Although today’s antiretroviral therapy (ART) allows people with HIV to live longer, healthier lives, they are at risk for metabolic side effects associated with increases in adipose tissue, including diabetes, hyperlipidemia and fatty livers.
“Weight gain associated with antiretroviral therapy is a major problem in HIV,” explained Allison Eckard, MD, a professor in the Departments of Pediatrics and Medicine, Divisions of Infectious Diseases, at the Medical University of South Carolina, in Charleston. “What’s more concerning, however, is the increased accumulation of visceral adipose fat and ectopic fat deposits in people with HIV-associated lipohypertrophy.”
Lipohypertrophy is associated with increased inflammation and can increase cardiometabolic risk.
“We currently have few treatments, and those treatments often show an ineffective response rate,” Dr. Eckard said.
Like many specialists whose patients are at risk for these conditions, HIV care providers are looking at glucagon-like peptide-1 (GLP-1) receptor agonists to see whether they would be effective for people with HIV.
“These medications have been nothing short of revolutionary in the diabetes world,” declared Todd Brown, MD, PhD, a professor of medicine and epidemiology, Division of Endocrinology, Diabetes and Metabolism, at the Johns Hopkins University, in Baltimore. For years, metformin had been the leading diabetes treatment, but in 2023, the American Diabetes Association stated that therapy should be individualized based on a person’s risk factors. If a person is at risk for cardiovascular disease (CVD), they should be given a GLP-1 receptor agonist, Dr. Brown explained at CROI 2024, held in Denver.
“That is a departure from what we had been teaching previously, and I think a good departure,” he said.
However, the best use of these products in people with HIV still needs to be determined. The number of studies using one of these products is growing exponentially, according to Dr. Brown, to about 14,000 papers per year. However, fewer than 10 of these studies focus on people with HIV.
Real-World Data
One study presented at CROI looked at real-world data about semaglutide’s use in people with HIV, who sought care at one of 10 HIV clinical care settings across the United States, which are part of the Centers for AIDS Research (CFAR) Network of Integrated Clinical Systems. They received semaglutide (now sold under the brand names Ozempic and Wegovy, Novo Nordisk) between 2018 and 2022 (abstract 797).
Heidi Crane, MD, MPH, the associate director of the University of Washington CFAR Clinical Epidemiology and Health Services Research Core, and a professor of medicine, Division of Allergy and Infectious Diseases, at the University of Washington, in Seattle, reported data from 222 people who were taking semaglutide and for whom they had measured baseline weight as well as kept track of their weight over time. Most of the patients were male, with one-fourth being female, and just over three-fourths had diabetes. The average hemoglobin A1c at initiation of treatment was 7.7%. Eighty-nine percent had a suppressed viral load at baseline, and the average CD4 T-cell count was just under 800 cells/mcL. The mean age of the patients was 53, and the average follow-up was 1.1 years.
On average, people with HIV lost 6.5 kg or 5.7% of body weight, according to Dr. Crane.
“We were curious about who it was that was losing this weight and whether it differed based on their baseline BMI [body mass index],” she said. They found “the greatest weight loss was among those with the highest weights at baseline.”
Those who were in obesity class III (patients who were =40 kg/m2 overweight), which was 50 people (22.5%) in the study, lost on average 8.8 kg (95% CI, 6.7-10.9 kg), according to Dr. Crane.
In addition, people who were taking a higher of dose of semaglutide were more likely to lose weight.
“Those with diabetes lost a smaller number of kilograms than those who did not have diabetes,” she said, but that was likely because “individuals with diabetes are more likely to be on the lower dose.”
Another analysis looked at people with lipohypertrophy in a randomized, double-blind, placebo-controlled trial of participants who received semaglutide or placebo for 32 weeks. There were 54 participants in each group. The patients, whose HIV was well controlled, were 18 years of age and older. The patients were overweight or obese and met the definition of HIV-associated lipohypertrophy. However, those with severe lipoatrophy were excluded. The patients also were not diagnosed with diabetes or CVD (poster 798).
“To date, we have not looked at the inflammation effects of semaglutide in HIV despite it showing anti-inflammatory effects and its potential modulation of the immune system in other populations,” Dr. Eckard said.
The median age of the patients was 53 years, 60% were male and 62% were Black. There were 15 (28%) smokers in the semaglutide group and 23 (43%) smokers in the placebo group.
“Our groups were well balanced at baseline,” Dr. Eckhard said, “except there were slightly more males in the semaglutide group and more smokers in the placebo group.”
The study was broken into four parts. The beginning of the study was spent assessing the stability and lifestyle of the participants, then began an interventional titration phase where people in the semaglutide group received a 0.25-mg dose weekly for four weeks and then a 0.5-mg dose weekly for four weeks. By week 9, they were on the full dose of 1 mg weekly for 24 weeks. The post-treatment observational phase lasted another 24 weeks, Dr. Eckhard explained.
Although they saw weight loss, some of that loss was in lean body mass, according to Dr. Eckhard. They also saw decreases in several inflammatory markers, such as C-reactive protein, interleukin-6 and sCD163.
“Semaglutide use caused notable decreases in several key biomarkers that appeared to be independent of that in weight loss in people with HIV-associated lipohypertrophy and without diabetes, suggesting, perhaps, semaglutide would be an effective treatment for HIV-associated lipohypertrophy,” Dr. Eckhard said.
Another research effort that includes people with HIV is the SLIM LIVER study (ACTG A5361; ClinicalTrials.gov Identifier: NCT04216589). Data reported at CROI 2024 looked at its effects on inflammatory markers and weight loss among people with HIV, as well as those with metabolic dysfunction–associated steatotic liver disease (MASLD) who received semaglutide from 2018 through the end of 2022.
Two analyses of the SLIM LIVER study enrolled participants with HIV who met the criteria for MASLD, according to Grace L. Ditzenberger, PT, DPT, a postdoctoral fellow at the University of Colorado Anschutz Medical Campus, in Aurora. Previously known as nonalcoholic fatty liver disease, MASLD is characterized by the accumulation of excess fat in the liver that is not caused by alcohol consumption or viral hepatitis. Over time, fat deposits may cause inflammation and cellular damage, and can result in liver disease. MASLD may also be associated with obesity, type 2 diabetes and other metabolic disorders.
MASLD is the most common cause of chronic liver disease in the United States, and is a leading reason for liver transplantation. An estimated 30% to 40% of people with HIV experience MASLD, slightly higher than the average among people without HIV, according to the National Institute of Allergy and Infectious Diseases.
The researchers found a weekly injection of semaglutide was safe and reduced the amount of fat in the liver by 31% in people with HIV who had MASLD.
The phase 2b pilot study enrolled people with HIV and MASLD who were 18 years and older and whose HIV viral load was suppressed to undetectable levels by ART. Participants were diverse with respect to ethnicity, race, gender and age. Of the 49 participants included in the analysis, 40 (82%) were taking ART regimens containing an integrase strand transfer inhibitor, which is a class of antiretroviral drugs shown to be highly effective at suppressing HIV but associated with weight gain in some people.
“The mean age was 50 years old, and it was a diverse population with almost 40% female, approximately 30% Black or African American, and around 40% Hispanic/Latino,” Dr. Ditzenberger said.
Participants self-injected semaglutide weekly at increasing doses until they reached a 1-mg dose at week 4 and participated in frequent safety monitoring visits. At 24 weeks, the study team assessed changes in participants’ liver fat content using a type of MRI specifically designed to measure the amount of fat in the liver. Participants experienced an average 31% reduction in liver fat, with 29% experiencing a complete resolution of MASLD, meaning their liver fat decreased to 5% or less of overall liver content (abstract 159). They also experienced weight loss, reduced fasting blood glucose and reduced fasting triglycerides, consistent with effects observed in studies of semaglutide in people without HIV (Ann Intern Med 2024 Apr 30. https://doi.org/10.7326/ M23-335).
“We also had 30% of our participants with complete MASLD resolution within the 24-week period,” said Jordan E. Lake, MD, MSc, a professor of infectious diseases with McGovern Medical School at UTHealth Houston, who is also the SLIM LIVER protocol co-chair.
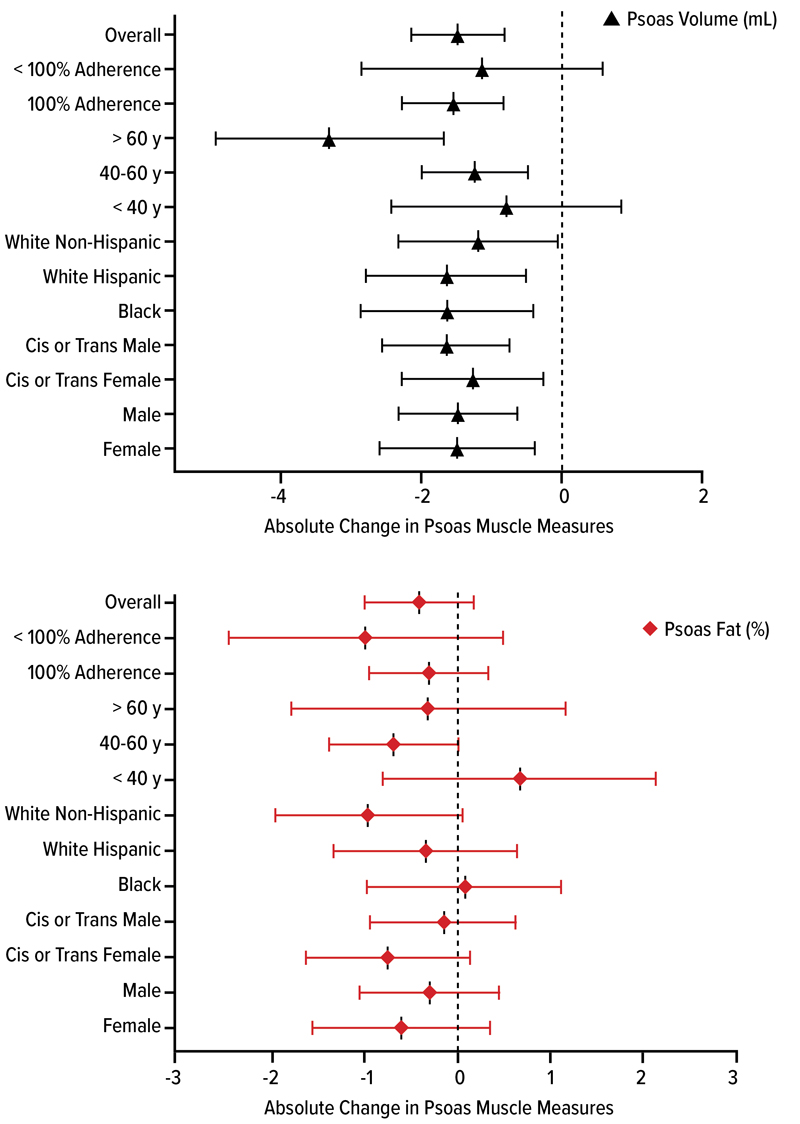
A subanalysis examined changes in muscle quality, quantity and function among people living with HIV who were enrolled in SLIM LIVER (abstract 799). Among the 46 participants for whom muscle measurements were available, the mean age was 50 years and their mean BMI was 35.5 kg/m2. Among people living with HIV taking semaglutide in SLIM LIVER, muscle volume decreased with weight loss, with no significant change in muscle fat or physical function, according to Dr. Ditzenberger.
In all the studies, semaglutide was generally well tolerated with an adverse event profile similar to people without HIV. The most common adverse events were gastrointestinal and included nausea, diarrhea, vomiting and abdominal pain.
These findings suggest that semaglutide is a safe and effective therapy for people with HIV, the researchers said. They may help inform healthcare decisions by people with HIV and their providers to enable healthier aging over the patient’s lifespan. According to the investigators, additional research is underway to understand whether people with HIV experience any unique immunologic or inflammatory pathway changes while taking semaglutide therapy.
“What we found is that semaglutide is an extremely effective treatment for MASLD in people with HIV,” Dr. Lake said. “Along with reducing liver fat in people with HIV, participants also experienced weight loss and improvements in blood sugar, insulin resistance and blood triglyceride levels.”
Dr. Brown reported relationships with Gilead Sciences, Janssen, Merck and ViiV Healthcare. Dr. Crane reported a relationship with ViiV Healthcare. Dr. Eckard reported a relationship with Theratechnologies. Dr. Lake reported relationships with Gilead and Theratechnologies.
This article is from the June 2024 print issue.