The benefits of vaccination with recombinant zoster vaccine (RZV; Shingrix, GSK) outweigh safety concerns in immunocompromised populations, according to a systematic review of studies involving multiple patient groups.
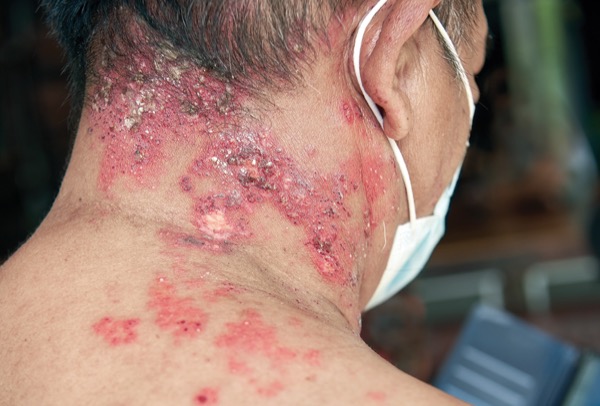
Patients who are immunocompromised, either due to their medical condition or immunosuppressive treatments, are at an elevated risk for herpes zoster. Bone marrow and stem cell transplant patients have among the highest risk, estimated at about nine to 21 times that of immunocompetent adults. Other notable groups with elevated risk include patients with hematologic malignancies (seven to 15 times), solid-organ transplants (four to nine times), solid tumors (three to five times), HIV (two to four times) and autoimmune conditions (two to three times).
Francis A. Farraye, MD, MSc, a gastroenterologist and a professor of medicine at the Mayo Clinic in Florida, in Jacksonville, who was a co-author of the review, stressed the importance of herpes zoster prophylaxis in these patient groups. “With the recent Advisory Committee on Immunization Practices [ACIP] recommendation to vaccinate immunocompromised patients or those likely to be immunocompromised in the future, we can dramatically lower the risk for shingles in these patients,” he told Infectious Disease Special Edition.
However, the vaccine’s strengths and weaknesses in different groups of patients had not been reviewed comprehensively. Dr. Farraye and his colleagues sought to fill this gap and better equip clinicians to make recommendations for their immunocompromised patients (Vaccine 2023;41[1]:36-48). Across the immunocompromised populations studied, recombinant RZV was safe and effective. (The key findings of their review for specific immunocompromised populations are detailed in the Table.)
| Table. Key RZV Efficacy and Safety in Selected Immunocompromised Populations | ||
| Patient population | Efficacy | Safety |
|---|---|---|
| HCT recipients | Post-transplant:
| Post-transplant:
|
| Hematologic malignancies |
| Most common AEs: pain, fatigue (phase 3) |
| Solid tumors | Durable immunologic response, particularly when given before chemotherapy initiation (phase 2/3) | Most common AE: myalgia (phase 2/3) |
| HIV | Data limited; potential for efficacy based on immune cell counts and antibody titers (phase 1/2a) | Data limited; promising safety record (phase 1/2a) |
| RA | Strong efficacy (observational, single-center studies) | AEs included RA flares (observational, single-center studies) |
| IBD | Vaccination associated with decreased odds of developing HZ (retrospective studies) | Low risk for post-vaccination IBD flare (retrospective study) |
| AE, adverse event; GVHD, graft-versus-host disease; HCT, hematopoietic cell transplant; HZ, herpes zoster; IBD, inflammatory bowel disease; RA, rheumatoid arthritis; RZV, recombinant zoster vaccine; VE, vaccine effectiveness. | ||
Joanna Schaenman, MD, PhD, an expert in transplant infectious diseases and a professor of medicine at the David Geffen School of Medicine at the University of California, Los Angeles, praised the review because it is a single source for zoster risk and vaccination data among several vulnerable patient populations. With the diverse set of patients at risk for shingles and the myriad of specialists involved in their treatment, “there’s a lot of fragmentation” in the care of these patients, Dr. Schaenman told Infectious Disease Special Edition. “And because the current model for ID consultation is generally treating infections rather than preventing infection, there are many lost opportunities for effective intervention.”
To combat these missed opportunities and prevent the significant morbidity and potential mortality associated with shingles, Dr. Schaenman recommended a multispecialty approach. “We in ID can do a better job getting out the message regarding prevention” with vaccination, she suggested. In addition, “primary care providers need to make sure that age-appropriate vaccination is part of their prevention messaging,” similar to recommendations for mammograms and colonoscopies.
Also, given the nuances involved in vaccinating different vulnerable patient populations, Dr. Schaenman suggested that the societies of the specialists involved in the care of patients who are immunocompromised should do more to “raise awareness about vaccination recommendations” so that vaccinations are performed prior to the start of immunosuppression when possible.
Dr. Farraye agreed that clinicians specializing in areas other than ID need to be aware of and heed shingles vaccination recommendations. He said in his field, for example, “it is up to the gastroenterologist to be certain their patients are up to date on appropriate vaccinations, as we are the primary caregiver for these patients, and they are unlikely to see ID specialists unless they develop an infection requiring ID input.”
Strategies
One strategy recommended by the reviewers to aid in identifying vaccination candidates is using electronic health records to remind clinicians to consider RVZ based on patient-specific factors. To maximize the utility of such an approach, the reviewers emphasized the need to generate data for more granular subgroups of patients, such as those taking certain immunosuppressive regimens for a given medical condition, and data on the risks and benefits of immunosuppressive therapy initiation delay, temporary withdrawal or continuation at the time of the RZV in these groups. Dr. Schaenman added that she would like to see more data about the “best timing for vaccine administration and the number of doses to optimize both the humoral and T-cell–mediated immune response.”
Although patients ideally would be vaccinated before immunosuppression to maximize efficacy, citing recent studies in transplant patients (Clin Infect Dis 2020;70[2]:181-190; Am J Transplant 2021;21[6]:2246-2253), Dr. Schaenman emphasized that it is important for clinicians to realize they “can safely vaccinate IC [immunocompromised] patients who are stable” and improve outcomes for these patients.
Vaccination can make sense from a health economics perspective as well, according to research conducted by Dr. Farraye, which found that “Shingrix vaccination is cost-effective and even cost-saving in populations of patients with IBD [inflammatory bowel disease]” (Aliment Pharmacol Ther 2023;57[11]:1326-1334).
The review was funded by GSK. Dr. Farraye reported financial relationships with AbbVie, Avalo Therapeutics, Braintree Laboratories, Bristol Myers Squibb, Fresenius Kabi, GI Reviewers, GSK, Iterative Health, Janssen, Lilly, Pfizer, Sandoz Immunology, Sebela and Viatris. Dr. Schaenman reported no relevant financial disclosures.
This article is from the June 2024 print issue.