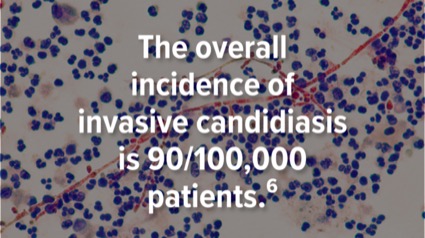
Candida spp are yeast that are found commonly on the skin and in the oropharyngeal cavity, gastrointestinal tract, and genitourinary tract.1,2 Although a normal part of the human flora, invasive candidiasis and candidemia are associated with mortality rates as high as 70% in the absence of adequate treatment.3-5 The overall incidence of invasive candidiasis in the United States has been estimated at 90 cases per 100,000 patients, due to increasing populations with risk factors, such as the need for central venous catheters, broad-spectrum antibiotic use, receipt of total parenteral nutrition, intraabdominal procedures, and/or immunocompromising conditions.3,6-8 Although Candida albicans is responsible for most infections, there is an evident epidemiological shift in Candida spp from albicans to non-albicans type and the emergence of C. auris—all which affect therapeutic decision-making.6,9 Importantly, Candida spp possess multiple virulence factors and are capable of forming biofilms that contribute to their observed persistence in deep-seated infections.10 These characteristics often necessitate prolonged antifungal therapy and, in some cases, lifelong suppressive therapy.1,11
While the development of novel antifungal classes is in progress, clinical management of invasive fungal infections continues to rely primarily on 3 major classes of antifungal agents: polyenes, azoles, and the echinocandins.12,13 Amphotericin B, a systemic polyene, and many azole antifungals offer broad-spectrum activity against both yeasts and molds.13 Traditional echinocandins, including caspofungin, anidulafungin, and micafungin, are semisynthetic compounds that are highly effective against both azole-susceptible and -resistant strains of Candida, although they generally lack efficacious activity against molds and require daily intravenous administration.12,14-15
More recently, rezafungin (Rezzayo, Melinta Therapeutics), a next-generation echinocandin, was developed by modifying the structure of anidulafungin to enhance its chemical stability and significantly prolong its half-life. Specifically, the pharmacokinetic and pharmacodynamic properties have been improved to maintain therapeutically effective levels against susceptible isolates for up to 1 week after doses.16 In addition to its potent in vitro activity against Candida. (Table 1), rezafungin has demonstrated in vitro activity against Aspergillus spp and Pneumocystis jirovecii, which may offer potential utility beyond invasive candidiasis, although clinical efficacy for these pathogens has not been established fully.14,17 In March 2023, the FDA granted limited-use approval to rezafungin for the treatment of invasive candidiasis and candidemia in adult patients with limited or no alternative treatment options. Under current package labeling, it is administered as a 400-mg IV loading dose, followed by 200 mg once weekly.18
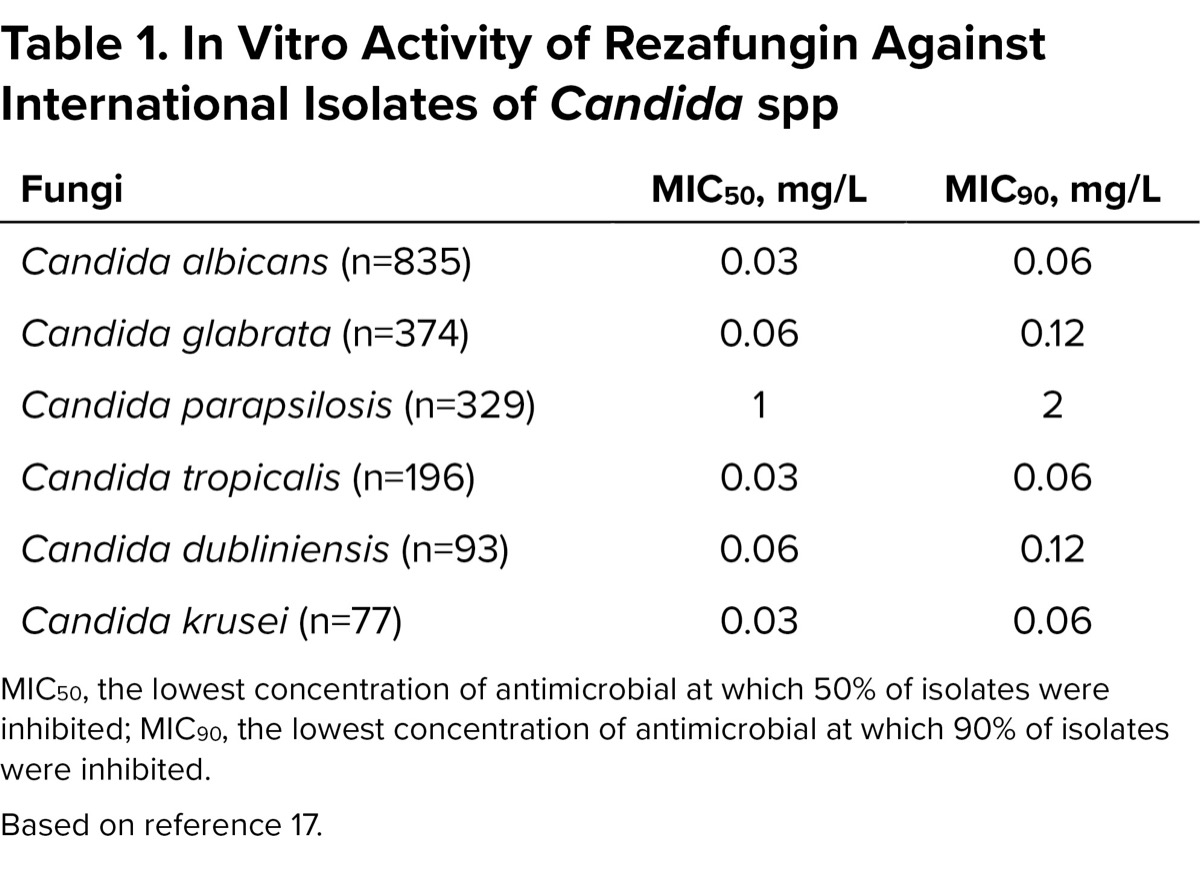
| Table 1. In Vitro Activity of Rezafungin Against International Isolates of Candida spp | ||
| Fungi | MIC50, mg/L | MIC90, mg/L |
|---|---|---|
| Candida albicans (n=835) | 0.03 | 0.06 |
| Candida glabrata (n=374) | 0.06 | 0.12 |
| Candida parapsilosis (n=329) | 1 | 2 |
| Candida tropicalis (n=196) | 0.03 | 0.06 |
| Candida dubliniensis (n=93) | 0.06 | 0.12 |
| Candida krusei (n=77) | 0.03 | 0.06 |
| MIC50, the lowest concentration of antimicrobial at which 50% of isolates were inhibited; MIC90, the lowest concentration of antimicrobial at which 90% of isolates were inhibited. Based on reference 17. | ||
Given the increasing burden of invasive candidiasis and the limitations of existing antifungal therapy—such as antifungal resistance, drug–drug interactions, corrected QT interval (QTc) prolongation, nephrotoxicity, and/or the need for daily parenteral administration—there is an urgent need for antifungal agents with similar efficacy, improved safety, and logistical practicality.13 Rezafungin’s activity against Candida spp, once-weekly dosing schedule, and favorable pharmacokinetic profile make it particularly well suited for patients in whom daily parenteral therapy is not optimal, such as those transitioning to discharge and/or with limited venous access.14,17,18 Additionally, its low potential for drug–drug interactions—including with frequently used immunosuppressant medications—and absence of QTc prolongation risk position it as an attractive option in complex, medically comorbid populations.18 These characteristics, combined with potential healthcare cost savings and decreased need for central venous catheter placement, underscore rezafungin’s potential to address several key clinical and operational challenges in the management of invasive fungal infections.19 Therefore, the primary objective of this review is to describe the pharmacokinetics and current clinical data for rezafungin, with a focus on invasive candidiasis and candidemia.
Pharmacokinetics
Similar to other echinocandins, rezafungin has low oral bioavailability and is only available parenterally for IV infusion.16,18 Rezafungin also does not penetrate the central nervous system or vitreous humor, which is in line with the other agents in this class. The drug has a volume of distribution of 67 L (±28 L), and is highly protein-bound (>85%) (Table 2).18,20,21
| Table 2. Pharmacokinetic Parameters of Rezafungin | |
| Absorption | Cmax ~ 19.2 mg/L (day 1) Cmax ~ 11.8 mg/L (day 15) |
| Distribution | Vd, 67 L >85% protein-bound |
| Metabolism | Hydroxylation (non-hepatic) |
| Elimination | Half-life ~ 152 h 74.3% in feces (unchanged) 25.7% in urine (inactive metabolites) |
| Cmax, maximum observed concentration; Vd, volume of distribution. | |
Because of the structural differences compared with the other echinocandins, rezafungin has a significantly longer half-life (152±29 hours), allowing for once-weekly dosing.18,22,23 The principal pharmacokinetic/pharmacodynamic index best predicted to optimize activity for rezafungin is the area under the curve0-168/minimum inhibitory concentration (AUC0-168/MIC), and it has been shown that rezafungin at approved doses has a high probability of target attainment against a range of observed MIC distributions against most Candida spp, including C. auris and C. glabrata.21 Rezafungin undergoes non-hepatic metabolism via hydroxylation and is eliminated primarily in the feces (74.3%) as active drug, and the urine (25.7%) as inactive metabolites. Rezafungin has not been shown to be a substrate of any major cytochrome CYP450 enzymes and thus, has minimal drug–drug interactions.18 Because of these optimized properties, rezafungin may have a role in reducing catheter overuse and allow for faster transitions of care to the outpatient setting, as well as acting as an alternative agent when azoles cannot be used due to antifungal resistance or drug–drug interactions.18,23
The STRIVE Trial
The STRIVE trial was a phase 2, multicenter, double-blind, randomized controlled trial conducted between July 2016 and April 2019, to compare the efficacy and safety of once-weekly IV rezafungin with daily IV caspofungin to treat invasive candidiasis and/or candidemia.24 Of note, a strong focus of this study was to determine the optimal dosing regimen to be used in phase 3 trials. Adults with systemic signs and mycologic confirmation of invasive candidiasis and/or candidemia were randomized to receive rezafungin as an IV loading dose of 400 mg followed by weekly IV doses of 400 mg (RZF 400/400 arm), rezafungin as an IV loading dose of 400 mg followed by weekly IV doses of 200 mg (RZF 400/200 arm), or caspofungin as an IV loading dose of 70 mg followed by a daily IV dose of 50 mg (CAS 70/50) for at least 2 weeks and up to 4 weeks. Patients in the caspofungin arm were permitted to transition to fluconazole as oral stepdown therapy based on predefined stability criteria.24
The primary end point was overall cure at day 14, which was defined as resolution of signs and symptoms and mycologic eradication; another efficacy assessment was 30-day all-cause mortality.24 There were 207 patients in the control and randomized (RZF 400/400, 81 patients; RZF 400/200, 57 patients; CAS 70/50, 69 patients). Baseline demographics were well balanced between the groups and the median (range) duration of therapy in each of the 3 groups was 14 days (1-28 days). The most frequently isolated pathogens included C. albicans (49.7%), C. glabrata (20.2%), and C. parapsilosis (15.3%). The overall cure rate was highest in the RZF 400/200 arm at 76.1% (RZF 400/400, 60.5%; CAS 70/50, 67.2%). Likewise, there were fewer patients in the RZF 400/200 arm for 30-day all-cause mortality at 4.4% than in the other arms (RZF 400/400, 15.8%; CAS 70/50, 13.1%).
No major safety events were observed.24 There is a potential that patients in the rezafungin 400/400 arm may have experienced a paradoxical effect phenomenon associated with Candida, in which attenuation of activity occurs with higher concentrations of echinocandins.25 However, it is important to note that divergence in overall cure between the 2 rezafungin arms was already observed by day 5, when all patients in the rezafungin groups had received the same, initial 400-mg loading dose (RZF 400/400, 55.3%; RZF 400/200, 73.9%).24
The ReSTORE and ReSPECT Trials
Following the completion of the STRIVE trial, the ReSTORE trial was conducted between October 2018 and August 2021, which was a phase 3, multicenter, double-blind, double-dummy, randomized controlled trial to compare the safety and efficacy of rezafungin and caspofungin in patients with invasive candidiasis and/or candidemia.24,26 Adult patients with systemic signs and mycologic confirmation of invasive candidiasis and/or candidemia were randomized in a 1:1 ratio to receive rezafungin as a 400-mg IV loading dose followed by 200 mg IV weekly for 2 to 4 doses or caspofungin as a 70-mg IV loading dose followed by 50 mg IV daily for up to 4 weeks. Patients in the caspofungin arm were permitted an optional stepdown therapy transition to fluconazole. A noninferiority margin of 20% was set and assessed in the modified intention-to-treat population.26 The primary end points were 30-day all-cause mortality and global cure, which was defined as clinical cure, mycologic eradication, and radiological cure.26
There were 187 patients in the modified intention-to-treat analysis (rezafungin, 93; caspofungin, 94) and baseline characteristics were similar in the 2 groups. The most common pathogen isolated was C. albicans, and more than 99% of all isolates were susceptible to rezafungin and caspofungin. Analysis found no differences in 30-day all-cause mortality (rezafungin, 23.7%; caspofungin, 21.3% [treatment difference, 2.4%; 95% CI,
It is important to note that the methodology of ReSTORE has been subject to criticism, primarily due to the relatively wide noninferiority margin of 20%.26,27 This margin is particularly notable given the high mortality rates associated with invasive fungal infections.3-5,26,27 Of note, the FDA’s decision to grant a limited-use indication was influenced by this broader margin, as a more conventional 10% noninferiority threshold could have supported a full indication without such restrictions.28
Lastly, the ongoing ReSPECT trial is an international, phase 3, multicenter, double-blind, randomized controlled trial evaluating the safety and efficacy of once-weekly rezafungin versus fluconazole or posaconazole and trimethoprim-sulfamethoxazole for the prevention of invasive fungal infections (such as Candida, Aspergillus, or Pneumocystis) in patients undergoing allogeneic hematopoietic stem cell transplantation.29 Although results are not available since the trial is still recruiting, study investigators plan to assess the primary outcome of fungal-free survival in approximately 450 patients across approximately 30 centers.29
Conclusion
Rezafungin represents a promising advancement in antifungal therapy, offering a favorable safety and efficacy profile for the treatment of candidemia and invasive candidiasis.14,24,26 Its once-weekly dosing and potent activity against a broad spectrum of fungal pathogens—including azole-resistant Candida—make it particularly suited for patients requiring prolonged antifungal treatment or those for whom daily parenteral therapy is impractical.14,17 Additionally, its minimal drug–drug interactions and reduced risk for QTc prolongation present advantages for patients who are poor candidates for azoles.13,14 Although more data are needed, rezafungin as sequential therapy may help streamline outpatient care, minimize the need for central venous catheters, and reduce hospital length of stay. Ongoing studies are assessing its role in prophylaxis in immunocompromised individuals.29 Although newly approved medications often incur higher up-front costs, economic analyses suggest that rezafungin may be cost-saving overall.19 Collectively, these attributes support rezafungin’s value as a novel and practical addition to the antifungal armamentarium, particularly in the context of rising antifungal resistance, complex patient needs, and enhancing logistical practicality.
References
- Talapko J, Juzbašic M, Matijevic T, et al. Candida albicans-the virulence factors and clinical manifestations of infection. J Fungi (Basel). 2021;7(2):79.
- Spaminato C, Leonardi D. Candida infections, causes, targets, and resistance mechanism: traditional and alternative antifungal agents. Biomed Res Int. 2013;2013:204237.
- Salmanton-García J, Cornely OA, Stemler J, et al. Attributable mortality of candidemia-results from the ECMM Candida III multinational European Observational Cohort Study. J Infect. 2024;89(3):106229.
- Andes DR, Safdar N, Baddley JW, et al. The epidemiology and outcomes of invasive Candida infections among organ transplant recipients in the United States: results of the Transplant-Associated Infection Surveillance Network (TRANSNET). Transpl Infect Dis. 2016;18(6):921-931.
- Pappas PG, Lionakis MS, Arendrup MC, et al. Invasive candidiasis. Nat Rev Dis Primers. 2018;4:18026.
- Ricotta EE, Lai YL, Babiker A, et al. Invasive candidiasis species distribution and trends, United States, 2009-2017. J Infect Dis. 2021;223(7):1295-1302.
- Wenzel RP. Nosocomial candidemia: risk factors and attributable mortality. Clin Infect Dis. 1995;20(6):1531-1534.
- Poissy J, Damonti L, Bignon A, et al. Risk factors for candidemia: a prospective matched case-control study. Crit Care. 2020;24(1):109.
- Quindós G, Marcos-Arias C, San-Millán R, et al. The continuous changes in the aetiology and epidemiology of invasive candidiasis: from familiar Candida albicans to multiresistant Candida auris. Int Microbiol. 2018;21(3):107-119.
- Malinovska Z, Conkova E, Váczi P. Biofilm formation in medically important Candida species. J Fungi (Basel). 2023;9(10):955.
- Pappas PG, Kauffman CA, Andes DR, et al. Clinical practice guidelines for the management of candidiasis: 2016 updated by the Infectious Diseases Society of America. Clin Infect Dis. 2016;62(4):e1-50.
- Vanreppelen G, Wuyts J, Dijck PV, et al. Sources of antifungal drugs. J Fungi (Basel). 2023;9(2):171.
- Nett JE, Andes DR. Antifungal agents: spectrum of activity, pharmacology, and clinical implications. Infect Dis Clin North Am. 2016;30(1):51-83.
- Garcia-Effron G. Rezafungin—mechanisms of action, susceptibility and resistance: similarities and differences with the other echinocandins. J Fungi (Basel). 2020;6(4):262.
- Morris MI, Villmann M. Echinocandins in the management of invasive fungal infections, part 1. Am J Health Syst Pharm. 2006;63(18):1693-1703.
- Symanki M, Chmielewska S, Czyzewska E, et al. Echinocandins-structure, mechanism of action and use in antifungal therapy. J Enzyme Inhib Med Chem. 2022;37(1):876-894.
- Pfaller MA, Carvalhaes C, Messer SA, et al. Activity of a long-acting echinocandin, rezafungin, and comparator antifungal agents tested against contemporary invasive fungal isolates (SENTRY Program, 2016 to 2018). Antimicrob Agents Chemother. 2020;64(4):e00099-20.
- Rezafungin. Package insert. Melinta Therapeutics; 2023. Accessed July 22, 2025. bit.ly/4f0Henc-IDSE
- Jeck J, Jakobs F, Kurte MS, et al. Health-economic modelling of cost savings due to the use of rezafungin based on a German cost-of-illness study of candidiasis. JAC-Antimicrobial Resistance. 2023;5(3):dlad079.
- Smith HL, Bensman TJ, Mishra S, et al. Regulatory considerations in the approval of rezafungin (Rezzayo) for the treatment of candidemia and invasive candidiasis in adults. J Infect Dis. 2024;230(2):505-513.
- Roepcke S, Passarell J, Walker H, et al. Population pharmacokinetic modeling and target attainment analyses of rezafungin for the treatment of candidemia and invasive candidiasis. Antimicrob Agents Chemother. 2023;67(12):e0091623.
- Forrister NM, McCarty TP, Pappas PG. New perspectives on antimicrobial agents: rezafungin. Antimicrob Agents Chemother. 2024;69(1):e00646-23.
- Bassetti M, Stewart A, Bartalucci C, et al. Rezafungin acetate for the treatment of candidemia and invasive candidiasis: a pharmacokinetic evaluation. Expert Opin Drug Metab Toxicol. 2025;21(2):125-132.
- Thompson GR, Soriano A, Skoutelis A, et al. Rezafungin versus caspofungin in a phase 2, randomized, double-blind study for the treatment of candidemia and invasive candidiasis: the STRIVE trial. Clin Infect Dis. 2021;73(11):e3647-e3655.
- Weiderhold NP. Paradoxical echinocandin activity: a limited in vitro phenomenon? Med Mycol. 2009;47(suppl):1:S369-S375.
- Thompson GR, Soriano A, Cornely OA, et al. Rezafungin versus caspofungin for treatment of candidaemia and invasive candidiasis (ReSTORE): a multicentre, double-blind, double-dummy, randomised phase 3 trial. Lancet. 2023;401(10370):49-59.
- Pong S, Urner M, Fowler RA, et al. Testing for non-inferiority mortality: a systematic review of non-inferiority margin sizes and trial characteristics. BMJ Open. 2021;11(4):e044480.
- FDA Briefing Document. FDA; 2023. fda.gov/media/164666/download
- Cidara Therapeutics doses first patient in pivotal phase 3 ReSPECT trial of rezafungin for prevention of invasive fungal disease in patients undergoing allogeneic blood and marrow transplantation. Cidara Therapeutics. May 20, 2020. Accessed April 20, 2025. bit.ly/4kysKwb-IDSE
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() A Week in the Life of Rezafungin: Advancements in Long-Acting Antifungal Therapy for Invasive Candidiasis
A Week in the Life of Rezafungin: Advancements in Long-Acting Antifungal Therapy for Invasive Candidiasis