By Marie Rosenthal, MS
Gilead Sciences warned that tampered and counterfeit versions of its HIV medications, Biktarvy and Descovy, are in circulation within U.S. drug distribution networks.
Biktarvy is a combination of bictegravir, emtricitabine (FTC) and tenofovir alafenamide (TAF) that is given once daily for HIV treatment. Descovy is a combination of FTC and TAF that is used for HIV preexposure prophylaxis and treatment.
Unauthorized distributors have sold these counterfeits to pharmacies in genuine Gilead bottles that have been tampered with a counterfeit foil induction seal or label and contain fake tablets.
Working with the FDA, Gilead has alerted potentially affected pharmacies to investigate the potential for counterfeit or tampered Gilead medication sold by these distributors that may be within their recent supply.
The authenticity and safety of Gilead-branded medicines can only be secure when obtained directly through Gilead’s authorized distributors, the company said.
Gilead said it is working closely with the FDA, pharmacies and legal authorities to remove counterfeit and tampered medication from circulation and to prevent future distribution of these medications.
“The safety of individuals taking Gilead medication is always our first priority,” said Merdad Parsey, MD, PhD, Gilead’s chief medical officer. “We are taking aggressive action to ensure that healthcare providers and people who rely on our medicines can confidently distinguish authentic Gilead products from counterfeit drugs.”
The following elements can ensure dispensed bottles of Biktarvy and Descovy are authentic:
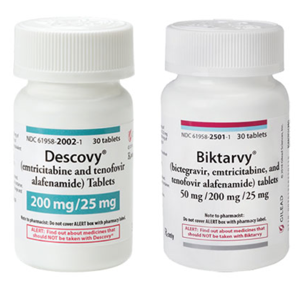
Authentic Biktarvy tablets are purplish-brown, capsule-shaped pills with “GSI” on one side and “9883” on the other.

Authentic Descovy tablets are blue, rectangular pills with “GSI” on one side and “225” on the other.

The FDA requires that Biktarvy and Descovy are dispensed in original packaging. Biktarvy and Descovy are dispensed in 30-count white plastic bottles, with white plastic caps and Gilead-branded labels.
Patients should be told to confirm that the pharmacy where they get their medication sourced their medications directly from authorized distributors. A list of Gilead authorized distributors can be found here.
Individuals who believe they have been dispensed a counterfeit and/or tampered Gilead medication should report the medicine to their doctor and pharmacy and Gilead Product Quality Complaints (800-445-3235 or QualityComplaints@gilead.com).