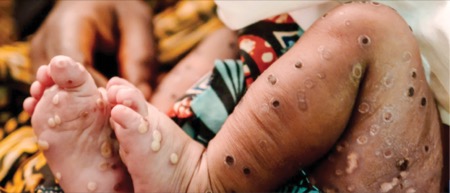
The antiviral drug tecovirimat (Tpoxx, SIGA Technologies) did not reduce the duration of mpox lesions among children and adults with clade I mpox in the Democratic Republic of the Congo (DRC), based on an early analysis of data from the PALM007 trial.
However, study participants saw a 1.7% overall mortality, regardless of whether they received the drug, which was much lower than the mpox mortality rate of 3.6% or higher reported among all cases in the DRC. The study demonstrated that people with mpox can achieve better outcomes when they are hospitalized and provided high-quality supportive care.
“These findings are disappointing, but they give us essential information and reinforce the need to identify other therapeutic candidates for mpox while we continue research on tecovirimat use in other populations with mpox,” said Jeanne Marrazzo, MD, MPH, the director of the National Institute of Allergy and Infectious Diseases.
The randomized, placebo-controlled PALM007 trial was launched in October 2022 to examine the safety and efficacy of tecovirimat for mpox treatment in adults and children. The study enrolled 597 people with laboratory-confirmed mpox at two sites in the DRC. Study participants were randomly assigned to receive tecovirimat or placebo and admitted to a hospital for at least 14 days, where they were monitored for safety and resolution of mpox lesions. All participants received supportive care including nutrition, hydration and treatment for secondary infections.
Tecovirimat was well tolerated. However, mortality was lower, and lesions resolved faster than anticipated regardless of whether the participants received tecovirimat or placebo. Although the results were not what they had hoped for, “this study delivered urgently needed evidence to guide the mpox response in Central Africa,” said co-principal investigator Jean-Jacques Muyembe-Tamfum, MD, PhD, the director-general of Institut National de la Recherche BiomÉdicale and a professor of microbiology at Kinshasa University Medical School, in the DRC.
Fifteen African countries had reported mpox by September, according to the WHO. A recent report from the CDC indicated that 67% of suspected mpox cases and 78% of suspected mpox deaths in the DRC, have occurred in people 15 years and younger.
In related news, the WHO and partners established a mechanism to allocate supplies of vaccines and diagnostics for mpox to those at the highest risk for infection, including for vaccinating contacts of confirmed cases, and providing access to point-of-care diagnostics to countries with ongoing mpox outbreaks. More than 3.6 million doses of vaccines have been pledged for the mpox response.
“WHO and partners are supporting the government of the Democratic Republic of the Congo and other countries to implement an integrated approach to case detection, contact tracing, targeted vaccination, clinical and home care, infection prevention and control, community engagement and mobilization, and specialized logistical support,” said Mike Ryan, MD, the executive director of WHO’s Health Emergencies Programme, adding it should provide an equitable and reliable pipeline of tools to interrupt transmission and reduce suffering.
The trial is sponsored by NIAID and co-led through a government-to-government partnership the INRB. Further analyses and detailed results will be released through scientific channels. The sources reported no relevant financial disclosures.
This article is from the October 2024 print issue.