By IDSE New Staff
The antiviral drug tecovirimat (Tpoxx, SIGA Technologies) did not reduce the time to lesion resolution or have an effect on pain among adults with mild to moderate clade II mpox and a low risk of developing severe disease, according to an interim data analysis from the international STOMP trial.
Therefore, the study’s data safety and monitoring board (DSMB) recommended stopping further enrollment of participants who were being randomized to tecovirimat or placebo. The National Institute of Allergy and Infectious Diseases accepted the DSMB’s recommendation. Given the lack of an efficacy signal, NIAID also closed enrollment into an open-label study arm for participants with or at elevated risk for severe disease that was not designed to estimate the drug’s efficacy.
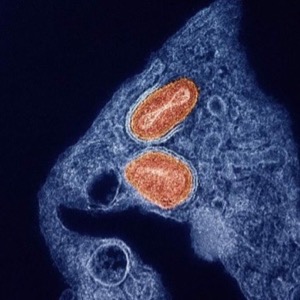 A colorized transmission electron micrograph of two particles of the virus that causes mpox, cultivated and purified from cell culture. Image courtesy of NIAID.
A colorized transmission electron micrograph of two particles of the virus that causes mpox, cultivated and purified from cell culture. Image courtesy of NIAID.The National Institute of Allergy and Infectious Diseases accepted the DSMB’s recommendation. Given the lack of an efficacy signal, NIAID also closed enrollment into an open-label study arm for participants with or at elevated risk for severe disease that was not designed to estimate the drug’s efficacy.
“The initial STOMP findings provide valuable insight to inform clade II mpox medical countermeasures and underscore the critical importance of conducting well-designed randomized clinical trials during infectious disease outbreaks,” said NIAID Director Jeanne Marrazzo, MD, MPH. “Before 2022, no treatment candidate had been studied in people with mpox, and this trial is a critical step in our systematic evaluation of existing antivirals like tecovirimat while pursuing novel antivirals and antibody-based mpox therapeutics.”
The STOMP study began in September 2022 as part of the U.S. response to the clade II mpox outbreak. The randomized international efficacy study enrolled participants in Argentina, Brazil, Japan, Mexico, Peru, Thailand and the United States, including Puerto Rico, who had been ill with mpox for fewer than 14 days.
In this double-blind study, participants were randomly assigned at a 2:1 ratio to receive tecovirimat or placebo. Children, pregnant people and other participants with severe disease, certain skin conditions or substantially suppressed immune systems were assigned to an open-label study arm. The STOMP study assessed all participants’ safety and, in randomized arms, evaluated whether a 14-day course of tecovirimat reduced the time to clinical resolution of visible mpox lesions and improved other outcome measures such as pain, compared with placebo.
A planned interim analysis at 75% of the study’s target enrollment showed there was no difference in the time to lesion resolution between participants treated with tecovirimat compared with those who received placebo. Pain decreased similarly between participants treated with tecovirimat and those who received placebo. At the DSMB’s request, an additional assessment was performed and showed there was a less than 1% chance that the study would show tecovirimat to be effective if it were to complete enrollment and follow-up, based on the study design and available data. At the time of analysis, reported adverse events were low and comparable between tecovirimat and placebo. By design, the open-label study arm did not assign participants to receive placebo, so STOMP will not draw conclusions about the efficacy of tecovirimat in participants with, or at elevated risk for, severe clade II mpox.
Further analyses of the study data are ongoing. Study participants are being notified of the findings, and study clinicians will make individual clinical care plans with participants based on their disease severity and symptoms. The CDC holds an expanded access investigational new drug protocol for mpox treatment outside of research settings. Eligible people include those with severe immunocompromise, including people with advanced HIV, for whom the role of tecovirimat treatment has not been fully established through a clinical trial.
“STOMP was a banner study for its speed of startup, inclusiveness and collaboration across governments and public health authorities,” said study chair Timothy Wilkin, MD, MPH, the chief of the Division of Infectious Diseases and Global Public Health at the University of California, San Diego. “This study may serve as a model for outbreak response, delivering essential scientific evidence while also enabling equitable treatment access.”
The STOMP findings are consistent with the results from an earlier trial of tecovirimat among children and adults with clade I mpox in the Democratic Republic of the Congo.
Mpox spreads mainly through close contact with an infected person. Two types of the virus have been identified, referred to as clades I and II, historically present in Central and West Africa, respectively. A clade II subtype virus caused a global mpox outbreak in 2022, and the virus continues to circulate at low levels. In 2024, a clade I outbreak in Central and East African countries was declared a public health emergency of international concern by the WHO. Travel-related cases of clade I mpox have been reported internationally, and the first reported case in the United States was diagnosed on Nov. 15.
The FDA initially approved tecovirimat to treat mpox’s close relative, smallpox, but the drug’s safety and efficacy as an mpox treatment have not been established prior to this year.
Throughout the study, NIAID and STOMP investigators collaborated closely with the CDC, FDA, other study country health authorities, teams conducting other studies of tecovirimat and additional key partners in the mpox response. These consultations allowed research partners to arrive at efficient answers to scientific questions, balance evidence needs with compassionate-use considerations and to avail up-to-date information for clinicians treating clade II mpox globally.
An additional study, UNITY, sponsored by ANRS Emerging Infectious Diseases, is evaluating tecovirimat with a similar study design to STOMP in Argentina, Brazil and Switzerland. More information about the UNITY study can also be found on ClinicalTrials.gov using the identifier NCT05597735.