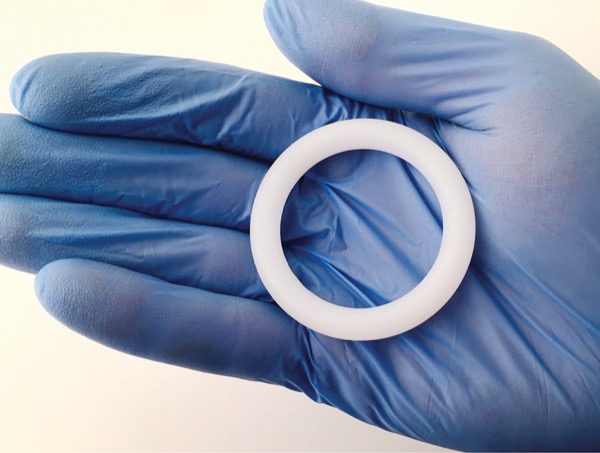
The dapivirine vaginal ring (DVR) and oral pre-exposure prophylaxis (PrEP) was safe and effective at preventing HIV in pregnant cisgender women, who are three times more likely to acquire HIV through sexual intercourse than similarly aged people who are not pregnant, according to Felix Mhlanga, Bsc HPT, MBChB, MMed, an obstetrician-gynocologist at the University of Zimbabwe Clinical Trials Centre.
This study, presented at CROI 2024, in Denver, marks a significant advancement in protecting the health of pregnant people and their babies against HIV, he explained. When discussing his findings from the DVR phase 3b clinical trial, Dr. Mhlanga, reported no significant difference in pregnancy outcomes, complications and congenital abnormalities between women using monthly DVR and women taking daily oral PrEP.
“It is clear that the sufficient data [we have] support the use of the DVR for HIV prevention together with the oral PrEP as a method for preventing HIV in pregnant women,” Dr. Mhlanga said.
The dapivirine ring made of flexible silicone, continuously releases dapivirine into the vagina, and is replaced monthly by the user. Although the World Health Organization recommends the DVR for PrEP and several African countries have approved it, there have been a paucity of data about the effects of the DVR during pregnancy, particularly as it relates to fetal toxicities and pregnancy complications. This study was the largest cohort to date with the longest duration of exposure to the DVR during pregnancy, according to Dr. Mhlanga.
Pregnant women in South Africa, Uganda and Zimbabwe ages 18 to 40 were randomly assigned to receive the DVR or oral PrEP with emtricitabine-tenofovir disoproxil fumarate starting in their second trimester until delivery or 41 weeks and six days, whichever came first.
Of the 248 pregnancies followed, 95% of deliveries were at term, with only two stillbirths and one miscarriage. While 26% of participants had hypertension and hemorrhages, Dr. Mhlanga maintained that such uncommon complications were similar to the community background rates of people not taking any type of PrEP, and therefore, were unrelated to the prophylaxis, regardless of the method. Of note, no study participants acquired HIV.
In addition to reporting no maternal deaths, Dr. Mhlanga also emphasized the lack of infectious complications such as endometritis, puerperal sepsis and chorioamnionitis.
“You could imagine that if you’re using a foreign body, which is the dapivirine ring, it could lead to infectious complications,” Dr. Mhlanga. “We didn’t see them in terms of outcomes.”
The vaginal ring, as well as oral PrEP, have the potential to safely prevent HIV during pregnancy and break the cycle of transmission among populations facing heightened vulnerability to infection in Africa, as well as contribute to global efforts to eliminate HIV, while giving the woman options, according to Dr. Mhlanga.
“This makes me super excited,” he said. “When I get back home we seem to have a path to provide care to people who urgently need it.”
The large clinical trial was conducted by the Microbicide Trials Network, with funding by the National Institute of Allergy and Infectious Diseases, Eunice Kennedy Shriver National Institute of Child Health and Human Development, and the National Institute of Mental Health. Dr. Mhlanga reported no relevant financial disclosures.
This article is from the June 2024 print issue.