How do you treat patients with severe mpox? Although we know that those who are immunocompromised or people living with HIV are at higher risk for severe disease, there continues to be no FDA-approved mpox therapy, three years after the 2022 global mpox outbreak.
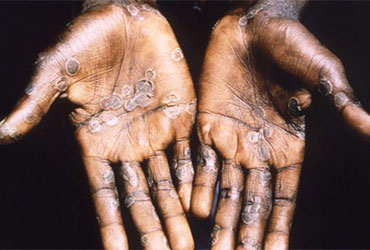
However, investigational treatments are available. Timmy Do, PharmD, BCPS, BCIDP, a clinical pharmacist on the Centralized Antimicrobial Stewardship Team, talked to Infectious Disease Special Edition about two people treated at his hospital, AdventHealth in Orlando, Florida. Both patients treated by Dr. Do and his colleague Monica Cozad, PharmD, BCIDP, also a clinical pharmacist on the Centralized Antimicrobial Stewardship Team, had uncontrolled HIV, were not compliant with antiretroviral therapy (ART), and had previously been infected with mpox. Consults with the CDC and research were key to treating these patients, Dr. Do told IDSE.
A Tale of 2 Cases
Patient 1
The first patient was a man in his 20s, who presented with worsening mpox lesions as well as fever and myalgias. His initial lab results showed his CD4+ count was 18 cells/mcg and his viral load 1,640 copies/mL. Dr. Cozad started him on broad-spectrum antibiotics and ART, as well as consulted with the hospital’s infectious disease department and the CDC. He also began oral tecovirimat (Tpoxx, SIGA Technologies) while waiting for an IV formulation. By day 6, he had started on IV tecovirimat and brincidofovir (Tembexa, Chimerix), and received one dose of vaccinia immunoglobulin (VIGIV). He also switched to IV tecovirimat. That was the day of his last recorded fever.
On day 12, he had his second dose of brincidofovir; however, his lesions became more hemorrhagic and confluent, which led the providers to suspect immune reconstitution inflammatory syndrome (IRIS). By day 17, his pain was better controlled, and his lesions were healing with no new lesions appearing. But one lesion on his knee worsened, and the providers suspected a superimposed skin and soft tissue infection. On day 20, he switched back to oral tecovirimat and continued the medication until discharge. On day 30, he was administered his third dose of brincidofovir. With no new lesions and better HIV markers—CD4+ at 47 cells/mcg and viral load at 47 copies/mL—the patients was discharged at day 34.
Patient 2
The second patient was a man in his 40s who had worsening mpox lesions. His lab results revealed a CD4+ count of 67 cells/mcg and viral load of more than 1 million copies/mL. Dr. Do started him on broad-spectrum antibiotics and oral tecovirimat. The providers consulted with the ID department and the CDC. On day 8, he began receiving ART, and on day 9, he received his first dose of IV cidofovir. On day 12, he started twice-daily topical cidofovir (topical was chosen due to his wound size), and on day 15, he had his first dose of VIGIV. He received his second dose of cidofovir on day 17.
However, multiple lesions were not improving, so by day 20, he started intralesional cidofovir in six areas. Day 21 marked the second dose of VIGIV and day 24 was the third dose of IV cidofovir. Intralesional cidofovir was repeated on day 28, followed by IV cidofovir on day 32. That same day, the providers noted lesion improvement, 12 days after starting the intralesional cidofovir. He repeated the drugs on days 36 and 38, respectively, and his lab results improved: His CD4+ count was 361 cells/mcg and viral load 4,400 copies/mL. At day 45, he was discharged with topical cidofovir and oral tecovirimat.
What to Know About Severe Mpox
The two cases clearly show the effect HIV/AIDS has on patients with mpox, Dr. Do told IDSE. Both patients needed multiple types of treatments and required significantly longer duration of treatment. “They received multiple novel treatments that were out of the ordinary, although there are already limited treatment for mpox,” he said. “Both patients experienced IRIS, which also made their mpox appear like current treatment was failing.”
The process of mapping out these treatment plans began with searching through case reports and consulting the CDC, Dr. Do explained. “For the patient that received intralesional and topical cidofovir, there was a lot of digging for literature on how they were made, used, and number of doses,” he said. Although most treatments were readily available—although they took several days to arrive at the hospital—the intralesional and topical cidofovir needed to be compounded in-house.
Drs. Cozad and Do reported no relevant financial disclosures. This research was presented at MAD-ID 2025 (abstract 92 OR).

