By Norah Chinn
More than 100 farms spanning 12 states have confirmed highly pathogenic avian influenza (HPAI) A (H5N1) infections in their dairy herds, according to Vivien Dugan, PhD, the director of the CDC’s Influenza Division, who updated the agency’s Advisory Committee on Immunization Practices (ACIP) during its June meeting.
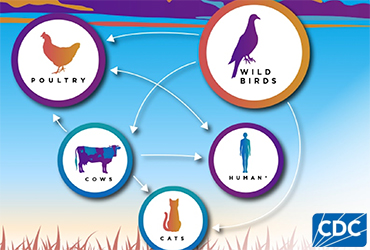
Since the outbreak was identified in March, only four human cases of the virus have been linked to exposure to infected cows, according to the CDC. All infections occurred in dairy workers with direct exposure to cattle presumed or confirmed to be infected, resulting in symptoms such as eye redness and watery eyes, with one patient having a cough without fever. Fortunately, none required hospitalization, all patients recovered fully and no human-to-human transmission was observed.
While the overall risk to the public remains low, individuals with prolonged, unprotected exposure to infected animals or contaminated environments face the highest risk, Dr. Dugan said. The CDC advises exposed individuals to monitor symptoms starting from their first exposure and continue for 10 days after their last exposure.
Those who are hunters are at risk, said Sandra Adamson Fryhofer, MD, a liaison member representing the American Medical Association. “I have a lot of patients who are hunters, and I wonder about their exposure to wild birds,” she said, asking whether current flu tests would be helpful in detecting the virus in these patients.
Dr. Dugan said current commercial tests used in physicians’ offices and urgent care facilities would produce positive results for influenza A, but samples would need to be sent to a state laboratory to subtype H5N1.
The CDC, the Department of Agriculture, the FDA, and state and local public health agencies are collaborating to lead public health efforts to protect human health and safety, understand the risk of H5N1 to people, prevent additional cases, and assess genetic changes indicating virus adaptation and spread.
The CDC continues to analyze emergency department data, influenza lab tests and other public health data, especially in areas with H5N1 detection in dairy cattle, poultry and wild birds. Since March 3, at least 690 people from affected farms have been monitored, with no unusual influenza activity reported, according to Dr. Dugan.
There was no impact on the current CDC influenza diagnostic assay’s ability to detect the virus, with no known resistance to FDA-approved antiviral drugs, Dr. Dugan said.
Two available candidate vaccine viruses closely related to the H5N1 virus are expected to provide effective protection and could serve as a basis for future vaccine development, if necessary. However, she added that an “H5 vaccine program is not currently planned,” she said, adding the government was focusing on a broader preparedness plan that would include a vaccine, if needed.
“I wonder whether it might be appropriate for the ACIP, through its influenza workgroup, to be presented data concerning the effectiveness or potential effectiveness of the currently available H5N1 vaccine and to at least begin discussions of a possible utilization in a preventive fashion of H5N1 vaccine,” said William Schaffner, MD, a past medical director of the National Foundation for Infectious Diseases and former NFID liaison to the ACIP at the CDC. “I just observed in Finland they are starting to use H5N1 in potentially exposed dairy workers. I think these are issues that are quite appropriate for this committee.” Dr. Schaffner is a professor of preventive medicine at Vanderbilt University School of Medicine, in Nashville, Tenn., and an expert in influenza.
Increased Surveillance
In May, the CDC began sharing influenza A virus wastewater data from across the United States. Wastewater surveillance detects influenza A viruses by comparing current levels with those from the 2023-2024 influenza season. High levels prompt outreach to state, local and federal partners in those areas. When a signal is detected, efforts are made to determine the driving factors, but so far, no potential human circulation has been identified, Dr. Dugan reported.
Epidemiological investigations conducted with health and agricultural partners at all levels of government are trying to answer key questions about evidence of infection evidence, spectrum of illness, asymptomatic rates, exposure types on farms and dairies, and behaviors linked to human infections or protection. Dr. Dugan explained the goal is to evaluate the risk for symptomatic and asymptomatic infections and to assess current detection and response procedures.
The CDC’s summer influenza surveillance strategy, developed with state and local public health agencies, monitors the spread of H5N1 beginning with continued monitoring of people with recent exposure on confirmed farms and then extending to the general population, according to Dr. Dugan. Enhanced national surveillance at seasonal influenza levels will aid the detection of H5N1 human cases. Any flagged syndromic and wastewater data will be followed up by the CDC, which will conduct provider outreach and continue influenza testing throughout the summer, especially for patients with recent relevant exposure.
“There are efforts looking at all our surveillance systems across the U.S. that we use for seasonal influenza, but they also serve the purpose of detecting novel influenza for any signals there. We are trying to support the states and monitoring those who are exposed to then test should they have symptoms,” Dr. Dugan said. “For serologic studies that is one of our high priorities here at CDC, and we are supporting the Michigan Department of Public Health in their efforts to initiate an epidemiologic serostudy, so those efforts are ongoing. But certainly, we are looking for more opportunities and working with our partners to develop more opportunities for serologic studies in people as well.”
The CDC’s global influenza program focuses on detecting and characterizing seasonal and novel influenza viruses like H5N1, Dr. Dugan said. It contributes data for biannual vaccine composition and generates and validates candidate vaccine viruses. The program works with global health offices to keep them informed about the U.S. situation.
Since January 2022, there have been 29 human cases of the H5N1 virus reported globally, according to Dr. Dugan. The virus was first found in birds in 1996 and is not a new threat to animals and people. (Read an in-depth story about the outbreak here.)
“We are looking very closely at the genetics, the genotypes, the phenotypes of viruses to understand that big question. From the scientific community, the public health community, the animal community, we do want to try to understand why is this happening now? What has changed?” Dr. Dugan said. “The H5N1 virus has been circulating for several years now in wild birds and it’s impacted poultry. We’ve had some experience with this particular version of the virus, but I think it’s a much bigger question and may take some more time to really figure out.”
In related news, the Department of Health and Human Services, through the Biomedical Advanced Research and Development Authority (BARDA), part of the Administration for Strategic Preparedness and Response, will provide $176 million to Moderna to develop an mRNA pandemic influenza vaccine.
The award was made through BARDA’s new Rapid Response Partnership Vehicle Consortium, a 10-year, multipurpose program designed to support advanced research and development countermeasures.
Moderna’s influenza vaccine candidate uses current mRNA technology leveraged successfully during the COVID-19 response, resulting in one of the first two FDA-authorized COVID-19 vaccines. In 2023, BARDA issued a request for proposal to Moderna and other companies to develop mRNA vaccines to prepare for potential public health emergencies caused by influenza viruses. mRNA vaccines have the potential to complement traditional vaccine technologies during a pandemic influenza emergency response.
This award should enable the rapid development of an influenza vaccine to target various viral strains of pandemic potential and enable the large-scale production of pandemic vaccines if needed, according to HHS.



