By Ethan Covey
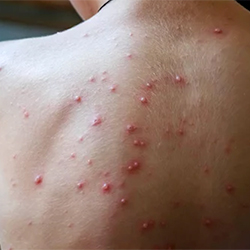
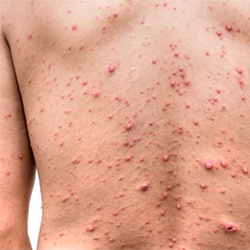
A program focused on expanding laboratory testing for varicella in Minnesota has highlighted the importance of lab confirmation to correctly diagnose the disease.
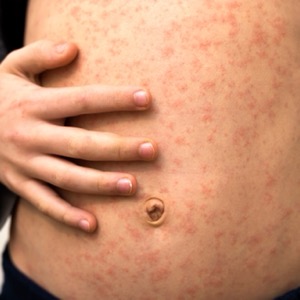
Varicella can present diagnostic challenges, particularly among patients who have received a varicella vaccine (MMWR Morb Mortal Wkly Rep 2024;73:245-246).
In 2016, the Minnesota Department of Health (MDH) established expanded laboratory testing for confirmation of varicella in the state. The MDH Public Health Laboratory provided free testing for people with suspected varicella, and polymerase chain reaction (PCR) testing for varicella-zoster virus (VZV), herpes simplex virus 1 (HSV-1), herpes simplex virus 2 (HSV-2) and enterovirus was conducted on all specimens submitted to that laboratory.
Educational outreach to healthcare providers was conducted, specimen collection kits were offered directly to people with suspected varicella, and notification letters were sent to families of children who had been exposed to varicella.
Following initiation of the laboratory testing program, the proportion of lab-confirmed cases of varicella rose from 17% to 36%. Additionally, the proportion of PCR-confirmed varicella cases increased by 62%.
The investigation also showed the difficulty of clinical diagnosis in correctly identifying the disease. Among 208 patients receiving a clinical diagnosis of varicella at a medical facility, 45% had positive VZV test results. Detection of VZV was far lower among those who had received varicella vaccination, compared with unvaccinated people (22% vs. 66%).
“These findings suggest clinical diagnosis can be unreliable, especially in vaccinated people, and emphasize the importance of laboratory testing for accurate diagnosis and management of varicella,” said Cynthia Kenyon, PhD, an epidemiologist supervisor, VPD Surveillance, at the MDH.
“Further studies should investigate the impact of implementing routine laboratory testing for varicella on the effectiveness of disease control efforts, including evaluation of how access to testing could contribute to disparities related to exclusion recommendations,” Dr. Kenyon added.
Dr. Kenyon reported no relevant financial disclosures.