inappropriate.38
NxTAG Respiratory Pathogen Panel v2
The NxTAG Respiratory Pathogen Panel (RPP; Diasorin) is a high-complexity, high-throughput, bead-based qualitative PCR assay that is capable of detecting 21 respiratory pathogens simultaneously.30 It is intended for the identification of nucleic acids from multiple respiratory viruses and bacteria extracted from upper respiratory tract specimens collected from patients with clinical signs and symptoms of a respiratory tract infection.39 The panel can run up to 96 samples in a single batch, with results available in less than 3 hours post-extraction. The NxTAG RPP was found to have an overall diagnostic sensitivity of 98.9% and specificity of 99.0%. Compared with the BioFire FilmArray and real-time PCR, it was the only multiplex assay that could detect human bocavirus, one of the causative agents of pediatric acute respiratory tract infections.40
VERIGENE Respiratory Pathogens Flex Test
The VERIGENE Respiratory Pathogens Flex Test is a moderate-complexity, customizable-throughput assay that uses reverse transcription, PCR, and microarray hybridization to detect gene sequences of 13 viruses and 3 bacteria.30 The cost-effective RP Flex is the first multiplex respiratory test that allows the user to pay for their reporting preference.28 In a study by Hwang et al, the authors compared the NxTAG and the VERIGENE for their ability to detect common targets like influenza A, influenza B, and respiratory syncytial virus in the respiratory specimens of hospitalized patients.41 A high degree of agreement was reported in detecting the presence or absence of respiratory viruses between the 2 panels. The NxTAG panel has a longer time requirement for results and more hands-on time of technicians but can analyze a broader range of respiratory viruses. The NxTAG panel would be useful in larger hospitals or laboratories requiring high-throughput assays and with experience in molecular testing. The VERIGENE panel may be useful in smaller hospitals or laboratories with fewer technicians, less experience in molecular testing, or in hospitals with outpatient settings.41
Cell-Free DNA Test
Karius Focus | BAL
Karius developed the Focus | BAL metagenomic sequencing of cell-free DNA to detect microorganisms in less than 1 mL of a bronchoalveolar lavage sample.42 This test is ideal to use for patients in whom a bronchoscopy is already scheduled to occur, are also immunocompromised, and are suspected of having a lung infection. It touts more than a 50% increased diagnostic yield compared with the standard-of-care methodology and can detect more than 350 bacteria, 15 DNA viruses, 115 fungi, and 30 parasites. Like the Karius Spectrum test, clinical data are primarily in pediatric patients, immunocompromised patients, and in patients with rare/unique organisms.29
Discussion
mPCR assays have been a game changer for rapidly identifying infectious pathogens. They are highly efficient, have rapid turnaround times and throughput, and can screen for multiple targets. The use of RDTs supports timely clinical decisions, leading to improved patient outcomes, decreased hospital LOS, prevention of unnecessary antibiotic use, and decreased disease transmission. When combined with AMS efforts, RDTs can further enhance the quality and impact of infectious disease management. A recent network meta-analysis demonstrated a significant mortality benefit associated with the use of RDTs and AMS programs compared with blood cultures alone (odds ratio [OR], 0.72; 95% CI, 0.59-0.87) and with the use of RDTs and AMS programs versus blood cultures and AMS programs (OR, 0.78; 95% CI, 0.63-0.96). However, neither RDTs alone nor blood cultures and AMS programs benefited survival compared with blood cultures alone.43 Ultimately, while the currently available RDTs have become the new standard of care for diagnosing infections, further advancements are still needed and on the horizon, including metagenomic next-generation sequencing and nanopore sequencing.44
References
- Morgan DJ, Malani P, Diekema DJ. Diagnostic stewardship—leveraging the laboratory to improve antimicrobial use. JAMA. 2017;318(7):607-608.
- Fabre V, Davis A, Diekema DJ, et al. Principles of diagnostic stewardship: a practical guide from the Society for Healthcare Epidemiology of America Diagnostic Stewardship Task Force. Infect Control Hosp Epidemiol. 2023;44(2):178-185.
- Peri AM, Chatfield MD, Ling W, et al. Rapid diagnostic tests and antimicrobial stewardship programs for the management of bloodstream infection: what is their relative contribution to improving clinical outcomes? A systematic review and network meta-analysis. Clin Infect Dis. 2024;79(2):502-515.
- Briggs N, Campbell S, Gupta S. Advances in rapid diagnostics for bloodstream infections. Diagn Microbiol Infect Dis. 2021;99(1):115219.
- Timsit JF, Ruppé E, Barbier F, et al. Bloodstream infections in critically ill patients: an expert statement. Intensive Care Med. 2020;46(2):266-284.
- Timbrook TT, Morton JB, McConeghy KW, et al. The effect of molecular rapid diagnostic testing on clinical outcomes in bloodstream infections: a systematic review and meta-analysis. Clin Infect Dis. 2017;64(1):15-23.
- Tjandra KC, Ram-Mohan N, Abe R, et al. Diagnosis of bloodstream infections: an evolution of technologies towards accurate and rapid identification and antibiotic susceptibility testing. Antibiotics. 2022;11(4):511.
- BIOFIRE Blood Culture Identification 2 (BCID2) Panel. bioMérieux. Accessed July 1, 2025. biofiredx.com/products/the-filmarray-panels/filmarraybcid/
- Messacar K, Hurst AL, Child J, et al. Clinical impact and provider acceptability of real-time antimicrobial stewardship decision support for rapid diagnostics in children with positive blood culture results. J Pediatr Infect Dis Soc. 2017;6(3):267-274.
- Pardo J, Klinker KP, Borgert SJ, et al. Clinical and economic impact of antimicrobial stewardship interventions with the FilmArray blood culture identification panel. Diagn Microbiol Infect Dis. 2016;84(2):159-164.
- Sparks R, Balgahom R, Janto C, et al. Evaluation of the BioFire Blood Culture Identification 2 panel and impact on patient management and antimicrobial stewardship. Pathology. 2021;53(7):889-895.
- Kim KJ, Yun SG, Cho Y, et al. Rapid direct identification of microbial pathogens and antimicrobial resistance genes in positive blood cultures using a fully automated multiplex PCR assay. J Korean Med Sci. 2024;39(17):e157.
- Andrews SR, Timbrook TT, Fisher MA, et al. Comparative analysis of a rapid diagnostic test and scoring tools for ESBL detection in Enterobacterales bloodstream infections for optimizing antimicrobial therapy. Microbiol Spectr. 2024;12(1):e03131-23.
- Young BA, Hanson KE, Gomez CA. Molecular diagnostic advances in transplant infectious diseases. Curr Infect Dis Rep. 2019;21(12):52.
- Tansarli GS, Chapin KC. A closer look at the laboratory impact of utilizing ePlex blood culture identification panels: a workflow analysis using rapid molecular detection for positive blood cultures. Microbiol Spectr. 2022;10(5):e0179622.
- cobas ePlex System. Roche. Accessed July 1, 2025. diagnostics.roche.com/global/en/products/systems/cobas-eplex-system-sys-447.html
- Claeys KC, Hopkins TL, Schlaffer K, et al. Comparing the clinical utility of rapid diagnostics for treatment of bloodstream infections using desirability of outcome ranking approach for the management of antibiotic therapy (DOOR-MAT). Antimicrob Agents Chemother. 2021;65(9):e00441-21.
- Kiszla BM, McCarty T, White C, et al. The impact of GenMark Dx ePlex blood-culture identification on the treatment and outcomes of gram-positive bacteremia. Antimicrob Steward Health Epidemiol. 2022;2(suppl S1):s44.
- Cox CR, Weghorn KN, Ruger K, et al. Clinical utility of multiplex PCR in the detection of pathogens from sterile body fluids. Bacteriology. 2024;62(4):e01611-23.
- Positive blood culture analysis. Bruker. Accessed July 1, 2025. bruker.com/en/products-and-solutions/microbiology-and-diagnostics/microbial-identification/positive-blood-culture-analysis.html
- Lin HH, Tseng KH, Tien N, et al. Evaluation of the Rapid Sepsityper protocol and specific MBT-Sepsityper® module for the identification of bacteremia and fungemia using Bruker Biotyper MALDI-TOF MS. J Microbiol Immune Infect. 2022;55(6 pt 2):1330-1333.
- Freiberg JA, Deri CR, Nesbit WJ, et al. Identification and antibiotic susceptibility patterns of clinical blood culture isolates not identified by a rapid microarray diagnostic system. Microbiol Spectr. 2021;9(1):e00175-21.
- The VERIGENE System. Diasorin. Accessed July 1, 2025. us.diasorin.com/en/molecular-diagnostics/tools/verigene-system
- Howard AK, Claeys K, Biggs JM, et al. Performance of Verigene rapid diagnostic testing for detection of inpatient pediatric bacteremia. J Ped Pharmacol Ther. 2021;26(5):472-477.
- Belknap A, Grosser DS, Hale DA, et al. Clinical uptake of antimicrobial stewardship recommendations following Nanosphere Verigene Blood Culture Gram-negative reporting. Proc (Bayl Univ Med Cent). 2017;30(4):395-399.
- Walker T, Dumadag S, Lee CJ, et al. Clinical impact of laboratory implementation of Verigene BC-GN microarray-based assay for detection of gram-negative bacteria in positive blood cultures. J Clin Microbiol. 2016;54(7):1789-1796.
- Swingler EA, Clark M, Moore SE, et al. Effect of a pharmacy-driven rapid bacteremia response program on outcomes in adult patients with extended-spectrum beta-lactamase bacteremia: a retrospective, quasi-experimental study. J Am Coll Clin Pharm. 2025;8(1):20-27.
- Karius Spectrum. Karius, Inc. Accessed July 1, 2025. kariusdx.com/our-solution/karius-spectrum
- Karius Spectrum. Liquid biopsy for infectious diseases. Karius, Inc. Accessed July 1, 2025. karius.showpad.com/share/f8HLLMjsWmfzuH2IH10J7
- Diaz-Decaro JD, Green NM, Godwin HA. Critical evaluation of FDA-approved respiratory multiplex assays for public health surveillance. Expert Rev Mol Diagn. 2018;18(7):631-643.
- BIOFIRE Respiratory 2.1 (RP2.1) Panel. bioMérieux. Accessed July 1, 2025. biomerieux.com/us/en/our-offer/clinical-products/biofire-respiratory-panels.html
- Lee CC, Chang JCY, Mao XW, et al. Combining procalcitonin and rapid multiplex respiratory virus testing for antibiotic stewardship in older adult patients with severe acute respiratory infection. J Am Med Dir Assoc. 2020;21(1):62-67.
- cobas ePlex System. Roche. Accessed July 1, 2025. diagnostics.roche.com/global/en/products/systems/cobas-eplex-system-sys-447.html
- Multiplex testing technology. Roche. Accessed July 1, 2025. diagnostics.roche.com/us/en/article-listing/innovations/esensor-technology.html
- Nijhuis RHT, Guerendiain D, Claas ECJ, et al. Comparison of ePlex Respiratory Pathogen Panel with laboratory-developed real-time PCR assays for detection of respiratory pathogens. J Clin Microbiol. 2017;55(6):1938-1945.
- Weiss ZF, Cunha CB, Chambers AB, et al. Opportunities revealed for antimicrobial stewardship and clinical practice with implementation of a rapid respiratory multiplex assay. J Clin Microbiol. 2019;57(10):e00861-19.
- Hanson KE, Azar MM, Chou A, et al. Molecular testing for acute respiratory tract infections: clinical and diagnostic recommendations from the IDSA’s diagnostics committee. Clin Infect Dis. 2020;71(10):2744-2751.
- Havers FP, Hicks LA, Chung JR, et al. Outpatient antibiotic prescribing for acute respiratory infections during influenza seasons. JAMA Netw Open. 2018;1(2):e180243.
- NxTAG Respiratory Pathogen Panel v2. Diasorin. Accessed July 1, 2025. us.diasorin.com/en/molecular-diagnostics/kits-reagents/nxtag-respiratory-pathogen-panel-V2
- Chen JHK, Lam HY, Yip CCY, et al. Clinical evaluation of the new high-throughput Luminex NxTAG respiratory pathogen panel assay for multiplex respiratory pathogen detection. J Clin Microbiol. 2016;54(7):1820-1825.
- Hwang SM, Lim MS, Han M, et al. Comparison of xTAG respiratory virus panel and Verigene Respiratory Virus Plus for detecting influenza virus and respiratory syncytial virus. J Clin Lab Anal. 2015;29(2):116-121.
- Karius Focus | BAL. Karius, Inc. Accessed July 1, 2025. karius.showpad.com/share/jksbzbr63Na3kVqZmRcjJ
- Liborio MP, Harris PNA, Ravi C, et al. Getting up to speed: rapid pathogen and antimicrobial resistance diagnostics in sepsis. Microorganisms. 2024;12(9):1824.
- Han D, Yu F, Zhang D, et al. Molecular rapid diagnostic testing for bloodstream infections: nanopore targeted sequencing with pathogen-specific primers. J Infect. 2024;88(6):106166.
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Common Bloodstream and Respiratory RDTs to Optimize Therapy Management
Common Bloodstream and Respiratory RDTs to Optimize Therapy Management