By Gina Shaw
Originally published by our sister publication Pharmacy Practice News
Updated Aug. 5
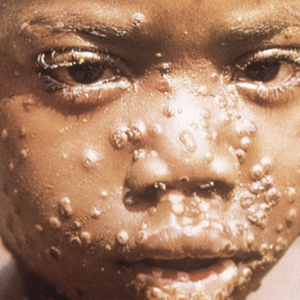
The Biden administration officially declared monkeypox to be a national public health emergency (PHE) on Thursday, Aug. 4, 2022, as cases of the disease in 48 states, Washington, D.C., and Puerto Rico rose to more than 6,600 nationwide, according to the CDC. (Only Montana and Wyoming have yet to report any cases.)
“This public health emergency will allow us to explore additional strategies to get vaccines and treatments more quickly out to the impacted communities. And it will allow us to get more data from jurisdictions so we can effectively track and attack this outbreak,” said Robert Fenton, the newly appointed White House national monkeypox response coordinator, at Thursday's media briefing.
The declaration comes as supplies of Jynneos (Bavarian Nordic), the only FDA-approved vaccine for monkeypox, continue to be limited. To help ease the backlog, on Thursday, the FDA announced a potential solution: dividing Jynneos doses into fifths to stretch current vaccine stocks.
ASHP has responded by releasing a letter calling on Health and Human Services Secretary Xavier Becerra to leverage the Public Readiness and Emergency Preparedness (PREP) Act to allow uniform, nationwide access to monkeypox immunizations by granting pharmacists the ability to order and administer the vaccine.
“Throughout the COVID-19 pandemic, pharmacists demonstrated their ability to be a source of timely access to vaccines on a national scale,” wrote ASHP vice president for government relations Tom Kraus, JD. “In the case of monkeypox, the [CDC] recommends that the vaccine be given within four days from the date of exposure for the best chance to prevent onset of the disease, and within 14 days to reduce symptoms of the disease. When patients access care from a pharmacist, directing the patient to another provider for vaccine administration would unnecessarily delay administration. To ensure pharmacists are able to expand access as vaccine supplies increase, we urge you to proactively authorize pharmacists to order and administer monkeypox vaccines.”
Limited Distribution
Although more than 336,000 doses of Jynneos have been distributed since May, there have been widespread reports of patients not being able to access the vaccine. “Those [vaccines] are being distributed out of the Strategic National Stockpile, with allocations being made to areas most in need similar to the way supplies of the COVID-19 vaccines were initially managed,” Michael Ganio, PharmD, senior director of pharmacy practice and quality for ASHP, told Pharmacy Practice News. “At present, most of that supply is going to state public health departments for distribution at either their own vaccine sites or with partner hospitals or health systems.”
With more than one-fourth of all monkeypox cases reported to date (1,553 as of Aug. 3), New York City has been hardest hit by the outbreak. In addition to New York state declaring a monkeypox PHE, the city declared its own on Aug. 1. On July 31, New York Gov. Kathy Hochul announced that the federal government would send approximately 110,000 more vaccines to New York state and New York City during phase 3 of the federal Jynneos vaccine distribution.
The state will receive 30,000 of these doses in three separate phases during the coming weeks, while the New York City Department of Health and Mental Hygiene will receive approximately 80,000 doses over the same period. Other states and cities will receive smaller allocations.
The next shipment of 500,000 doses from Denmark-based Bavarian Nordic is not expected to arrive until the end of October, a company spokesperson told The Washington Post.
Because of tight supplies of Jynneos, the CDC has been limiting vaccine availability only to the high-risk contacts of monkeypox patients and the healthcare workers treating them. The official criteria include:
· people who have been identified by public health officials as a contact of someone with monkeypox;
· people who are aware that one of their sexual partners in the past two weeks has been diagnosed with monkeypox;
· people who had multiple sexual partners in the past two weeks in an area with known monkeypox; and
· people whose jobs may expose them to orthopoxviruses, such as laboratory workers who perform testing for orthopoxviruses; laboratory workers who handle cultures or animals with orthopoxviruses; and some designated healthcare or public health workers.
ACAM2000, an alternative vaccine, is in much greater supply, but it has a number of significant risks and drawbacks, Dr. Ganio said. Approved for vaccination against smallpox, ACAM2000 has millions of doses in the Strategic National Stockpile in case of a bioterror attack involving the disease. But unlike Jynneos, which is based on the modified vaccinia virus Ankara and does not replicate in the body, ACAM2000 is made with unmodified vaccinia virus that can replicate after vaccination.
“It can cause serious illness in people who are severely immunocompromised, individuals who are pregnant, people with eczema and infants,” Dr. Ganio said. “It also has been associated with cases of myocarditis, which were relatively few and mild in studies leading to its approval, but that could be more significant if large populations were vaccinated with ACAM2000.”
A pustule also develops at the vaccination site that can shed live virus, putting close contacts of the vaccinated person at risk for infection until the pustule scabs over and the scab falls off, usually about two weeks later.
Technical Limitations
The declaration of the PHE does eliminate some of the technical barriers to using ACAM2000. Because it is only approved for smallpox, not monkeypox, any institution wanting to offer the vaccine previously had to go through the Investigational New Drug (IND) application process for expanded access. “Since this is a government entity procuring and distributing these vaccines, they are following strict approval processes,” Dr. Ganio said. “A lot of paperwork is needed for an IND, because this is essentially a drug trial, although not as complicated as a randomized controlled trial.”
ASHP has established a web-based resource center on the monkeypox outbreak at www.ashp.org/pharmacy-practice/resource-centers/monkeypox-resources.
FDA’s ‘Dose-Sparing’
On Thursday, the FDA commissioner, Robert Califf, MD, detailed the agency's strategy for stretching vaccine supplies. “We’re considering an approach for the current doses of Jynneos that would allow health care providers to use an existing one dose vial of the vaccine to administer a total of up to five separate doses,” he explained. “This approach, which we’re referring to as dose-sparing, would change the method of administration for Jynneos, which currently is administered subcutaneously.”
Specifically, the vaccine would be injected intradermally—a route that offers “some advantages,” Dr. Califf said, “including an improved immune response to the vaccine.” However, “it’s important to note that overall safety and efficacy profile will not be sacrificed with this approach.”
In Related News:
Protesters interrupted a symposium about the monkeypox outbreak during AIDS 2022, held in Montreal July 29 to Aug 2. The protesters took the stage and the podium to urge better distribution of monkeypox vaccine, especially in developing countries.
They criticized the Biden administration and the CDC for its slow response. The protest started right after Geneviève Bergeron, MD, MPH, of Montreal’s public health department, announced that anyone who was attending the AIDS 2022 conference could receive a free vaccine. Montreal has more than 300 cases as of Aug. 5.
Immediately following the protest, Demetre Daskalakis, MD, who was recently named the White House National Monkeypox Response Deputy Coordinator, resumed the symposium, thanking the protesting for their comments and assuring them they were heard. However, he did not address any of their concerns. Instead, he gave the original presentation he planned, about how well public health officials in the United States were communicating with the LBGTQ+ community.