By IDSE News Staff
A recent case report provides evidence that enterovirus D68 (EV-D68) directly infects spinal cord neurons, leading to a robust immune response—a direct causation of acute flaccid myelitis (AFM), which resulted in large outbreaks seen in 2014, 2016 and 2018 (N Engl J Med 2022;386:2059-2060).
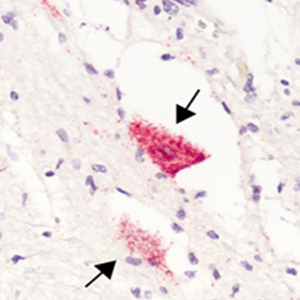 Arrows point to the motor neurons of a child who had AFM that contain EV-D68 RNA, stained red. Image by Dr. Matthew Vogt.
Arrows point to the motor neurons of a child who had AFM that contain EV-D68 RNA, stained red. Image by Dr. Matthew Vogt.Increasing evidence demonstrated a causal link between AFM and EV-D68 infection, one of more than 100 non-polio enteroviruses. EV-D68 can cause mild to severe respiratory illness like runny nose, wheezing, cough, body aches and muscle aches. In some cases, AFM can develop.
“Enterovirus D68 is much like the rhinovirus, where it typically starts with common cold-type symptoms,” said Matthew Vogt, MD, PhD, an assistant professor in the Division of Pediatric Infectious Diseases, University of North Carolina at Chapel Hill School of Medicine, and a member of the UNC Children’s Research Institute. “Right when those symptoms start to get better, within five days or so, then weakness will begin to appear. It’s highly variable. The weakness can range from subtle to paralysis of every muscle in the body,” he said.
EV-D68 is often detected in respiratory specimens of patients with AFM, but rarely detected in their cerebrospinal fluid. Few autopsies have examined human AFM pathogenesis, so most understanding of pathogenesis is from mouse models of infection. More evidence was supporting the link between EV-D68 and AFM, but proof of direct causation was lacking.
This study began in 2019, when Dr. Vogt was a pediatric infectious diseases fellow in the laboratory of James Crowe, MD, at Vanderbilt University Medical Center, in Nashville, Tenn. At that time, his research involved investigating the types of antibodies humans make in response to EV-D68 infection.
“Antibody and B cell research is very powerful now, since the techniques used identify mechanisms of immunity and, at the same time, can be used to isolate therapeutic molecules,” Dr. Crowe said.
Twelve years after a 5-year-old boy died from AFM in 2008, Dr. Vogt and other researchers returned to this patient’s autopsy specimens to investigate the pathology and immune response to the virus. The pathogenesis was con-firmed when the study revealed EV-D68 directly infecting spinal cord neurons. The region of infected neurons clinically corresponded to upper limb weakness, and a robust immune response was also present.
Dr. Vogt called the central nervous system an “immune privileged site,” meaning the immune system works dif-ferently in the spinal cord and brain from the rest of the body. “Sometimes when there’s an immune response to an infection, it can cause lasting damage to the infected tissues, even affecting surrounding cells that are not infected. By design, cytolytic CD8+ T cells help to kill infected cells, which is a normal part of clearing viruses in most tissues. But when this happens in the central nervous system, we do not think the neurons regenerate. In the study, we found a lot of these CD8+ T cells right in the area of where these infected neurons were,” Dr. Vogt said.
These findings lend additional plausibility to the role of immunopathogenesis contributing to AFM and connects EV-D68 with the disease. So, optimal acute treatment of AFM likely requires a multipronged approach focused on antiviral and anti-inflammatory strategies.
“There probably needs to be a balance,” Dr. Vogt said. “Antiviral or antibody drugs combined with an anti-inflammatory type of medicine could be good for treatment. It’s currently common for AFM patients to be treated with an immune-modulating medicine, trying to limit that immune-mediated damage like what the cytolytic T cells can do. The challenge is if you take away the entire immune response, then you might take the brakes off that virus infec-tion, causing the virus to go out of control. My hypothesis after seeing the data is that a patient should be treated with both options when they are available. Unfortunately, we don’t have antiviral or antibody drugs available for treating EV-D68 infection in humans right now.”
Dr. Vogt and his colleagues have used monoclonal antibodies to neutralize EV-D68. The antibody given to infected mice during the onset of paralysis improves overall paralysis. Since this piece of evidence in the study leaves no doubt that EV-D68 causes AFM, information derived from this case report may inform treatment approaches and future direction of laboratory studies, they said.
This research also validates the high value of conducting autopsies and biobanking tissues for cases of poorly ex-plained infectious syndromes.
AFM is a serious neurologic polio-like condition that causes muscle weakness, sometimes leading to permanent pa-ralysis. The condition is uncommon, and it affects the nervous system, specifically the spinal cord, which causes the muscles and reflexes in the body to become weak. It can often lead to paralysis of the skeletal muscles, and in severe cases, it can affect the muscles of swallowing and breathing. More than 90% of cases have occurred in children. Recovery from AFM can vary but rarely involves full recovery of overall strength. Mortality is low, with only two re-ported deaths out of 682 confirmed AFM cases in the United States since 2014.
Funding for the study was provided by the National Institute of Allergy and Infectious Diseases and the Pediatric In-fectious Diseases Society-St. Jude Children’s Hospital Fellowship Program in Basic Research. Dr. Vogt was supported by grant K08 AI153125 from the National Institute of Allergy and Infectious Diseases and the Pediatric Infectious Diseases Society-St. Jude Children’s Hospital Fellowship Program in Basic Research. Dr. Crowe was supported by grant U19 AI117905 from the National Institute of Allergy and Infectious Diseases.




